Medical Device Registration Changes CIS
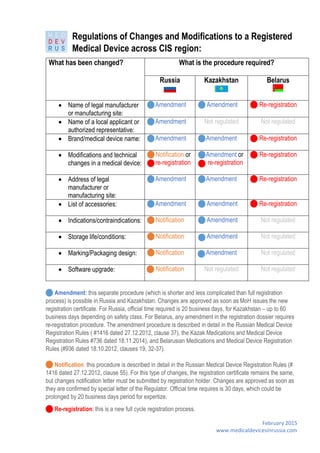
- 1. February 2015 www.medicaldevicesinrussia.com Regulations of Changes and Modifications to a Registered Medical Device across CIS region: Amendment: this separate procedure (which is shorter and less complicated than full registration process) is possible in Russia and Kazakhstan. Changes are approved as soon as MoH issues the new registration certificate. For Russia, official time required is 20 business days, for Kazakhstan – up to 60 business days depending on safety class. For Belarus, any amendment in the registration dossier requires re-registration procedure. The amendment procedure is described in detail in the Russian Medical Device Registration Rules ( #1416 dated 27.12.2012, clause 37), the Kazak Medications and Medical Device Registration Rules #736 dated 18.11.2014), and Belarusian Medications and Medical Device Registration Rules (#936 dated 18.10.2012, clauses 19, 32-37). Notification: this procedure is described in detail in the Russian Medical Device Registration Rules (# 1416 dated 27.12.2012, clause 55). For this type of changes, the registration certificate remains the same, but changes notification letter must be submitted by registration holder. Changes are approved as soon as they are confirmed by special letter of the Regulator. Official time requires is 30 days, which could be prolonged by 20 business days period for expertize. Re-registration: this is a new full cycle registration process. What has been changed? What is the procedure required? Russia Kazakhstan Belarus Name of legal manufacturer or manufacturing site: Amendment Amendment Re-registration Name of a local applicant or authorized representative: Amendment Not regulated Not regulated Brand/medical device name: Amendment Amendment Re-registration Modifications and technical changes in a medical device: Notification or re-registration Amendment or re-registration Re-registration Address of legal manufacturer or manufacturing site: Amendment Amendment Re-registration List of accessories: Amendment Amendment Re-registration Indications/contraindications: Notification Amendment Not regulated Storage life/conditions: Notification Amendment Not regulated Marking/Packaging design: Notification Amendment Not regulated Software upgrade: Notification Not regulated Not regulated
