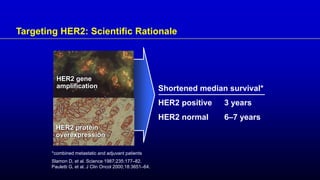
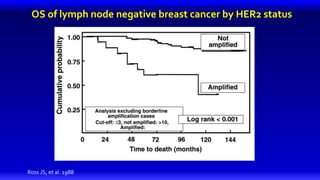
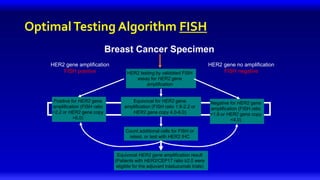
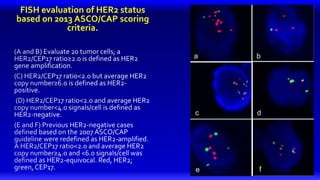
1) HER2 positive breast cancer accounts for around 15-20% of cases and has a poorer prognosis than other subtypes without HER2 targeted therapy.
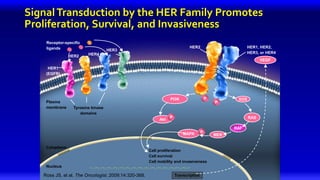
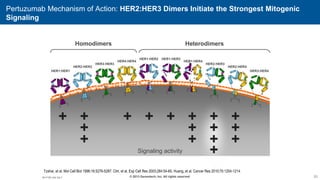
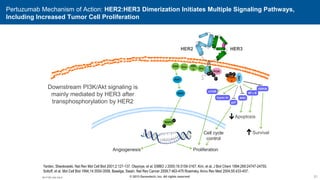
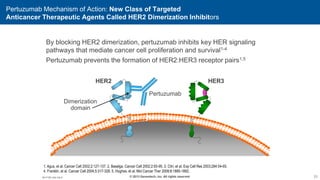
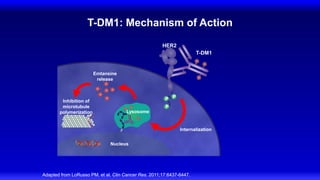
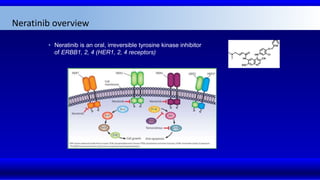
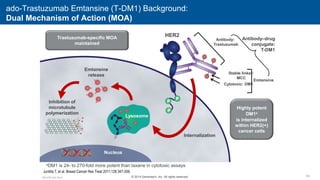
2) Multiple HER2 targeted agents are available including trastuzumab, lapatinib, pertuzumab, T-DM1, neratinib, and tucatinib which inhibit HER2 signaling through different mechanisms such as antibody binding or tyrosine kinase inhibition.
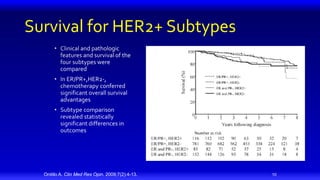
3) Combining HER2 targeted therapies such as trastuzumab with chemotherapy improves outcomes for patients with metastatic HER2 positive breast cancer compared to chemotherapy alone.







![15
© 2013 Genentech, Inc. All rights reserved.
The HER2-Targeted Therapeutic:
Trastuzumab, a Humanized Anti-HER2 mAb
Targets HER2 protein
Selectively binds with high
affinity
HER2 epitopes recognized by
hypervariable murine sequences
Human
IgG1
Carter P, et al. Proc Natl Acad Sci U S A 1992;89:4285-4289. Herceptin® (trastuzumab) [prescribing information]. South San Francisco, CA:
Genentech, Inc.; 2010.
mAb=monoclonal antibody
M1.H.BC.Adv.Ow.17](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-15-320.jpg)
![16
© 2013 Genentech, Inc. All rights reserved.
The HER2-Targeted Therapeutic: Trastuzumab MOA
Humanized monoclonal
antibody specific for
HER2
Targets HER2 protein-
overexpressing cells
Proposed MOA based
on preclinical studies
● Extracellular
● Intracellular
Dimerized
HER2
receptors
signal tumor
cells to
proliferate
Extracellular
trastuzumab
binds to subdomain
IV of HER2 receptors
on tumor cells,
flagging them for
destruction by the
immune system
Intracellular
trastuzumab
blocks HER2
signaling to
inhibit
proliferation
of tumor cells
Arnould L, et al. Br J Cancer 2006;94:259-267. Bianco AR. J Chemother
2004;16(suppl 4):52-54. Harari D, Yarden Y. Oncogene 2000;19;6102-6114.
Lewis GD, et al. Cancer Immunol Immunother 1993;37:255-263.
Sliwkowski MX, et al. Semin Oncol 1999;26(suppl 12):60-70. Yakes FM, et al. Cancer Res 2002;62:4132-4141. Yarden Y. Oncology 2001;61(suppl 2):1-13.
Herceptin® (trastuzumab) [prescribing information]. South San Francisco, CA: Genentech, Inc.; 2010.
MOA=mechanism of action](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-16-320.jpg)
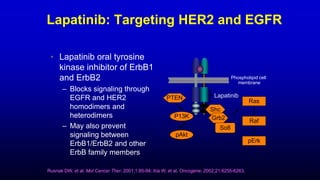
![Lapatinib
Drug Profile
• Belongs to the 4-
anilinoquinazoline class of
tyrosine kinase inhibitors
• Binds reversibly to the
cytoplasmic ATP-binding site of
the kinase, thereby preventing
receptor phosphorylation and
activation
• Works intracellularly
N-{3-Chloro-4-[(3-fluorobenzyl)oxy]phenyl}-6-
[5-({[2(methylsulfonyl)ethyl]amino}methyl)-2-furyl]-4-
quinazolinamine
Lapatinib is the first-in-class oral small-molecule
inhibitor HER2 tyrosine kinase:
Xia W, et al. Oncogene. 2002;21:6255-6263.](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-17-320.jpg)



![27
© 2013 Genentech, Inc. All rights reserved.
The HER2-Targeted Therapeutic:
Trastuzumab, a Humanized Anti-HER2 mAb
Targets HER2 protein
Selectively binds with high
affinity
HER2 epitopes recognized by
hypervariable murine sequences
Human
IgG1
Carter P, et al. Proc Natl Acad Sci U S A 1992;89:4285-4289. Herceptin® (trastuzumab) [prescribing information]. South San Francisco, CA:
Genentech, Inc.; 2010.
mAb=monoclonal antibody
M1.H.BC.Adv.Ow.17](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-27-320.jpg)

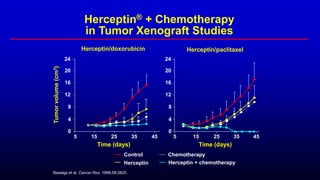
![AC=doxorubicin (60mg/m2) [or epirubicin (75mg/m2)] + cyclophosphamide (600mg/m2) q3w for 6 cycles;
Taxol (175mg/m2 x 3 h) q3w for 6 cycles; Herceptin (4mg/kg) loading dose, 2mg/kg qw until progression;
CT=chemotherapy; KPS=Karnofsky Performance Score; q3w=every three weeks; qw=weekly
No prior adjuvant AC Prior adjuvant AC
Taxol
(n=96)
Herceptin +
Taxol (n=92)
AC
(n=138)
Herceptin +
AC (n=143)
Stratify
Randomize Randomize
Slamon D, et al. N Engl J Med 2001;344:783.
n=469
MBC
HER2+
No prior CT for MBC
Measurable disease
KPS 60%
Herceptin Combination Pivotal Trial in
First-line MBC: Schema](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-30-320.jpg)
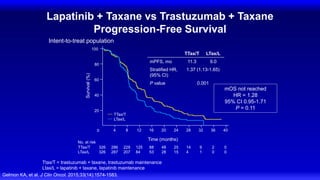
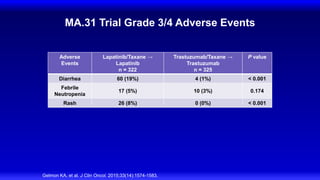
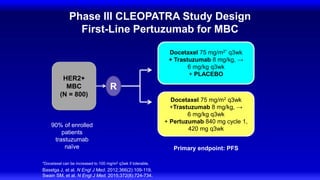
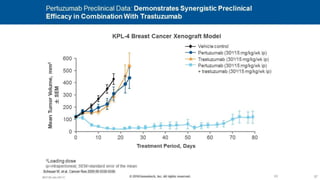
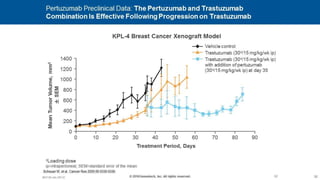
![CLEOPATRA: PFS
Investigator Assessment
Swain SM, et al. N Engl J Med. 2015;372(8):724-734.
100
90
80
70
60
50
40
30
20
10
0
PFS
(%)
Time (months)
HR 0.68, 95% CI [0.58, 0.80], P < 0.001
Pertuzumab + T + D: median = 18.7 months
Placebo + T + D: median = 12.4 months
6.3
Months
80
70
60
50
40
30
20
10
0
402
406
284
223
179
110
121
75
87
51
37
21
6
6
0
0
0
0
n at risk
T, trastuzumab; D, docetaxel.
Pertuzumab
Placebo](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-39-320.jpg)
![CLEOPATRA: OS
Swain SM, et al. N Engl J Med. 2015;372(8):724-734.
Pertuzumab +Trastuzumab + Docetaxel
Median OS: 56.5 months
Placebo + Trastuzumab + Docetaxel
Median OS: 40.8 months
HR = 0.68, 95% CI [0.56-0.84],P < 0.001
Median follow-up: 50 months (range, 0-70 months)
100
90
80
70
60
50
40
30
20
10
0
Time (months)
OS
(%)
70
60
50
40
30
20
10
0
1
28
104
226
268
318
371
402
0
23
91
179
230
289
350
406
n at risk
Δ = 15.7 months
Pertuzumab
Placebo](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-40-320.jpg)
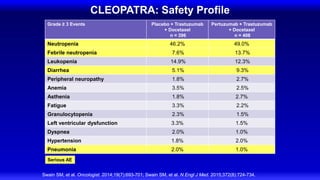

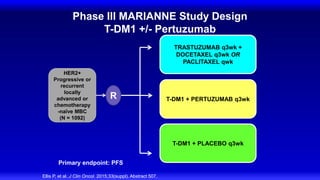


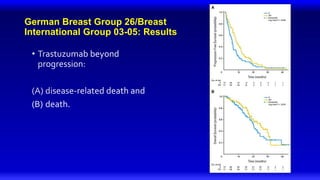
![Lapatinib
Drug Profile
• Belongs to the 4-
anilinoquinazoline class of
tyrosine kinase inhibitors
• Binds reversibly to the
cytoplasmic ATP-binding site of
the kinase, thereby preventing
receptor phosphorylation and
activation
• Works intracellularly
N-{3-Chloro-4-[(3-fluorobenzyl)oxy]phenyl}-6-
[5-({[2(methylsulfonyl)ethyl]amino}methyl)-2-furyl]-4-
quinazolinamine
Lapatinib is the first-in-class oral small-molecule
inhibitor HER2 tyrosine kinase:
Xia W, et al. Oncogene. 2002;21:6255-6263.](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-47-320.jpg)

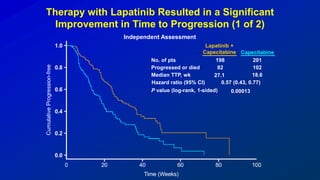


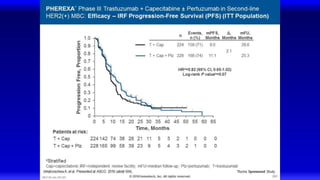
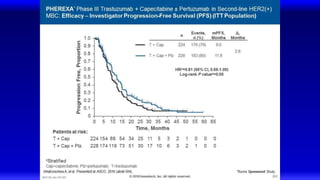

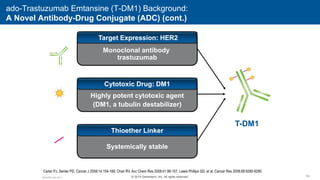

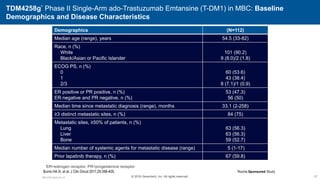
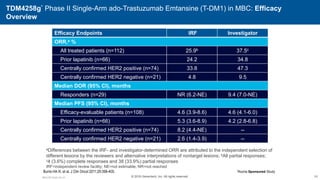
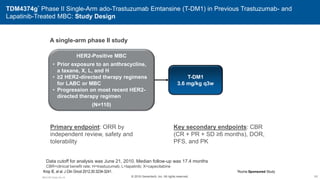
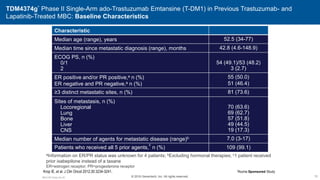


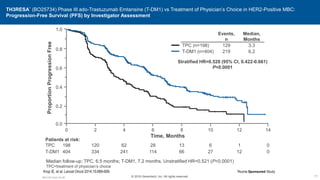
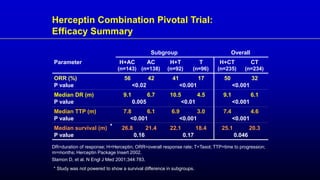
![EMILIA* (TDM4370g) Phase III ado-Trastuzumab Emtansine (T-DM1) vs Capecitabine + Lapatinib in HER2-Positive MBC:
Progression-Free Survival (PFS) by Independent Review
496 404 310 176 129 73 53 35 25 14 9 8 5 1 0 0
495 419 341 236 183 130 101 72 54 44 30 18 9 3 1 0
Cap + Lap
T-DM1
Patients at risk by independent review:
Median, Months Events, n
Cap + Lap 6.4 304
T-DM1 9.6 265
0.0
0.2
0.4
0.6
0.8
1.0
0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30
Proportion
Progression
Free
Time, Months
Verma S, et al. N Engl J Med 2012;367:1783-1791 [including supplementary appendix].
Unstratified HR=0.66 (95% CI, 0.56-0.78; P<0.0001)
Cap=capecitabine; Lap=lapatinib
Median PFS by investigator review:
9.4 for T-DM1 vs 5.8 months for Cap + Lap;
HR=0.66; P<0.001
73
Stratified HR=0.65 (95% CI, 0.55-0.77)
P<0.001
M5.K.BC.Early.Ow.46
*Roche Sponsored Study
© 2016 Genentech, Inc. All rights reserved.](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-73-320.jpg)
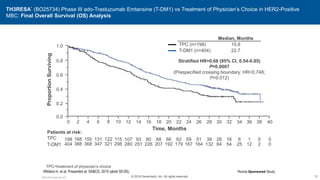
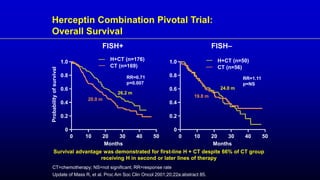
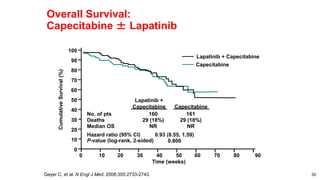
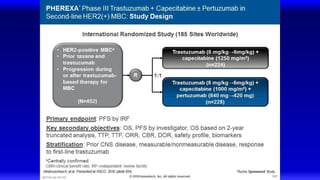
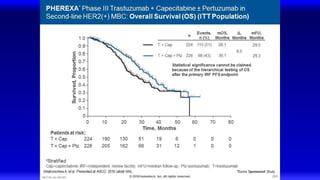
![EMILIA: Overall Survival
Verma S, et al. N Engl J Med. 2012;367(19):1783-1791.
Overall
Survival
(%)
100
80
60
40
20
0
0 6 12 18 24 30
4 10 16 22 28
2 8 14 20 26
Months
No. at Risk
Lapatinib–
capecitabine
T-DM1
496
495
471
485
453
474
435
457
403
439
368
418
297
349
240
293
204
242
159
197
133
164
110
136
86
111
45
62
63
86
27
38
17
28
7
13
32 34 36
4
5
85.2% (95% CI, 82.0-88.5)
64.7% (95% CI, 59.3-70.2)
Lapatinib–Capecitabine
T-DM1
78.4% (95% CI, 74.6-82.3)
51.8% (95% CI, 45.9-57.7)
Median No.
of Months
No. of
Events
Lapatinib-Capecitabine 25.1 182
T-DM1 30.9 149
Stratified hazard ratio 0.68 95% CI [0.55-0.85]
P < 0.001
Efficacy stopping boundary P = 0.0037](https://image.slidesharecdn.com/metastaticher2garcia-220722225510-635bf9d4/85/Metastatic-HER2-Garcia-pptx-74-320.jpg)