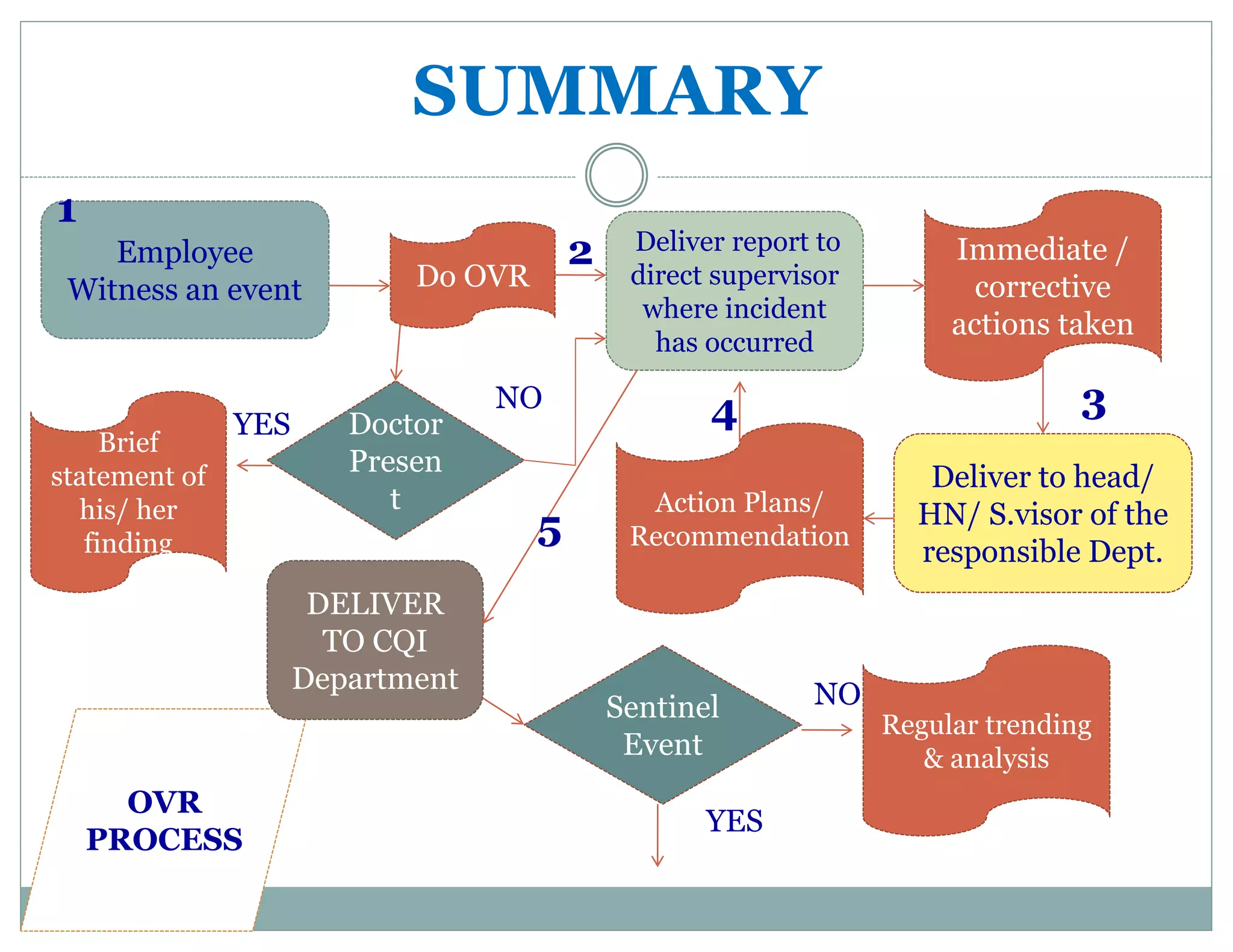
This document provides information about quality management and occurrence variance reporting (OVR) processes. It defines key terms like quality, adverse events, near misses and sentinel events. It outlines the OVR process which involves any staff member witnessing and reporting an event, investigation by relevant departments, and submission to the continuous quality improvement department for trend analysis. The purpose of OVR is to identify areas for improvement, implement corrective actions, and establish preventative measures through a non-blame approach.