8 hole 16 a
•Download as PPTX, PDF•
0 likes•436 views
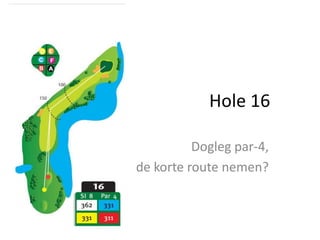
Wim laat zien hoe een van de nieuwere holes het best bespeeld kan worden
Report
Share
Report
Share
Recommended
Cpcu 530 chapter 1 cases

This document summarizes four Supreme Court cases related to education, criminal procedure, corporate law, and insurance regulation. Brown v Board of Education declared segregation in public schools unconstitutional. Miranda v Arizona established that suspects must be informed of their rights and inadmissible confessions cannot be used as evidence. Paul v Virginia ruled that corporations are not citizens under the Privileges and Immunities Clause and that insurance is not interstate commerce. South-Eastern Underwriters overturned Paul v Virginia, finding that insurance transactions are subject to federal regulation under the Commerce Clause.
Pepug 65 integracja lync i cisco

Podstawy integracji Lync z systemami Cisco w zakresie integracji głosowej i video
FDA Social Media Review From Dose Of Digital

A handful of new information came out at the FDA pharma social media hearings, but quite a bit was missing as well. Here's my brief overview of what I learned and what I thought was missing
Zarządzanie użytkownikami i komputerami w firmie - dlaczego warto znać Active...

Dlaczego warto podnosić kwalifikacje i poznać Active Directory, krótkie wprowadzenie do AD
FDA Form 483 (Inspectional Observations) - Top Violations 2013

FDA Attorney Marc Sanchez discusses the Top FDA Form 483 (Inspectional Observations) from fiscal year 2013. The top observations provide a foundation for preparing for a pre-inspection, for-cause inspection, or routine surveillance inspection.
3 top ten fda warning letter findings by the

The document outlines the top ten most common Good Manufacturing Practice (GMP) findings by the FDA in fiscal year 2003 based on Form 483 observations. The most frequent finding was a failure to follow and/or document written production and process control procedures, which was referenced on 317 Form 483s. Other top findings included responsibilities of the quality control unit, written procedures for production and process controls, testing and release for distribution, and validation. The document also provides examples of common GMP violations found during inspections, such as dirty facilities, unqualified equipment, and lack of corrective and preventative action procedures.
FDA 483 observations in the lab

Real life FDA 483 observations in the lab that you should avoid. If you have a FDA Inspection scheduled, this presentation is for you.
Recommended
Cpcu 530 chapter 1 cases

This document summarizes four Supreme Court cases related to education, criminal procedure, corporate law, and insurance regulation. Brown v Board of Education declared segregation in public schools unconstitutional. Miranda v Arizona established that suspects must be informed of their rights and inadmissible confessions cannot be used as evidence. Paul v Virginia ruled that corporations are not citizens under the Privileges and Immunities Clause and that insurance is not interstate commerce. South-Eastern Underwriters overturned Paul v Virginia, finding that insurance transactions are subject to federal regulation under the Commerce Clause.
Pepug 65 integracja lync i cisco

Podstawy integracji Lync z systemami Cisco w zakresie integracji głosowej i video
FDA Social Media Review From Dose Of Digital

A handful of new information came out at the FDA pharma social media hearings, but quite a bit was missing as well. Here's my brief overview of what I learned and what I thought was missing
Zarządzanie użytkownikami i komputerami w firmie - dlaczego warto znać Active...

Dlaczego warto podnosić kwalifikacje i poznać Active Directory, krótkie wprowadzenie do AD
FDA Form 483 (Inspectional Observations) - Top Violations 2013

FDA Attorney Marc Sanchez discusses the Top FDA Form 483 (Inspectional Observations) from fiscal year 2013. The top observations provide a foundation for preparing for a pre-inspection, for-cause inspection, or routine surveillance inspection.
3 top ten fda warning letter findings by the

The document outlines the top ten most common Good Manufacturing Practice (GMP) findings by the FDA in fiscal year 2003 based on Form 483 observations. The most frequent finding was a failure to follow and/or document written production and process control procedures, which was referenced on 317 Form 483s. Other top findings included responsibilities of the quality control unit, written procedures for production and process controls, testing and release for distribution, and validation. The document also provides examples of common GMP violations found during inspections, such as dirty facilities, unqualified equipment, and lack of corrective and preventative action procedures.
FDA 483 observations in the lab

Real life FDA 483 observations in the lab that you should avoid. If you have a FDA Inspection scheduled, this presentation is for you.
OGC sociale media maart 2011

Introductie in Sociale Media voor de Golfclub Oosterhout, gegeven op 8 en 22 maart.
9 Community René

René legt uit dat er voor de Twilight spelers meerdere mogelijkheden zijn om met elkaar af te spreken, via de website of via sociale netwerken
6 golfschool Wim

Wim laat zien wat er in zijn 'golftas' zit. Medewerkers, stagiaires en de mogelijkheden voor lessen passeren de revue!
5 wedstrijdcie Vincent

Vincent laat zien hoe je je kunt inschrijven voor de Oranjewedstrijd en vertelt verder over wat de wedstrijdcommissie allemaal doet.
3 HRC Jan

Jan vertelt wat de Handicap en Regelcommissie allemaal doet. Het is de basis van de Golfsport, en er zijn een paar regels speciaal voor de OGC!
1a OGC organisatie Nico

Nico laat zien hoe je je inschrijft om een rondje te lopen, en maakt duidelijk wat de organisatie allemaal te bieden heeft.
1 Welkom twilight spelers René

René vertelt iets over de achtergronden van de Oosterhoutse, en loopt kort door de agenda. Ook komen de diverse communicatiemiddelen aan bod.
More Related Content
More from Oosterhoutse Golfclub
OGC sociale media maart 2011

Introductie in Sociale Media voor de Golfclub Oosterhout, gegeven op 8 en 22 maart.
9 Community René

René legt uit dat er voor de Twilight spelers meerdere mogelijkheden zijn om met elkaar af te spreken, via de website of via sociale netwerken
6 golfschool Wim

Wim laat zien wat er in zijn 'golftas' zit. Medewerkers, stagiaires en de mogelijkheden voor lessen passeren de revue!
5 wedstrijdcie Vincent

Vincent laat zien hoe je je kunt inschrijven voor de Oranjewedstrijd en vertelt verder over wat de wedstrijdcommissie allemaal doet.
3 HRC Jan

Jan vertelt wat de Handicap en Regelcommissie allemaal doet. Het is de basis van de Golfsport, en er zijn een paar regels speciaal voor de OGC!
1a OGC organisatie Nico

Nico laat zien hoe je je inschrijft om een rondje te lopen, en maakt duidelijk wat de organisatie allemaal te bieden heeft.
1 Welkom twilight spelers René

René vertelt iets over de achtergronden van de Oosterhoutse, en loopt kort door de agenda. Ook komen de diverse communicatiemiddelen aan bod.
More from Oosterhoutse Golfclub (11)
8 hole 16 a
- 1. Hole 16 Dogleg par-4, de korte route nemen?
- 2. Het teeshot iets links De volgende slag is gemakkelijker door te spelen.
- 3. Visueel focussen Teeshot naar links Teeshot over rechts
- 4. De approach via rechts Dit komt door de bunker aan de linkerzijde.
- 5. Approach via links Approach via rechts
