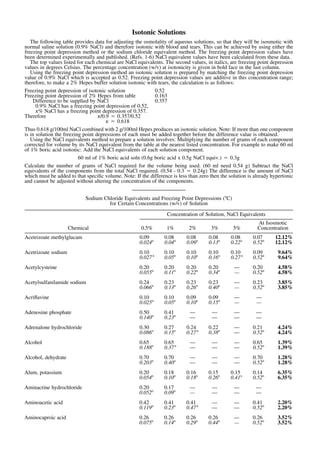
tabla de equivalentes NaCl y punto crioscópico
- 1. 40270 -- Merck BATCH LEFT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions The following table provides data for adjusting the osmolality of aqueous solutions, so that they will be isosmotic with normal saline solution (0.9% NaCl) and therefore isotonic with blood and tears. This can be achieved by using either the freezing point depression method or the sodium chloride equivalent method. The freezing point depression values have been determined experimentally and published. (Refs. 1-6) NaCl equivalent values have been calculated from these data. The top values listed for each chemical are NaCl equivalents. The second values, in italics, are freezing point depression values in degrees Celsius. The percentage concentration (w/v) at isotonicity is given in bold face in the last column. Using the freezing point depression method an isotonic solution is prepared by matching the freezing point depression value of 0.9% NaCl which is accepted as 0.52. Freezing point depression values are additive in this concentration range; therefore, to make a 2% Hepes buffer solution isotonic with tears, the calculation is as follows: Freezing point depression of isotonic solution 0.52 Freezing point depression of 2% Hepes from table 0.163 Difference to be supplied by NaCl 0.357 0.9% NaCl has a freezing point depression of 0.52, x% NaCl has a freezing point depression of 0.357. Therefore x/0.9 ⳱ 0.357/0.52 x ⳱ 0.618 Thus 0.618 g/100ml NaCl combined with 2 g/100ml Hepes produces an isotonic solution. Note: If more than one component is in solution the freezing point depressions of each must be added together before the difference value is obtained. Using the NaCl equivalents method to prepare a solution involves: Multiplying the number of grams of each component corrected for volume by its NaCl equivalent from the table at the nearest listed concentration. For example to make 60 ml of 1% boric acid isotonic: Add the NaCl equivalents of each solution component. 60 ml of 1% boric acid soln (0.6g boric acid x 0.5g NaCl equiv.) ⳱ 0.3g Calculate the number of grams of NaCl required for the volume being used. (60 ml need 0.54 g) Subtract the NaCl equivalents of the components from the total NaCl required. (0.54 - 0.3 ⳱ 0.24g) The difference is the amount of NaCl which must be added to that specific volume. Note: If the difference is less than zero then the solution is already hypertonic and cannot be adjusted without altering the concentration of the components. Sodium Chloride Equivalents and Freezing Point Depressions (⬚C) for Certain Concentrations (w/v) of Solution Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Acetrizoate methylglucam 0.09 0.08 0.08 0.08 0.08 0.07 12.12% 0.024⬚ 0.04⬚ 0.09⬚ 0.13⬚ 0.22⬚ 0.52⬚ 12.12% Acetrizoate sodium 0.10 0.10 0.10 0.10 0.10 0.09 9.64% 0.027⬚ 0.05⬚ 0.10⬚ 0.16⬚ 0.27⬚ 0.52⬚ 9.64% Acetylcysteine 0.20 0.20 0.20 0.20 — 0.20 4.58% 0.055⬚ 0.11⬚ 0.22⬚ 0.34⬚ — 0.52⬚ 4.58% Acetylsulfanilamide sodium 0.24 0.23 0.23 0.23 — 0.23 3.85% 0.066⬚ 0.13⬚ 0.26⬚ 0.40⬚ — 0.52⬚ 3.85% Acriflavine 0.10 0.10 0.09 0.09 — — 0.025⬚ 0.05⬚ 0.10⬚ 0.15⬚ — — Adenosine phosphate 0.50 0.41 — — — — 0.140⬚ 0.23⬚ — — — — Adrenalone hydrochloride 0.30 0.27 0.24 0.22 — 0.21 4.24% 0.086⬚ 0.15⬚ 0.27⬚ 0.38⬚ — 0.52⬚ 4.24% Alcohol 0.65 0.65 — — — 0.65 1.39% 0.188⬚ 0.37⬚ — — — 0.52⬚ 1.39% Alcohol, dehydrate 0.70 0.70 — — — 0.70 1.28% 0.203⬚ 0.40⬚ — — — 0.52⬚ 1.28% Alum, potassium 0.20 0.18 0.16 0.15 0.15 0.14 6.35% 0.054⬚ 0.10⬚ 0.18⬚ 0.26⬚ 0.41⬚ 0.52⬚ 6.35% Aminacrine hydrochloride 0.20 0.17 — — — — 0.052⬚ 0.09⬚ — — — — Aminoacetic acid 0.42 0.41 0.41 — — 0.41 2.20% 0.119⬚ 0.23⬚ 0.47⬚ — — 0.52⬚ 2.20% Aminocaproic acid 0.26 0.26 0.26 0.26 — 0.26 3.52% 0.075⬚ 0.14⬚ 0.29⬚ 0.44⬚ — 0.52⬚ 3.52%
- 2. 40270 -- Merck BATCH RIGHT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration p-Aminohippuric acid 0.13 0.13 — — — — 0.035⬚ 0.07⬚ — — — — p-Aminosalicylate sodium 0.30 0.29 0.29 0.28 — 0.27 3.27% 0.086⬚ 0.16⬚ 0.32⬚ 0.47⬚ — 0.52⬚ 3.27% Ammonium carbonate 0.70 0.70 — — — 0.70 1.29% 0.202⬚ 0.40⬚ — — — 0.52⬚ 1.29% Ammonium chloride 1.10 — — — — 1.07 0.84% 0.315⬚ — — — — 0.52⬚ 0.84% Ammonium lactate 0.33 0.33 0.33 — — 0.33 2.76% 0.093⬚ 0.18⬚ 0.37⬚ — — 0.52⬚ 2.76% Ammonium nitrate 0.69 0.69 — — — 0.69 1.30% 0.200⬚ 0.40⬚ — — — 0.52 1.30% Ammonium phosphate, dibasic 0.58 0.55 — — — 0.51 1.76% 0.165⬚ 0.31⬚ — — — 0.52⬚ 1.76% Ammonium sulfate 0.55 0.55 — — — 0.54 1.68% 0.158⬚ 0.31⬚ — — — 0.52⬚ 1.68% Antimony potassium tartrate 0.22 0.18 0.15 0.13 0.10 — 0.065⬚ 0.10⬚ 0.17⬚ 0.23⬚ 0.33⬚ — Antipyrine 0.18 0.17 0.16 0.14 0.14 0.13 6.81% 0.050⬚ 0.09⬚ 0.17⬚ 0.25⬚ 0.39⬚ 0.52⬚ 6.81% Arecoline hydrobromide 0.030 0.27 0.25 0.24 — 0.23 3.88% 0.084⬚ 0.15⬚ 0.28⬚ 0.41⬚ — 0.52⬚ 3.88% Arginine glutamate 0.17 0.17 0.17 0.17 0.17 0.17 5.37% 0.048⬚ 0.09⬚ 0.19⬚ 0.29⬚ 0.48⬚ 0.52⬚ 5.37% L-Arginine hydrochloride 0.31 0.30 0.28 0.27 — 0.26 3.43% 0.087⬚ 0.17⬚ 0.32⬚ 0.46⬚ — 0.52⬚ 3.43% Arsenic trioxide 0.30 0.30 — — — — 0.085⬚ 0.16⬚ — — — — Ascorbic acid 0.20 0.18 0.18 0.18 0.18 0.18 5.94% 0.053⬚ 0.10⬚ 0.20⬚ 0.31⬚ 0.51⬚ 0.52⬚ 5.94% Atropine methylnitrate 0.20 0.18 0.16 0.15 0.14 0.14 6.52% 0.055⬚ 0.10⬚ 0.18⬚ 0.26⬚ 0.41⬚ 0.52⬚ 6.52% Atropine sulfate 0.14 0.13 0.12 0.11 0.11 0.10 8.85% 0.039⬚ 0.07⬚ 0.13⬚ 0.19⬚ 0.31⬚ 0.52⬚ 8.85% Aurothioglucose 0.03 0.03 0.03 0.03 0.03 — 0.007⬚ 0.01⬚ 0.02⬚ 0.04⬚ 0.07⬚ — Barbital sodium 0.32 0.30 0.29 0.29 — 0.29 3.12% 0.087⬚ 0.17⬚ 0.33⬚ 0.50⬚ — 0.52⬚ 3.12% Benzalkonium chloride 0.18 0.16 0.15 0.14 0.13 — 0.048⬚ 0.09⬚ 0.17⬚ 0.24⬚ 0.38⬚ — Benzethonium chloride 0.08 0.05 0.03 0.02 0.02 — 0.022⬚ 0.02⬚ 0.03⬚ 0.04⬚ 0.05⬚ — Benzyl alcohol 0.18 0.17 0.16 0.15 — — 0.049⬚ 0.09⬚ 0.18⬚ 0.26⬚ — — Bismuth potassium tartrate 0.10 0.09 0.07 0.06 0.05 — 0.033⬚ 0.05⬚ 0.08⬚ 0.10⬚ 0.14⬚ — Bismuth sodium tartrate 0.14 0.13 0.13 0.12 0.11 0.10 8.91% 0.041⬚ 0.07⬚ 0.13⬚ 0.19⬚ 0.31⬚ 0.52⬚ 8.91%
- 3. 40270 -- Merck BATCH LEFT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Boric acid 0.52 0.50 — — — 0.47 1.9% 0.146⬚ 0.28⬚ — — — 0.52⬚ 1.9% Bromodiphenhydramine hydrochloride 0.20 0.17 0.14 0.10 0.07 — 0.067⬚ 0.10⬚ 0.16⬚ 0.18⬚ 0.20⬚ — Butabarbital sodium 0.27 0.27 0.27 0.27 — 0.27 3.33% 0.078⬚ 0.15⬚ 0.31⬚ 0.47⬚ — 0.52⬚ 3.33% Caffeine 0.08 0.08 — — — — 0.025⬚ 0.04⬚ — — — — Calcium aminosalicylate 0.30 0.27 0.23 0.21 — — 0.091⬚ 0.15⬚ 0.26⬚ 0.36⬚ — — Calcium chloride dihydrate 0.50 0.51 — — — 0.53 1.70% 0.145⬚ 0.29⬚ — — — 0.52⬚ 1.70% Calcium chloride hexahydrate 0.34 0.35 0.36 — — 0.36 2.5% 0.097⬚ 0.20⬚ 0.41⬚ — — 0.52⬚ 2.5% Calcium chloride, anhydrous 0.70 0.70 — — — 0.70 1.29% 0.206⬚ 0.40⬚ — — — 0.52⬚ 1.29% Calcium disodium edetate 0.21 0.21 0.21 0.20 — 0.20 4.50% 0.061⬚ 0.12⬚ 0.24⬚ 0.35⬚ — 0.52⬚ 4.50% Calcium gluconate 0.18 0.16 0.15 0.14 — — 0.050⬚ 0.09⬚ 0.16⬚ 0.23⬚ — — Calcium lactate 0.26 0.23 0.22 0.21 — 0.20 4.5% 0.073⬚ 0.13⬚ 0.25⬚ 0.37⬚ — 0.52⬚ 4.5% Chiniofon 0.14 0.13 0.12 0.11 — — 0.039⬚ 0.07⬚ 0.13⬚ 0.20⬚ — — Chloramine-T 0.24 0.23 0.22 0.22 — 0.22 4.1% 0.064⬚ 0.12⬚ 0.25⬚ 0.38⬚ — 0.52⬚ 4.1% Chlorobutanol, hydrated 0.24 — — — — — — 0.071⬚ — — — — — — Chlorophyll 0.14 0.10 0.08 0.06 0.05 — 0.037⬚ 0.05⬚ 0.08⬚ 0.11⬚ 0.15⬚ — Citric acid 0.18 0.18 0.17 0.17 0.16 0.16 5.52% 0.050⬚ 0.09⬚ 0.19⬚ 0.28⬚ 0.47⬚ 0.52⬚ 5.52% Congo red 0.05 0.05 0.05 0.05 0.05 — 0.015⬚ 0.03⬚ 0.05⬚ 0.09⬚ 0.15⬚ — Cromolyn sodium 0.16 0.14 0.11 0.09 0.05 0.046⬚ 0.08⬚ 0.12⬚ 0.14⬚ 0.15⬚ Cupric sulfate 0.20 0.18 0.16 0.15 0.14 0.13 6.85% 0.054⬚ 0.09⬚ 0.17⬚ 0.25⬚ 0.39⬚ 0.52⬚ 6.85% Cupric sulfate, anhydrous 0.30 0.27 0.25 0.23 — 0.22 4.09% 0.084⬚ 0.15⬚ 0.28⬚ 0.39⬚ — 0.52⬚ 4.09% Cyclophosphamide 0.10 0.10 0.10 — — — 0.031⬚ 0.06⬚ 0.12⬚ — — — Dextrose 0.16 0.16 0.16 0.16 0.16 0.16 5.51% 0.045⬚ 0.09⬚ 0.18⬚ 0.27⬚ 0.47⬚ 0.52⬚ 5.51% Dextrose, anhydrous 0.18 0.18 0.18 0.18 0.18 0.18 5.05% 0.050⬚ 0.10⬚ 0.20⬚ 0.31⬚ 0.51⬚ 0.52⬚ 5.05% Diatrizoate sodium 0.10 0.09 0.09 0.09 0.09 0.09 10.55% 0.025⬚ 0.04⬚ 0.09⬚ 0.14⬚ 0.24⬚ 0.52⬚ 10.55%
- 4. 40270 -- Merck BATCH RIGHT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Diethanolamine 0.31 0.31 0.31 — — 0.31 2.90% 0.089⬚ 0.17⬚ 0.35⬚ — — 0.52⬚ 2.90% Dimethyl sulfoxide 0.42 0.42 0.42 — — 0.42 2.16% 0.122⬚ 0.24⬚ 0.48⬚ — — 0.52⬚ 2.16% Disodium edetate 0.24 0.23 0.22 0.21 — 0.20 4.44% 0.070⬚ 0.13⬚ 0.24⬚ 0.36⬚ — 0.52⬚ 4.44% Dopamine hydrochloride 0.30 0.30 0.29 0.29 — 0.29 3.11% 0.085⬚ 0.17⬚ 0.33⬚ 0.50⬚ — 0.52⬚ 3.11% Ephedrine hydrochloride 0.32 0.30 0.29 0.28 — 0.28 3.2% 0.087⬚ 0.16⬚ 0.33⬚ 0.48⬚ — 0.52⬚ 3.2% Ephedrine lactate 0.28 0.26 0.25 0.24 — 0.24 3.72% 0.075⬚ 0.14⬚ 0.28⬚ 0.42⬚ — 0.52⬚ 3.72% Ephedrine sulfate 0.24 0.23 0.22 0.20 — 0.20 4.54% 0.070⬚ 0.13⬚ 0.24⬚ 0.35⬚ — 0.52⬚ 4.54% Epinephrine bitartrate 0.18 0.18 0.17 0.16 0.16 0.16 5.7% 0.050⬚ 0.09⬚ 0.19⬚ 0.28⬚ 0.45⬚ 0.52⬚ 5.7% Epinephrine hydrochloride 0.30 0.29 0.27 0.26 — 0.26 3.47% 0.088⬚ 0.16⬚ 0.31⬚ 0.45⬚ — 0.52⬚ 3.47% Ergonovine maleate 0.20 0.16 0.13 — — — 0.055⬚ 0.08⬚ 0.14⬚ — — — Ethylenediamine 0.46 0.44 0.43 — — — 0.130⬚ 0.25⬚ 0.50⬚ — — — Evans blue 0.06 0.06 0.06 0.05 0.05 — 0.017⬚ 0.03⬚ 0.06⬚ 0.09⬚ 0.14⬚ — Ferric ammonium citrate, green 0.18 0.17 0.16 0.15 0.14 — 0.054⬚ 0.09⬚ 0.17⬚ 0.25⬚ 0.39⬚ — Ferric cacodylate 0.10 0.09 0.08 — — — 0.023⬚ 0.04⬚ 0.09⬚ — — — Ferrous gluconate 0.16 0.15 0.14 0.12 0.11 — 0.048⬚ 0.08⬚ 0.15⬚ 0.21⬚ 0.33⬚ — Ferrous lactate 0.22 0.21 0.21 — — — 0.062⬚ 0.12⬚ 0.23⬚ — — — Fluorescein sodium 0.36 0.31 0.29 0.27 — 0.27 3.34% 0.099⬚ 0.18⬚ 0.33⬚ 0.47⬚ — 0.52⬚ 3.34% Fluorouracil 0.16 0.13 — — — — 0.045⬚ 0.07⬚ — — — — Folinic acid-SF calcium 0.06 0.05 0.05 0.04 0.04 — 0.013⬚ 0.02⬚ 0.05⬚ 0.07⬚ 0.12⬚ — D-Fructose 0.18 0.18 0.18 0.18 0.18 0.18 5.05% 0.050⬚ 0.10⬚ 0.20⬚ 0.31⬚ 0.51⬚ 0.52⬚ 5.05% Galactose, anhydrous 0.18 0.18 0.18 0.18 — 0.18 4.92% 0.053⬚ 0.10⬚ 0.21⬚ 0.31⬚ — 0.52⬚ 4.92% Gentamicin sulfate 0.05 0.05 0.05 0.05 0.05 — 0.015⬚ 0.03⬚ 0.06⬚ 0.09⬚ 0.15⬚ — Glucoheptonate calcium 0.12 0.12 0.11 0.10 0.10 — 0.037⬚ 0.06⬚ 0.12⬚ 0.17⬚ 0.27⬚ — Glucosulfone sodium 0.18 0.16 0.14 0.13 0.13 — 0.049⬚ 0.08⬚ 0.16⬚ 0.23⬚ 0.36⬚ —
- 5. 40270 -- Merck BATCH LEFT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration D-Glucuronic acid 0.20 0.20 0.19 0.19 0.18 0.18 5.02% 0.061⬚ 0.11⬚ 0.22⬚ 0.32⬚ 0.51⬚ 0.52⬚ 5.02% L-Glutamic acid 0.25 0.25 0.25 — — — 0.070⬚ 0.14⬚ 0.29⬚ — — — Glycerin 0.36 0.35 0.35 — — 0.35 2.6% 0.104⬚ 0.20⬚ 0.40⬚ — — 0.52⬚ 2.6% Glycine 0.41 0.41 0.41 — — 0.41 2.19% 0.118⬚ 0.23⬚ 0.47⬚ — — 0.52⬚ 2.19% Gold sodium thiomalate 0.10 0.10 0.10 0.09 0.09 — 0.032⬚ 0.06⬚ 0.11⬚ 0.15⬚ 0.25⬚ — Guanidine hydrochloride 0.72 0.65 — — — 0.61 1.47% 0.208⬚ 0.37⬚ — — — 0.52⬚ 1.47% Heparin sodium 0.07 0.07 0.07 0.07 0.07 0.07 12.2% 0.021⬚ 0.04⬚ 0.08⬚ 0.12⬚ 0.21⬚ 0.52⬚ 12.2% Hepes 0.17 0.16 0.14 0.14 0.13 0.13 6.80% 0.048⬚ 0.08⬚ 0.16⬚ 0.23⬚ 0.38⬚ 0.52⬚ 6.80% Histamine dihydrochloride 0.40 0.40 0.40 — — 0.40 2.24% 0.115⬚ 0.23⬚ 0.46⬚ — — 0.52⬚ 2.24% Histamine phosphate 0.28 0.25 0.24 0.23 — 0.22 4.1% 0.080⬚ 0.14⬚ 0.27⬚ 0.39⬚ — 0.52⬚ 4.1% Histidine monohydrochloride 0.30 0.29 0.28 0.26 — — 0.082⬚ 0.16⬚ 0.31⬚ 0.46⬚ — — Hyaluronidase 0.01 0.01 0.01 0.01 0.01 — 0.004⬚ 0.00⬚ 0.01⬚ 0.02⬚ 0.03⬚ — Indigotindisulfonate sodium 0.30 0.30 — — — — 0.085⬚ 0.17⬚ — — — — o-Iodohippurate sodium 0.16 0.16 0.16 0.15 0.15 0.15 5.92% 0.047⬚ 0.09⬚ 0.18⬚ 0.26⬚ 0.44⬚ 0.52⬚ 5.92% Iodophthalein sodium 0.20 0.17 0.14 0.12 0.11 0.09 9.58% 0.055⬚ 0.09⬚ 0.15⬚ 0.21⬚ 0.31⬚ 0.52⬚ 9.58% Iodopyracet 0.12 0.11 0.11 0.11 0.10 0.10 9.21% 0.036⬚ 0.06⬚ 0.12⬚ 0.18⬚ 0.29⬚ 0.52⬚ 9.21% Iodopyracet diethylamine 0.14 0.12 0.12 0.11 0.11 0.10 8.73% 0.035⬚ 0.06⬚ 0.13⬚ 0.19⬚ 0.30⬚ 0.52⬚ 8.73% Iopamidol 0.03 0.03 0.03 0.03 0.03 — 0.008⬚ 0.01⬚ 0.03⬚ 0.05⬚ 0.08⬚ — Isopropyl Alcohol 0.53 0.53 — — — 0.53 1.71% 0.153⬚ 0.30⬚ — — — 0.52⬚ 1.71% Kanamycin sulfate 0.08 0.07 0.07 0.07 0.07 — 0.021⬚ 0.04⬚ 0.08⬚ 0.12⬚ 0.21⬚ — Lactic acid 0.44 0.41 0.39 — — 0.39 2.3% 0.124⬚ 0.23⬚ 0.45⬚ — — 0.52⬚ 2.3% Lactose 0.06 0.07 0.08 0.08 0.09 0.09 9.75% 0.019⬚ 0.04⬚ 0.08⬚ 0.13⬚ 0.24⬚ 0.52⬚ 9.75% Lithium carbonate 1.06 — — — — 0.98 0.92% 0.303⬚ — — — — 0.52⬚ 0.92% Lithium chloride 1.40 — — — — 1.40 0.65% 0.405⬚ — — — — 0.52⬚ 0.65%
- 6. 40270 -- Merck BATCH RIGHT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Lyapolate sodium 0.10 0.09 0.09 0.09 0.09 0.09 9.96% 0.025⬚ 0.05⬚ 0.10⬚ 0.15⬚ 0.26⬚ 0.52⬚ 9.96% Magnesium chloride 0.48 0.45 0.45 — — 0.45 2.02% 0.136⬚ 0.26⬚ 0.51⬚ — — 0.52⬚ 2.02% Magnesium sulfate 0.18 0.17 0.16 0.15 0.15 0.14 6.3% 0.049⬚ 0.09⬚ 0.17⬚ 0.26⬚ 0.41⬚ 0.52⬚ 6.3% Magnesium sulfate, anhydrous 0.34 0.32 0.30 0.29 — 0.28 3.18% 0.093⬚ 0.18⬚ 0.34⬚ 0.49⬚ — 0.52⬚ 3.18% Mannitol 0.16 0.17 0.17 0.17 0.18 0.18 5.07% 0.047⬚ 0.09⬚ 0.20⬚ 0.30⬚ 0.51⬚ 0.52⬚ 5.07% Merbromin 0.16 0.14 0.12 0.11 0.09 — 0.044⬚ 0.08⬚ 0.13⬚ 0.18⬚ 0.27⬚ — Mercuric cyanide 0.16 0.15 0.15 0.14 0.13 — 0.047⬚ 0.08⬚ 0.16⬚ 0.23⬚ 0.38⬚ — Mercury bichloride 0.14 0.13 0.12 0.12 0.10 — 0.038⬚ 0.07⬚ 0.14⬚ 0.20⬚ 0.33⬚ — Methacholine bromide 0.29 0.28 0.26 0.24 — 0.24 3.77% 0.087⬚ 0.16⬚ 0.29⬚ 0.42⬚ — 0.52 3.77% Methacholine chloride 0.34 0.32 0.30 0.28 — 0.28 3.21% 0.099⬚ 0.18⬚ 0.38⬚ 0.49⬚ — 0.52⬚ 3.21% Methenamine 0.22 0.23 0.24 0.24 — 0.24 3.68% 0.061⬚ 0.12⬚ 0.27⬚ 0.41⬚ — 0.52⬚ 3.68% Methiodal sodium 0.24 0.24 0.24 0.24 — 0.24 3.81% 0.068⬚ 0.13⬚ 0.27⬚ 0.41⬚ — 0.52⬚ 3.81% Methionine 0.32 0.28 0.25 — — — 0.091⬚ 0.16⬚ 0.28⬚ — — — Methylergonovine maleate 0.10 0.10 — — — — 0.028⬚ 0.05⬚ — — — — N-Methylglucamine 0.20 0.20 0.18 0.18 0.18 0.18 5.02% 0.057⬚ 0.11⬚ 0.21⬚ 0.31⬚ 0.51⬚ 0.52⬚ 5.02% Metrizamide 0.04 0.04 0.03 0.03 — — 0.010⬚ 0.02⬚ 0.04⬚ 0.06⬚ — — Monoethanolamine 0.53 0.53 — — — 0.53 1.70% 0.154⬚ 0.30⬚ — — — 0.52⬚ 1.70% Morphine hydrochloride 0.16 0.15 0.15 0.14 — — 0.044⬚ 0.08⬚ 0.16⬚ 0.24⬚ — — Morphine nitrate 0.22 0.19 0.16 0.15 — — 0.061⬚ 0.10⬚ 0.18⬚ 0.25⬚ — — Morphine sulfate 0.16 0.14 0.12 0.11 0.09 — 0.046⬚ 0.07⬚ 0.13⬚ 0.17⬚ 0.25⬚ — Neoarsphenamine 0.42 0.40 0.39 — — 0.39 2.32% 0.116⬚ 0.22⬚ 0.44⬚ — — 0.52⬚ 2.32% Neomycin sulfate 0.14 0.12 0.10 0.09 0.08 — 0.041⬚ 0.06⬚ 0.11⬚ 0.15⬚ 0.22⬚ — Nicotinamide 0.30 0.26 0.23 0.21 — 0.20 4.49% 0.083⬚ 0.14⬚ 0.26⬚ 0.37⬚ — 0.52⬚ 4.49% Nicotinic acid 0.26 0.25 — — — — 0.074⬚ 0.14⬚ — — — —
- 7. 40270 -- Merck BATCH LEFT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Papaverine hydrochloride 0.10 0.10 0.10 — — — 0.028⬚ 0.06⬚ 0.12⬚ — — — Paraldehyde 0.25 0.25 0.25 0.25 — 0.25 3.65% 0.071⬚ 0.14⬚ 0.28⬚ 0.43⬚ — 0.52 3.65% Phenobarbital sodium 0.24 0.24 0.23 0.23 — 0.23 3.95% 0.069⬚ 0.13⬚ 0.26⬚ 0.39⬚ — 0.52⬚ 3.95% Phenol 0.38 0.35 0.33 — — 0.32 2.8% 0.104⬚ 0.19⬚ 0.38⬚ — — 0.52⬚ 2.8% Phentolamine mesylate 0.18 0.17 0.16 0.14 0.13 0.11 8.23% 0.052⬚ 0.09⬚ 0.17⬚ 0.24⬚ 0.36⬚ 0.52⬚ 8.23% Phenylethyl alcohol 0.25 0.25 0.25 — — — 0.070⬚ 0.14⬚ 0.28⬚ — — — Polyethylene glycol 300 0.12 0.12 0.12 0.12 0.13 0.13 6.73% 0.034⬚ 0.06⬚ 0.14⬚ 0.21⬚ 0.37⬚ 0.52⬚ 6.73% Polyethylene glycol 400 0.08 0.08 0.09 0.09 0.09 0.11 8.50% 0.022⬚ 0.04⬚ 0.09⬚ 0.15⬚ 0.27⬚ 0.52⬚ 8.50% Polyethlene glycol 1500 0.06 0.06 0.07 0.07 0.07 0.09 10.00% 0.015⬚ 0.03⬚ 0.07⬚ 0.12⬚ 0.21⬚ 0.52⬚ 10.00% Polyethylene glycol 1540 0.02 0.02 0.02 0.03 0.03 — 0.005⬚ 0.01⬚ 0.02⬚ 0.04⬚ 0.09⬚ — Polyethylene glycol 4000 0.02 0.02 0.02 0.02 0.02 — 0.004⬚ 0.00⬚ 0.02⬚ 0.03⬚ 0.06⬚ — Polymyxin B sulfate 0.10 0.09 0.07 0.06 0.04 — 0.033⬚ 0.04⬚ 0.07⬚ 0.09⬚ 0.13⬚ — Polysorbate 80 0.02 0.02 0.02 0.02 0.02 — 0.005⬚ 0.01⬚ 0.02⬚ 0.03⬚ 0.05⬚ — Polyvinyl alcohol (99% hydrolyzed) 0.02 0.02 0.02 0.02 0.03 — 0.004⬚ 0.00⬚ 0.02⬚ 0.03⬚ 0.07⬚ — Potassium acetate 0.59 0.59 — — — 0.59 1.53% 0.172⬚ 0.34⬚ — — — 0.52⬚ 1.53% Potassium chlorate 0.50 0.49 — — — 0.48 1.88% 0.140⬚ 0.27⬚ — — — 0.52⬚ 1.88% Potassium chloride 0.76 0.76 — — — 0.76 1.19% 0.219⬚ 0.43⬚ — — — 0.52⬚ 1.19% Potassium iodide 0.34 0.34 0.34 — — 0.34 2.59% 0.104⬚ 0.20⬚ 0.40⬚ — — 0.52⬚ 2.59% Potassium nitrate 0.58 0.56 — — — 0.56 1.62% 0.163⬚ 0.32⬚ — — — 0.52⬚ 1.62% Potassium permanganate 0.39 0.39 0.39 — — — 0.112⬚ 0.22⬚ 0.44⬚ — — — Potassium phosphate, anhydrous 0.50 0.46 0.43 — — 0.43 2.11% 0.140⬚ 0.26⬚ 0.49⬚ — — 0.52⬚ 2.11% Potassium phosphate, monobasic 0.48 0.44 0.42 — — 0.41 2.18% 0.133⬚ 0.25⬚ 0.48⬚ — — 0.52⬚ 2.18% Potassium sorbate 0.44 0.41 0.40 — — 0.40 2.23% 0.125⬚ 0.23⬚ 0.46⬚ — — 0.52⬚ 2.23% Potassium sulfate 0.46 0.44 0.43 — — 0.43 2.11% 0.132⬚ 0.25⬚ 0.49⬚ — — 0.52⬚ 2.11%
- 8. 40270 -- Merck BATCH RIGHT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Potassium thiocyanate 0.61 0.59 — — — 0.59 1.52% 0.180⬚ 0.34⬚ — — — 0.52⬚ 1.52% Povidone 0.01 0.01 0.01 0.10 0.01 — 0.004⬚ 0.00⬚ 0.01⬚ 0.01⬚ 0.03⬚ — Propylene glycol 0.44 0.43 0.43 — — 0.43 2.10% 0.126⬚ 0.25⬚ 0.49⬚ — — 0.52⬚ 2.10% Pyridoxine hydrochloride 0.41 0.36 0.32 0.29 — — 0.118⬚ 0.20⬚ 0.36⬚ 0.51⬚ — — Resorcinol 0.28 0.28 0.28 0.27 — 0.27 3.3% 0.082⬚ 0.16⬚ 0.31⬚ 0.47⬚ — 0.52⬚ 3.3% Riboflavin phosphate, sodium 0.08 0.08 0.08 0.08 — — 0.022⬚ 0.04⬚ 0.09⬚ 0.15⬚ — — Rose bengal 0.08 0.07 0.07 0.07 0.07 0.06 14.9% 0.020⬚ 0.04⬚ 0.08⬚ 0.12⬚ 0.19⬚ 0.52⬚ 14.9% Rose bengal B 0.08 0.08 0.08 0.08 0.08 — 0.022⬚ 0.04⬚ 0.08⬚ 0.15⬚ 0.21⬚ — Silver nitrate 0.33 0.33 0.33 — — 0.33 2.74% 0.095⬚ 0.19⬚ 0.38⬚ — — 0.52⬚ 2.74% Silver protein, mild 0.17 0.17 0.17 0.17 0.16 0.16 5.51% 0.047⬚ 0.09⬚ 0.18⬚ 0.28⬚ 0.47⬚ 0.52⬚ 5.51% Silver protein, strong 0.12 0.08 0.06 0.05 0.04 — 0.033⬚ 0.04⬚ 0.06⬚ 0.08⬚ 0.10⬚ — Sodium acetate 0.47 0.46 0.45 — — 0.45 2.03% 0.136⬚ 0.26⬚ 0.51⬚ — — 0.52⬚ 2.03% Sodium acetate, anhydrous 0.08 0.77 — — — 0.76 1.18% 0.226⬚ 0.44⬚ — — — 0.52⬚ 1.18% Sodium antimonyl tartrate 0.14 0.13 0.13 0.12 0.12 0.11 7.9% 0.039⬚ 0.07⬚ 0.14⬚ 0.20⬚ 0.33⬚ 0.52⬚ 7.9% Sodium arsenate, dibasic 0.26 0.25 0.25 0.24 — 0.24 3.83% 0.074⬚ 0.14⬚ 0.27⬚ 0.41⬚ — 0.52 3.83% Sodium ascorbate 0.34 0.32 0.30 — — 0.30 2.99% 0.097⬚ 0.18⬚ 0.35⬚ — — 0.52⬚ 2.99% Sodium benzoate 0.40 0.40 0.40 — — 0.40 2.25% 0.116⬚ 0.23⬚ 0.46⬚ — — 0.52⬚ 2.25% Sodium bicarbonate 0.68 0.65 — — — 0.65 1.39% 0.197⬚ 0.38⬚ — — — 0.52⬚ 1.39% Sodium biphosphate monohydrate 0.45 0.43 0.41 — — 0.41 2.21% 0.128⬚ 0.24⬚ 0.47⬚ — — 0.52⬚ 2.21% Sodium biphosphate dihydrate 0.40 0.36 0.34 — — 0.32 2.77% 0.109⬚ 0.20⬚ 0.38⬚ — — 0.52⬚ 2.77% Sodium biphosphate, anhydrous 0.50 0.46 0.43 — — 0.43 2.1% 0.142⬚ 0.26⬚ 0.49⬚ — — 0.52⬚ 2.1% Sodium bismuth thioglycolate 0.20 0.19 0.18 0.18 0.17 0.17 5.29% 0.055⬚ 0.10⬚ 0.20⬚ 0.30⬚ 0.49⬚ 0.52⬚ 5.29% Sodium bisulfite 0.64 0.61 — — — 0.60 1.5% 0.186⬚ 0.35⬚ — — — 0.52⬚ 1.5% Sodium borate 0.48 0.42 0.37 — — 0.35 2.6% 0.137⬚ 0.24⬚ 0.42⬚ — — 0.52⬚ 2.6%
- 9. 40270 -- Merck BATCH LEFT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Sodium bromide 0.58 0.58 — — — 0.57 1.6% 0.166⬚ 0.32⬚ — — — 0.52⬚ 1.6% Sodium cacodylate 0.38 0.32 0.30 0.28 — 0.27 3.3% 0.104⬚ 0.18⬚ 0.33⬚ 0.48⬚ — 0.52⬚ 3.3% Sodium carbonate, anhydrous 0.74 0.70 — — — 0.68 1.32% 0.214⬚ 0.40⬚ — — — 0.52⬚ 1.32% Sodium carbonate, monohydrated 0.64 0.60 — — — 0.58 1.56% 0.183⬚ 0.34⬚ — — — 0.52⬚ 1.56% Sodium carboxymethyl cellulose 0.03 0.03 — — — — 0.007⬚ 0.01⬚ — — — — Sodium chloride 1.00 — — — — 1.00 0.9% 0.289⬚ — — — — 0.52⬚ 0.9% Sodium citrate 0.32 0.31 0.30 0.30 — 0.30 3.02% 0.091⬚ 0.17⬚ 0.34⬚ 0.51⬚ — 0.52⬚ 3.02% Sodium colistimethate 0.16 0.15 0.14 0.14 0.13 0.13 6.85% 0.045⬚ 0.08⬚ 0.16⬚ 0.23⬚ 0.38⬚ 0.52⬚ 6.85% Sodium folate 0.14 0.12 0.11 0.10 — — 0.040⬚ 0.06⬚ 0.12⬚ 0.16⬚ — — Sodium hypophosphite 0.68 0.61 — — — — 0.190⬚ 0.35⬚ — — — — Sodium iodide 0.41 0.39 0.39 — — 0.38 2.37% 0.113⬚ 0.22⬚ 0.44⬚ — — 0.52⬚ 2.37% Sodium lactate 0.58 0.55 — — — 0.52 1.72% 0.164⬚ 0.31⬚ — — — 0.52⬚ 1.72% Sodium lauryl sulfate 0.10 0.08 0.07 0.05 — — — 0.029⬚ 0.04⬚ 0.06⬚ 0.08⬚ — — — Sodium metabisulfite 0.70 0.67 — — — 0.65 1.38% 0.206⬚ 0.38⬚ — — — 0.52⬚ 1.38% Sodium nitrate 0.74 0.68 — — — 0.66 1.36% 0.214⬚ 0.39⬚ — — — 0.52 1.36% Sodium nitrite 0.86 0.84 — — — 0.83 1.08% 0.248⬚ 0.48⬚ — — — 0.52 1.08% Sodium nitroferricyanide 0.30 0.29 0.28 0.28 — 0.27 3.30% 0.086⬚ 0.16⬚ 0.32⬚ 0.47⬚ — 0.52⬚ 3.30% Sodium phosphate 0.30 0.29 0.28 0.27 — 0.27 3.33% 0.086⬚ 0.16⬚ 0.32⬚ 0.47⬚ — 0.52⬚ 3.33% Sodium phosphate, dibasic dihydrate 044 0.42 0.41 — — 0.40 2.23% 0.127⬚ 0.24⬚ 0.47⬚ — — 0.52⬚ 2.23% Sodium phosphate, dibasic dodecahydrate 0.24 0.22 0.21 0.21 — 0.20 4.45% 0.064⬚ 0.12⬚ 0.24⬚ 0.35⬚ — 0.52⬚ 4.45% Sodium phosphate, exsiccated 0.56 0.53 — — — 0.51 1.75% 0.159⬚ 0.30⬚ — — — 0.52⬚ 1.75% Sodium propionate 0.62 0.61 — — — 0.61 1.47% 0.177⬚ 0.35⬚ — — — 0.52⬚ 1.47% Sodium ricinoleate 0.10 0.10 0.10 0.09 0.09 — 0.033⬚ 0.06⬚ 0.11⬚ 0.16⬚ 0.25⬚ — Sodium salicylate 0.38 0.36 0.36 — — 0.36 2.53% 0.106⬚ 0.20⬚ 0.41⬚ — — 0.52⬚ 2.53%
- 10. 40270 -- Merck BATCH RIGHT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Sodium succinate 0.32 0.32 0.31 — — 0.31 2.90% 0.092⬚ 0.18⬚ 0.36⬚ — — 0.52⬚ 2.90% Sodium sulfate 0.28 0.26 0.25 0.23 — 0.23 3.95% 0.079⬚ 0.14⬚ 0.28⬚ 0.40⬚ — 0.52⬚ 3.95% Sodium sulfate, anhydrous 0.58 0.54 — — — 0.51 1.78% 0.165⬚ 0.30⬚ — — — 0.52⬚ 1.78% Sodium sulfite, exsiccated 0.72 0.65 — — — — 0.204⬚ 0.37⬚ — — — — Sodium tartrate 0.33 0.33 0.33 — — 0.33 2.72% 0.098⬚ 0.19⬚ 0.38⬚ — — 0.52⬚ 2.72% Sodium thiosulfate 0.32 0.31 0.31 — — 0.30 2.98% 0.092⬚ 0.18⬚ 0.35⬚ — — 0.52⬚ 2.98% Sorbitol hemihydrate 0.16 0.16 0.16 0.16 0.16 0.16 5.48% 0.045⬚ 0.09⬚ 0.19⬚ 0.28⬚ 0.48⬚ 0.52⬚ 5.48% Streptomycin calcium chloride 0.20 0.20 0.19 0.19 0.18 0.18 5.0% 0.057⬚ 0.11⬚ 0.21⬚ 0.32⬚ 0.52⬚ 0.52⬚ 5.0% Streptomycin hydrochloride 0.18 0.17 0.17 0.16 0.16 — 0.050⬚ 0.09⬚ 0.19⬚ 0.28⬚ 0.46⬚ — Streptomycin sulfate 0.08 0.07 0.07 0.06 0.06 — 0.020⬚ 0.03⬚ 0.07⬚ 0.10⬚ 0.17⬚ — Strychnine hydrochloride 0.20 0.18 0.14 — — — 0.060⬚ 0.09⬚ 0.16⬚ — — — Strychnine nitrate 0.12 0.12 — — — — 0.035⬚ 0.06⬚ — — — — Sucrose 0.08 0.08 0.09 0.09 0.09 0.10 9.25% 0.023⬚ 0.04⬚ 0.09⬚ 0.15⬚ 0.26⬚ 0.52⬚ 9.25% Sulfobromophthalein sodium 0.07 0.06 0.05 0.05 0.04 — 0.019⬚ 0.03⬚ 0.06⬚ 0.08⬚ 0.12⬚ — Tannic acid 0.03 0.03 0.03 0.03 0.03 — 0.009⬚ 0.01⬚ 0.03⬚ 0.05⬚ 0.08⬚ — Tartaric acid 0.26 0.25 0.24 0.23 — 0.23 3.9% 0.075⬚ 0.14⬚ 0.27⬚ 0.40⬚ — 0.52⬚ 3.9% Tetraethylammonium bromide 0.36 0.33 0.30 0.28 — 0.28 3.17% 0.098⬚ 0.18⬚ 0.34⬚ 0.49⬚ — 0.52⬚ 3.17% Tetraethylammonium chloride 0.36 0.34 0.33 — — 0.33 2.67% 0.100⬚ 0.19⬚ 0.38⬚ — — 0.52⬚ 2.67% Thiamine hydrochloride 0.26 0.25 0.23 0.22 — 0.21 4.24% 0.074⬚ 0.13⬚ 0.26⬚ 0.37⬚ — 0.52⬚ 4.24% Thiocyanate sodium 0.71 0.17 — — — 0.17 1.27% 0.205⬚ 0.41⬚ — — — 0.52⬚ 1.27% Thio-TEPA 0.16 0.16 0.16 0.16 0.16 0.16 5.67% 0.045⬚ 0.09⬚ 0.18⬚ 0.27⬚ 0.46⬚ 0.52⬚ 5.67% Tribromoethanol 0.06 0.05 0.05 — — — 0.015⬚ 0.03⬚ 0.05⬚ — — — Triethanolamine 0.20 0.21 0.22 0.22 — 0.22 4.05% 0.058⬚ 0.12⬚ 0.25⬚ 0.38⬚ — 0.52⬚ 4.05% Trisodium edetate, monohydrate 0.29 0.29 0.28 0.27 — 0.27 3.31% 0.079⬚ 0.15⬚ 0.31⬚ 0.47⬚ — 0.52⬚ 3.31%
- 11. 40270 -- Merck BATCH LEFT top of RH base of RH top of text base of text short standard top of RF base of RF Isotonic Solutions (Continued) Chemical Concentration of Solution, NaCl Equivalents 0.5% 1% 2% 3% 5% At Isosmotic Concentration Tromethamine 0.26 0.26 0.26 0.26 — 0.26 3.41% 0.075⬚ 0.15⬚ 0.30⬚ 0.45⬚ — 0.52⬚ 3.41% Trypan blue 0.26 0.26 — — — — 0.075⬚ 0.15⬚ — — — — Tubocurarine chloride 0.14 0.13 0.11 0.10 0.09 — 0.042⬚ 0.07⬚ 0.12⬚ 0.17⬚ 0.26⬚ — Urea 0.55 0.53 — — — 0.52 1.73% 0.158⬚ 0.30⬚ — — — 0.52⬚ 1.73% Urethan 0.31 0.31 0.31 — — 0.31 2.93% 0.089⬚ 0.17⬚ 0.35⬚ — — 0.52⬚ 2.93% Uridine 0.12 0.12 0.12 0.12 0.12 0.11 8.18% 0.035⬚ 0.06⬚ 0.13⬚ 0.20⬚ 0.33⬚ 0.52⬚ 8.18% Warfarin sodium 0.18 0.17 0.16 0.15 0.15 0.15 6.10% 0.049⬚ 0.09⬚ 0.18⬚ 0.26⬚ 0.43⬚ 0.52⬚ 6.10% Zinc chloride 0.66 0.61 — — — — 0.190⬚ 0.35⬚ — — — — Zinc p-phenosulfonate 0.18 0.18 0.18 0.17 0.17 — 0.053⬚ 0.10⬚ 0.19⬚ 0.29⬚ 0.48⬚ — Zinc sulfanilate 0.22 0.21 0.20 0.19 0.18 — 0.066⬚ 0.12⬚ 0.22⬚ 0.32⬚ 0.51⬚ — Zinc sulfate 0.16 0.15 0.14 0.13 0.12 0.12 7.65% 0.045⬚ 0.08⬚ 0.15⬚ 0.22⬚ 0.35⬚ 0.52⬚ 7.65% Zinc sulfate (dried) 0.24 0.23 0.22 0.21 — 0.20 4.52% 0.072⬚ 0.136⬚ 0.252⬚ 0.362⬚ — 0.52⬚ 4.52% References 1. E. R. Hammarlund, K. Pedersen-Bjergaard, J. Am. Pharm. Assoc., Sci. Ed. 47, 107 (1958). 2. E. R. Hammarlund et al., J. Pharm. Sci. 54, 160 (1965). 3. E. R. Hammarlund, G. L. Van Pevenage, ibid. 55, 1448 (1996). 4. W. E. Fassett et al., ibid. 58, 1540 (1969). 5. C. Sapp et al., ibid. 64, 1884 (1975). 6. E. R. Hammarlund, ibid. 70, 1161 (1981). 7. A. R. Gennaro (Ed)., Remington’s Pharmaceutical Sciences (Mack Publ. Co., Easton, PA., 17th ed., 1985) pp 1455- 1472. 8. E. R. Hammarlund, J. Pharm. Sci. 78, 519 (1989).