Report
Share
Recommended
Recommended
More Related Content
Similar to Biogen
Similar to Biogen (20)
PD1 & PD L1 Targeted Agents - Nivolumab Clinical & Commercial Development (11...

PD1 & PD L1 Targeted Agents - Nivolumab Clinical & Commercial Development (11...
Presentation the 38th jp morgan healthcare conference chinese

Presentation the 38th jp morgan healthcare conference chinese
Presentation at The 38th JP Morgan Healthcare Conference_English

Presentation at The 38th JP Morgan Healthcare Conference_English
Recently uploaded
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE AbudhabiAbortion pills in Kuwait Cytotec pills in Kuwait
VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...

VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Recently uploaded (20)
Call Girls In Noida 959961⊹3876 Independent Escort Service Noida

Call Girls In Noida 959961⊹3876 Independent Escort Service Noida
Call Girls In DLf Gurgaon ➥99902@11544 ( Best price)100% Genuine Escort In 24...

Call Girls In DLf Gurgaon ➥99902@11544 ( Best price)100% Genuine Escort In 24...
Call Girls in Delhi, Escort Service Available 24x7 in Delhi 959961-/-3876

Call Girls in Delhi, Escort Service Available 24x7 in Delhi 959961-/-3876
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi
How to Get Started in Social Media for Art League City

How to Get Started in Social Media for Art League City
Falcon's Invoice Discounting: Your Path to Prosperity

Falcon's Invoice Discounting: Your Path to Prosperity
Call Girls In Panjim North Goa 9971646499 Genuine Service

Call Girls In Panjim North Goa 9971646499 Genuine Service
FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756

FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756
Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...

Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...
Call Girls Hebbal Just Call 👗 7737669865 👗 Top Class Call Girl Service Bangalore

Call Girls Hebbal Just Call 👗 7737669865 👗 Top Class Call Girl Service Bangalore
BAGALUR CALL GIRL IN 98274*61493 ❤CALL GIRLS IN ESCORT SERVICE❤CALL GIRL

BAGALUR CALL GIRL IN 98274*61493 ❤CALL GIRLS IN ESCORT SERVICE❤CALL GIRL
Call Girls Kengeri Satellite Town Just Call 👗 7737669865 👗 Top Class Call Gir...

Call Girls Kengeri Satellite Town Just Call 👗 7737669865 👗 Top Class Call Gir...
Insurers' journeys to build a mastery in the IoT usage

Insurers' journeys to build a mastery in the IoT usage
B.COM Unit – 4 ( CORPORATE SOCIAL RESPONSIBILITY ( CSR ).pptx

B.COM Unit – 4 ( CORPORATE SOCIAL RESPONSIBILITY ( CSR ).pptx
VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...

VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...
The Path to Product Excellence: Avoiding Common Pitfalls and Enhancing Commun...

The Path to Product Excellence: Avoiding Common Pitfalls and Enhancing Commun...
Biogen
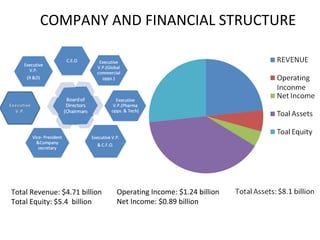
- 1. COMPANY AND FINANCIAL STRUCTURE Total Revenue: $4.71 billion Total Equity: $5.4 billion Operating Income: $1.24 billion Net Income: $0.89 billion
- 2. CEO: George A. Scangos CFO & Management: Paul Clancy VP of R&D: Douglas Williams Regulatory: Nadine Cohen The Brains and Goals Behind Biogen Idec Business Goals as of 2007: -- 100,000 patients on TYSABRI(R) (natalizumab); Not yet met. -- More than 40% of the Company's revenue coming from its International business; Not yet met. -- Four new products and/or existing products launched in new indications; Not yet met. -- Six programs in late-stage clinical development. Goal achieved.
- 4. PHASES DRUGS FDA APPROVED DRUGS PHASE 1 ANTI-LINGO, NEBULASTIN, CD40L, ANTI-TWEAK, BIIB037 (HUMAN ANTI-AMYLOID B MAB) PHASE 2 OCRELIZUMAB (HUMANIZED ANTI-CD20 MAB). PHASE 3 PEG IFN (PEGYLATED INTERFERON BETA-1A), BG-12 (DIMETHYLFUMARATE), DACLIZUMAB, LONG LASTING r FACTOR IX, LONG LASING r FACTOR VIII, GA101 (HUMANIZED ANTI-CD20 MAB), DEXPRAMIPEXOLE. PHASE 4 ------- FILLING -------
- 5. WORK DONE IN QUALITY ASSURANCE DEVELOPMENT, MAINTENANCE AND MANAGEMENT OF QUALITY SYSTEMS ENSURE THAT EXCEPTIONS ARE ANALYZED FOR PREVENTIVE ACTIONS AND PRODUCT IMPACT. DEVELOP AND MAINTAIN DATA ANALYSIS AND TRENDING SYSTEMS FOR PRODUCT SPECIFIC DATA GENERATED AT THE FACILITY. PROVIDE TECHNICAL EXPERTISE FOR PRODUCT QUALITY DECISIONS. MAINTAIN PRODUCT DISPOSITION PROCEDURES. EVALUATION OF FACILITY, PROCEDURES, EQUIPMENT, PERSONNEL AND PROCESSES. PROCESSES USED MASS SPECTROSCOPY: ESI TOF MS, MALDI MS,LC UV MS. CHROMATOGRAPHY: HIC, RPLC, SEC, ION EXCHANGE CHROMATOGRAPHY, SDS PAGE cIEF