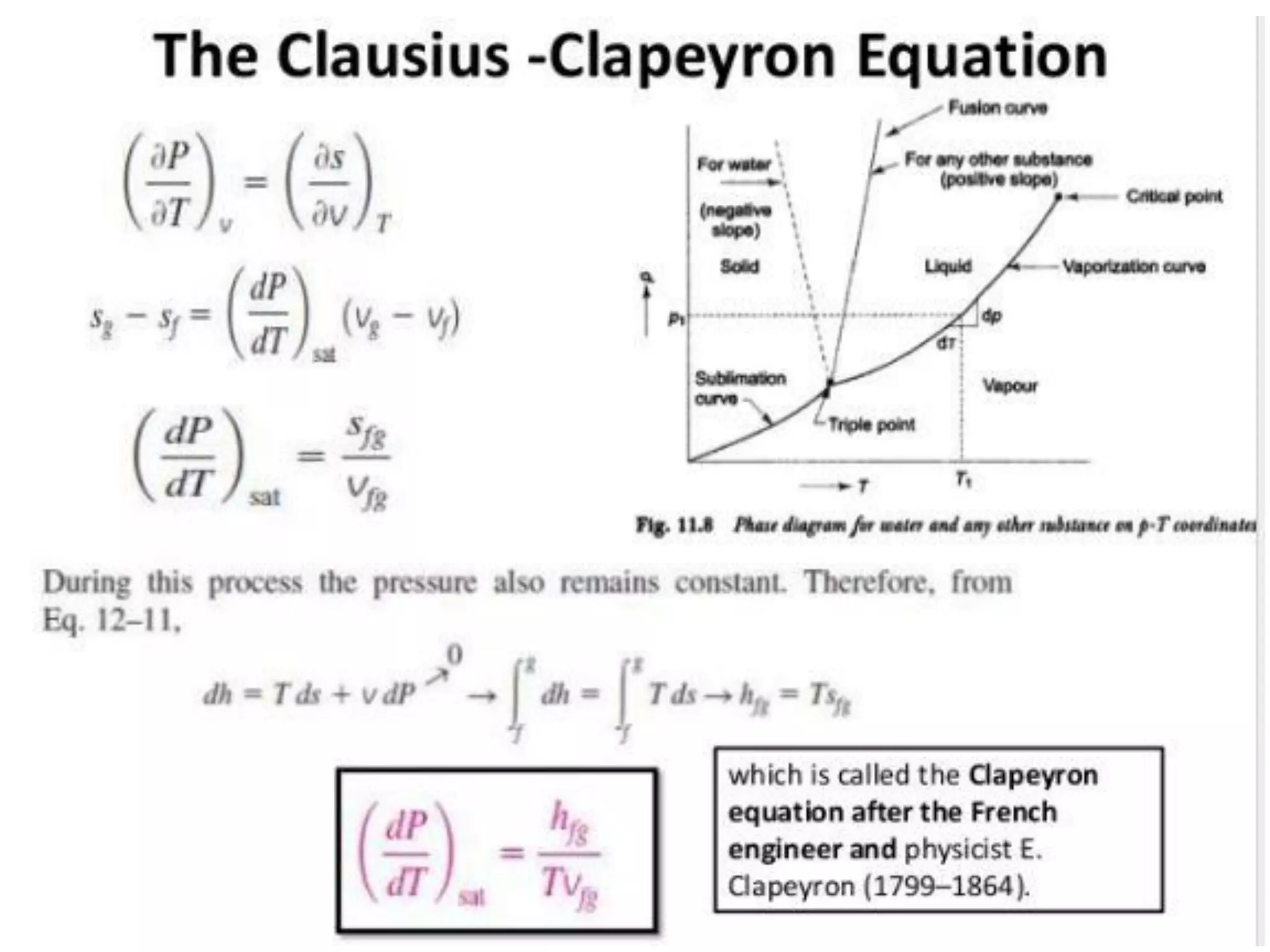
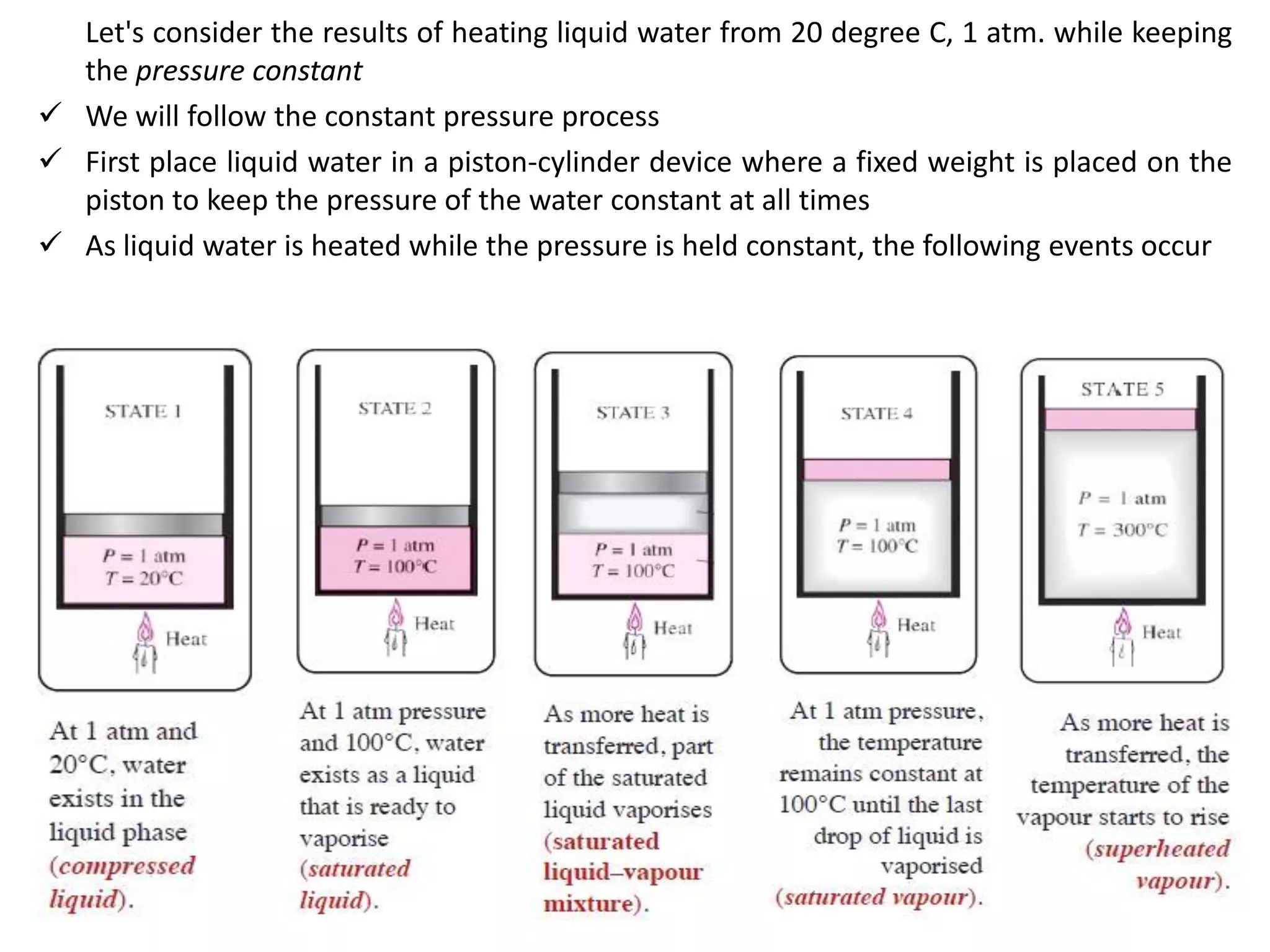
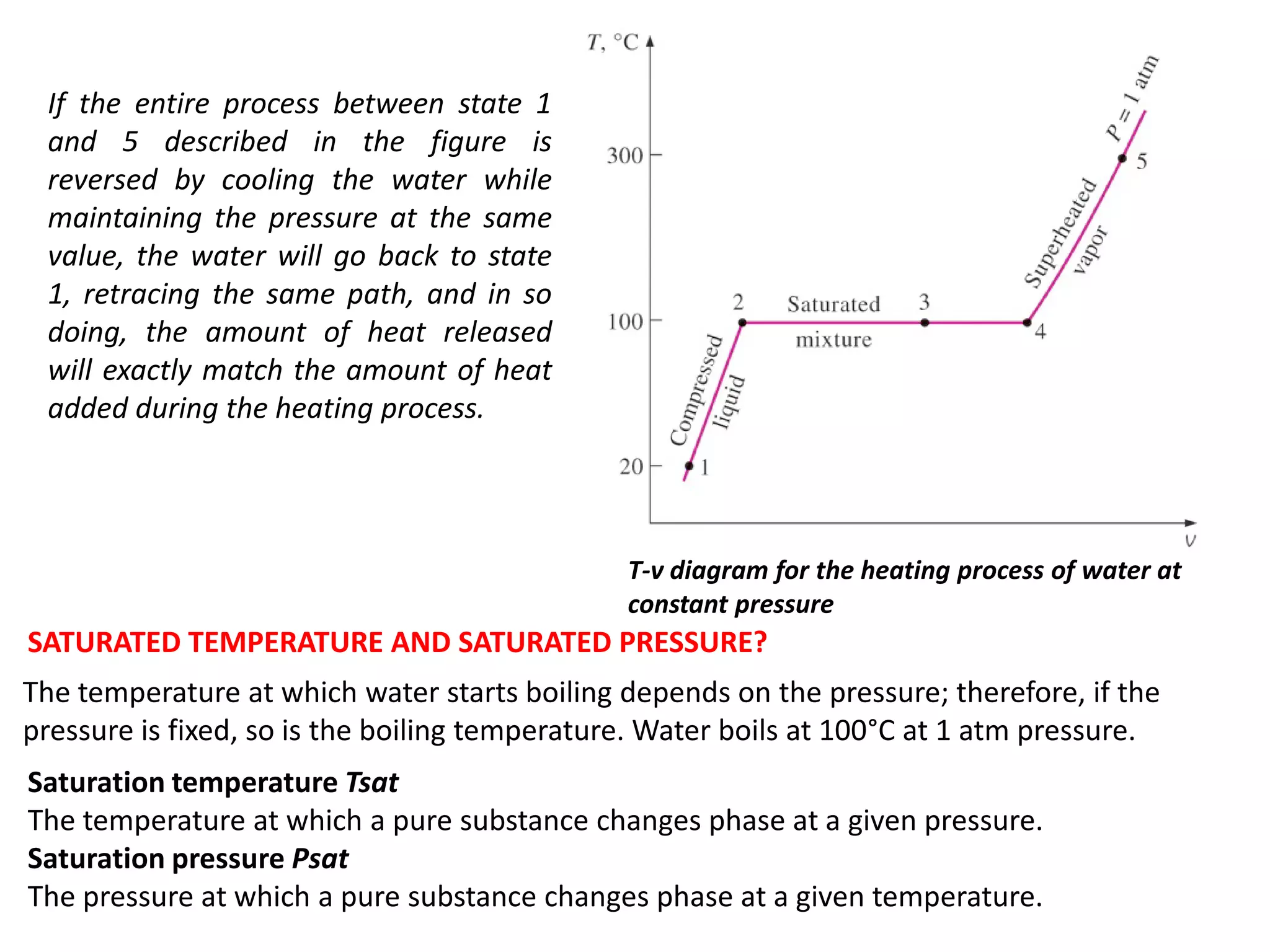
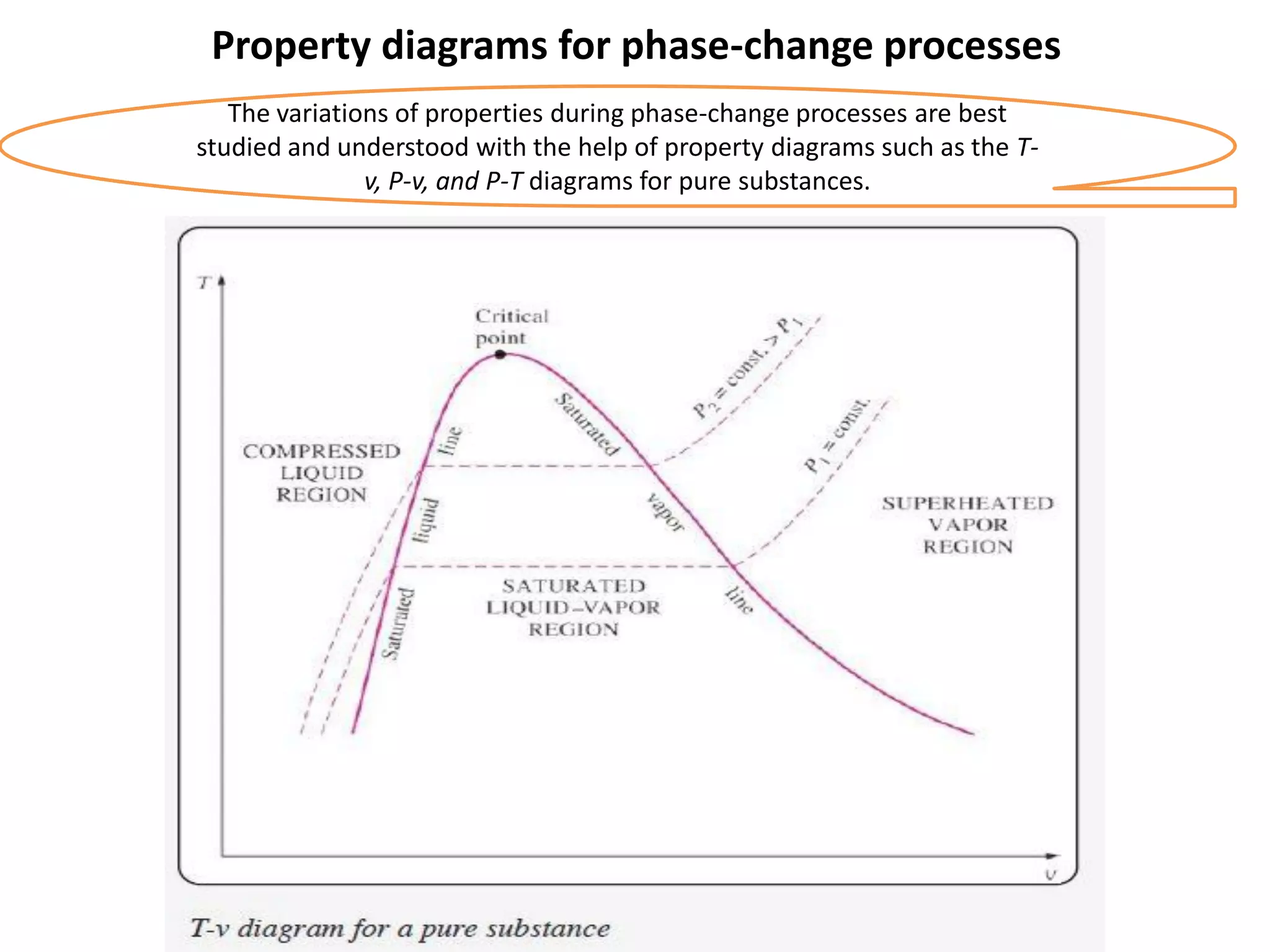
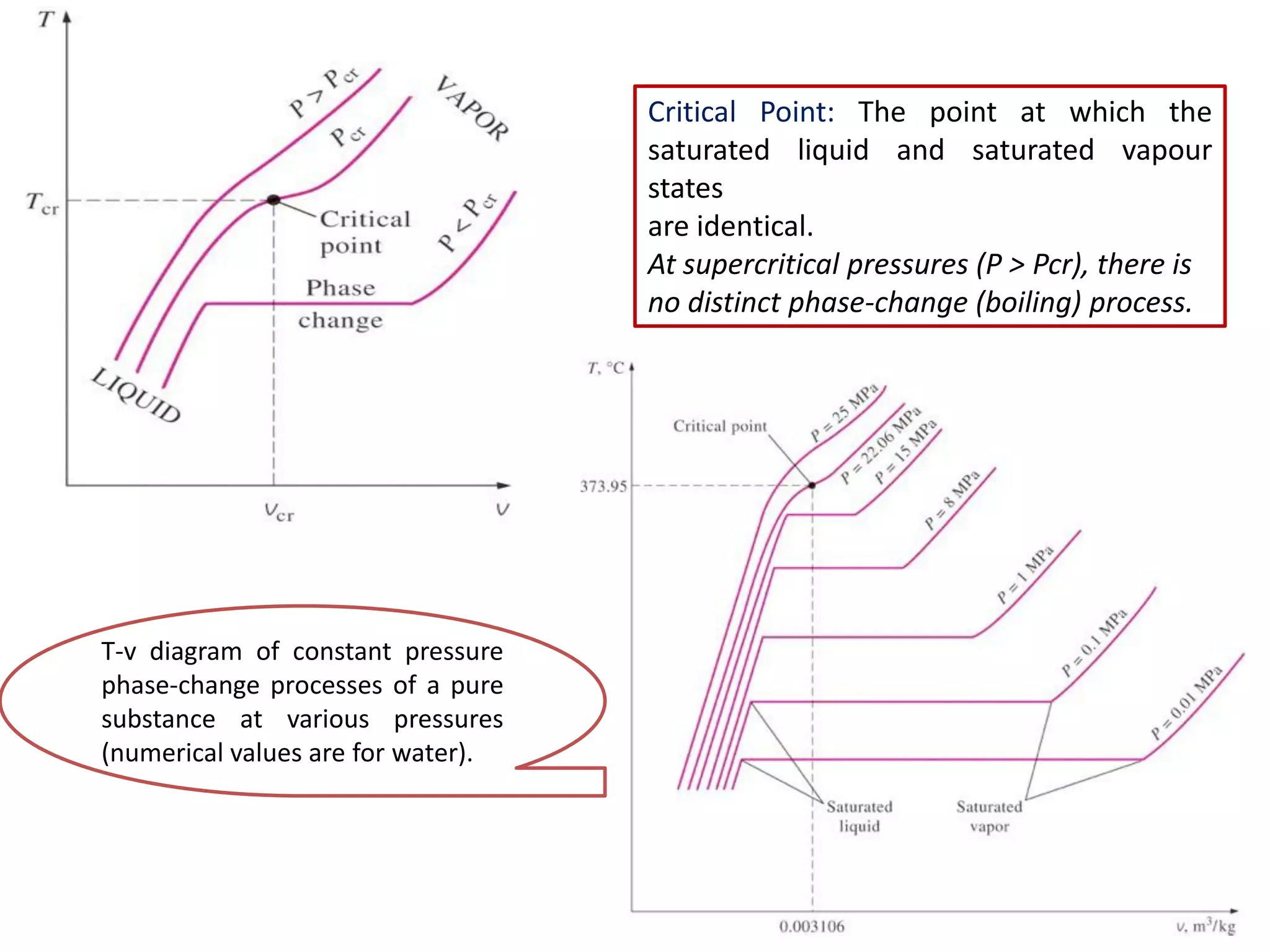
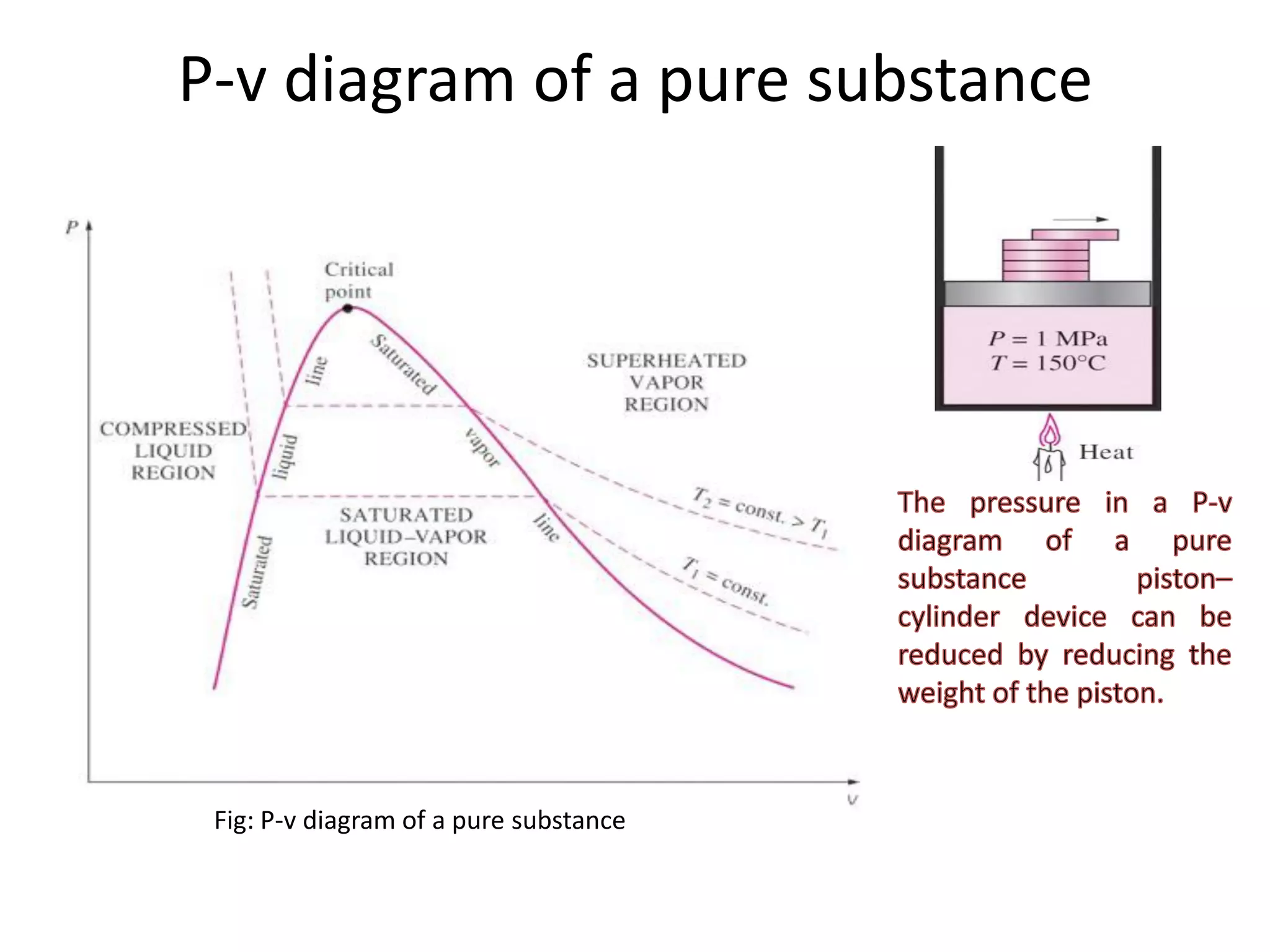
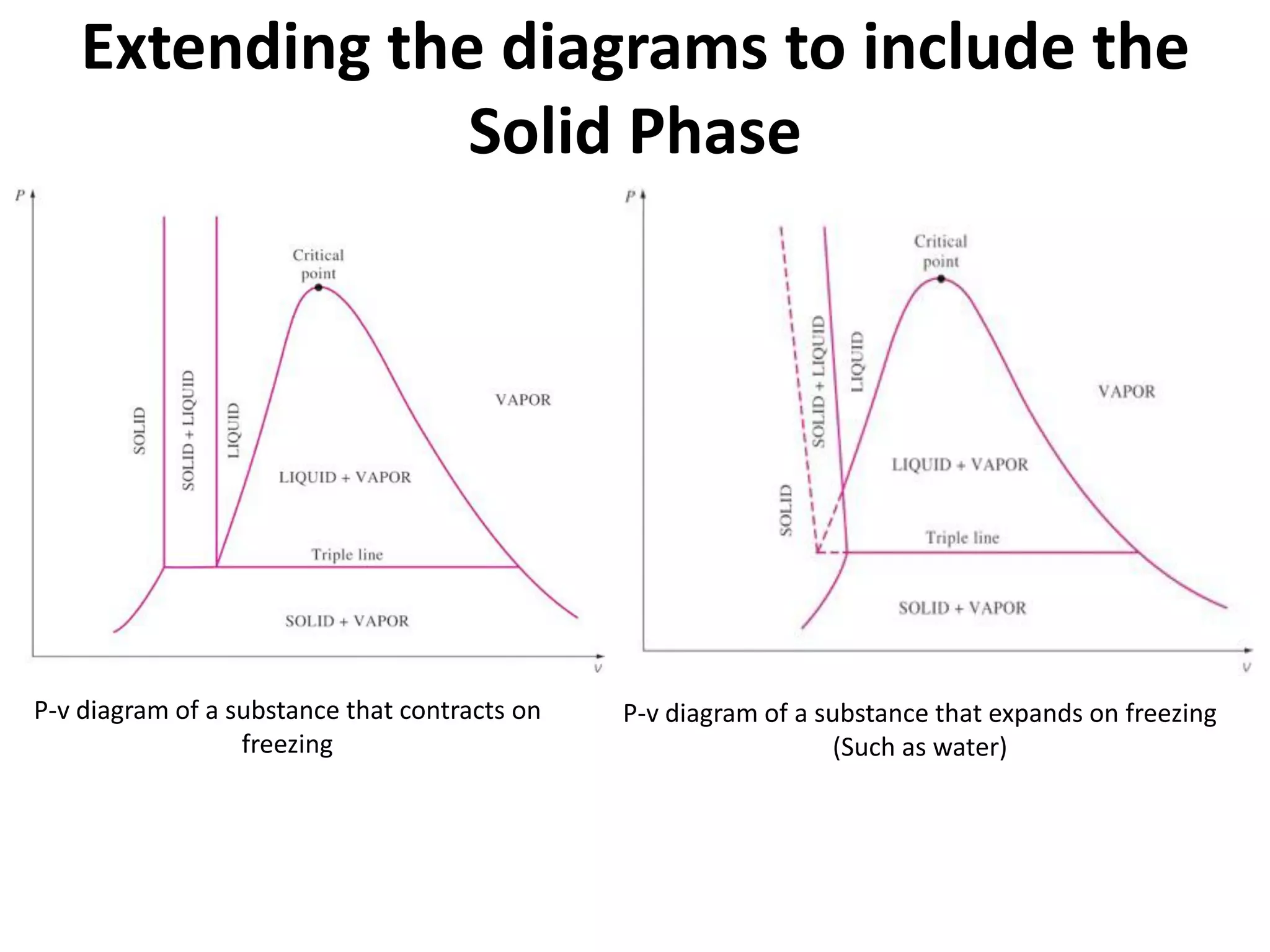
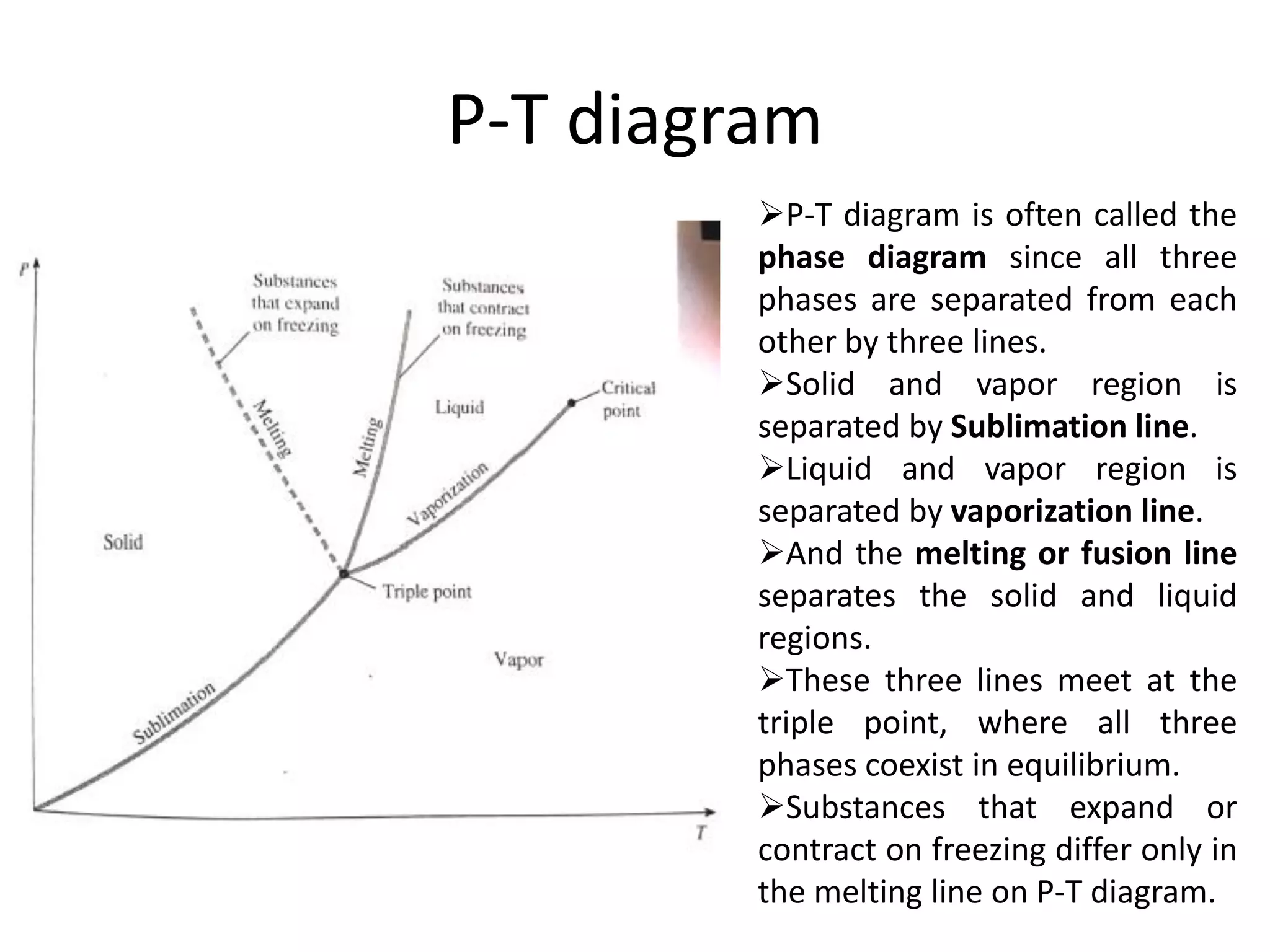
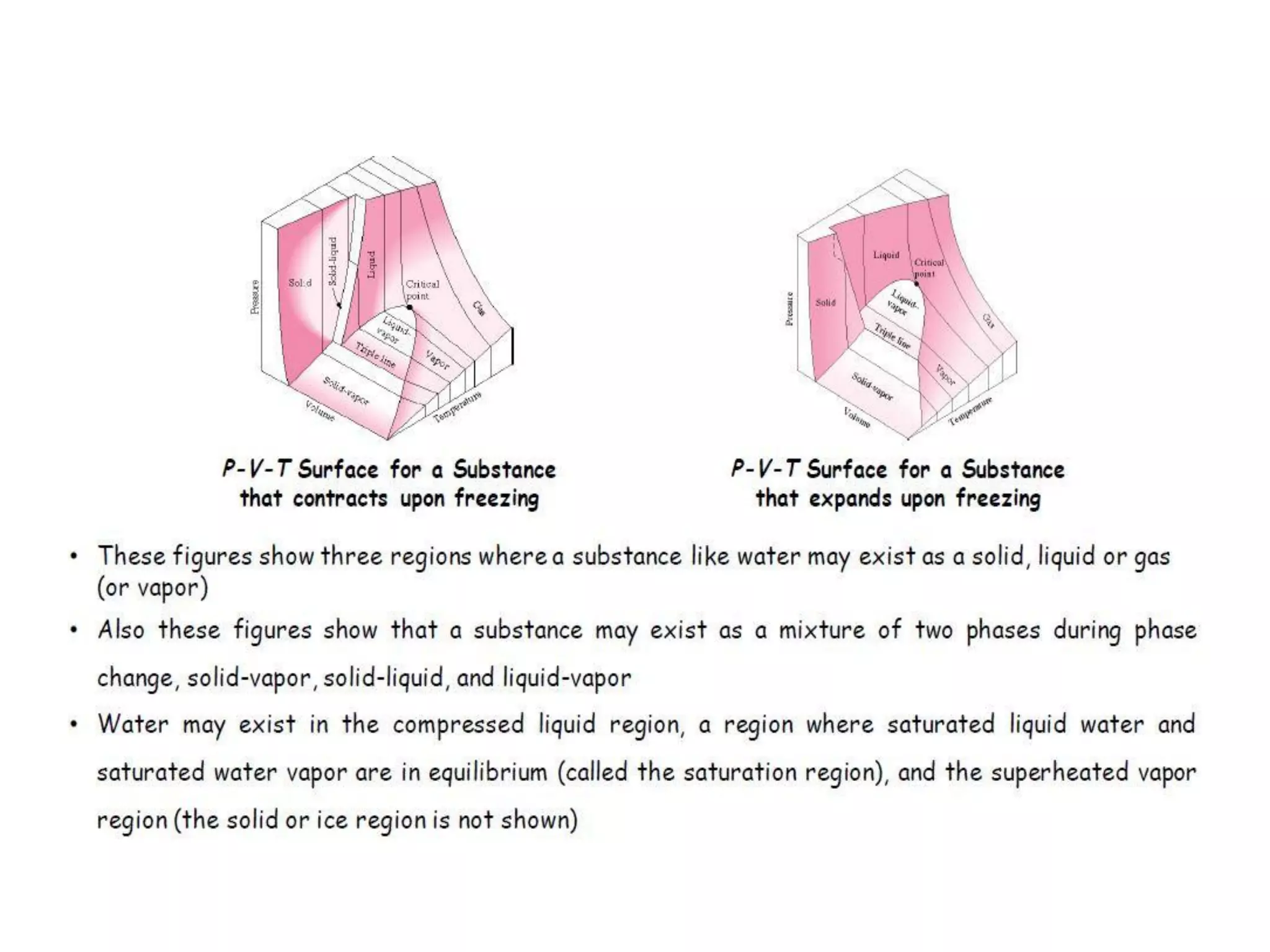
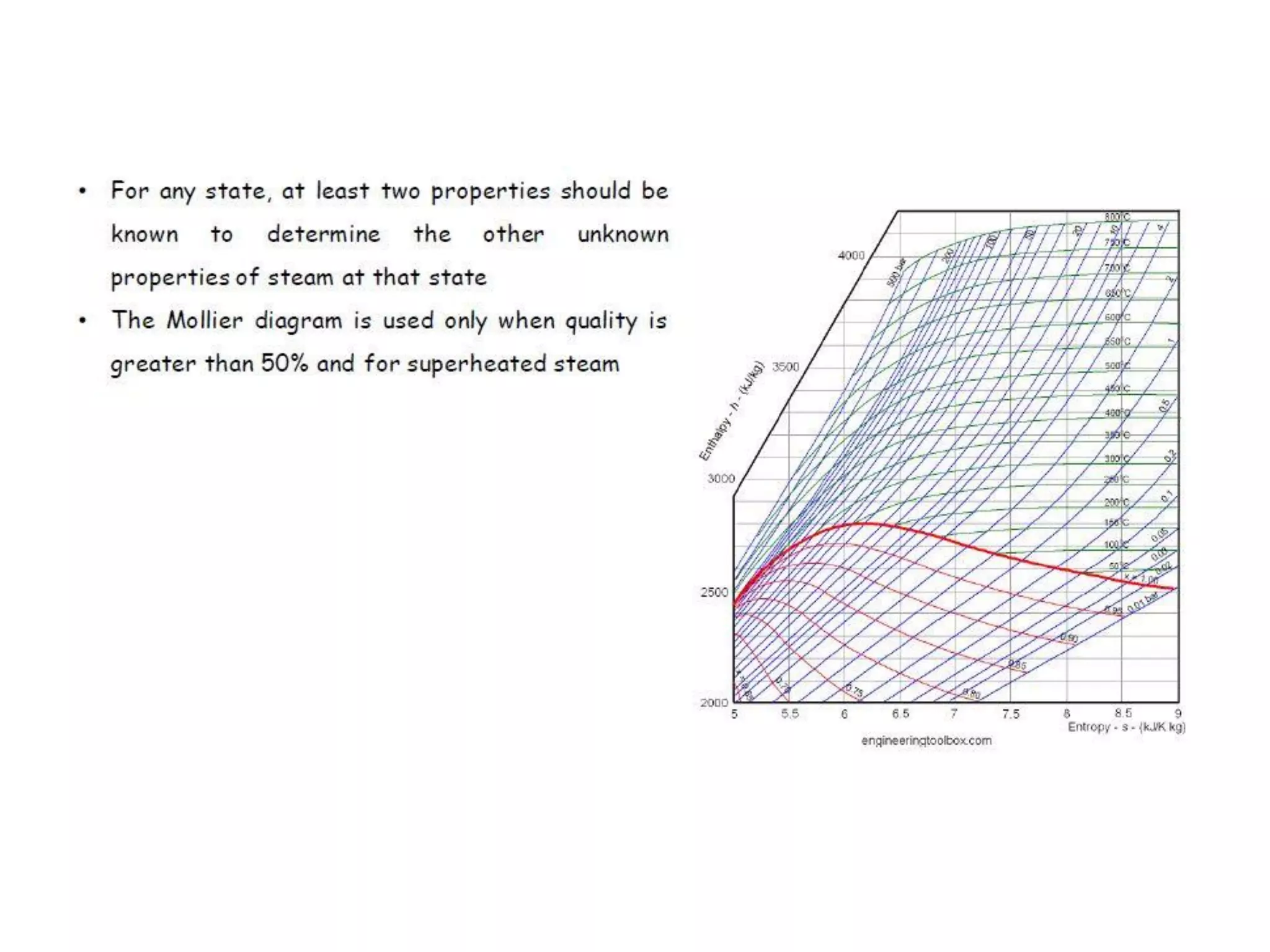
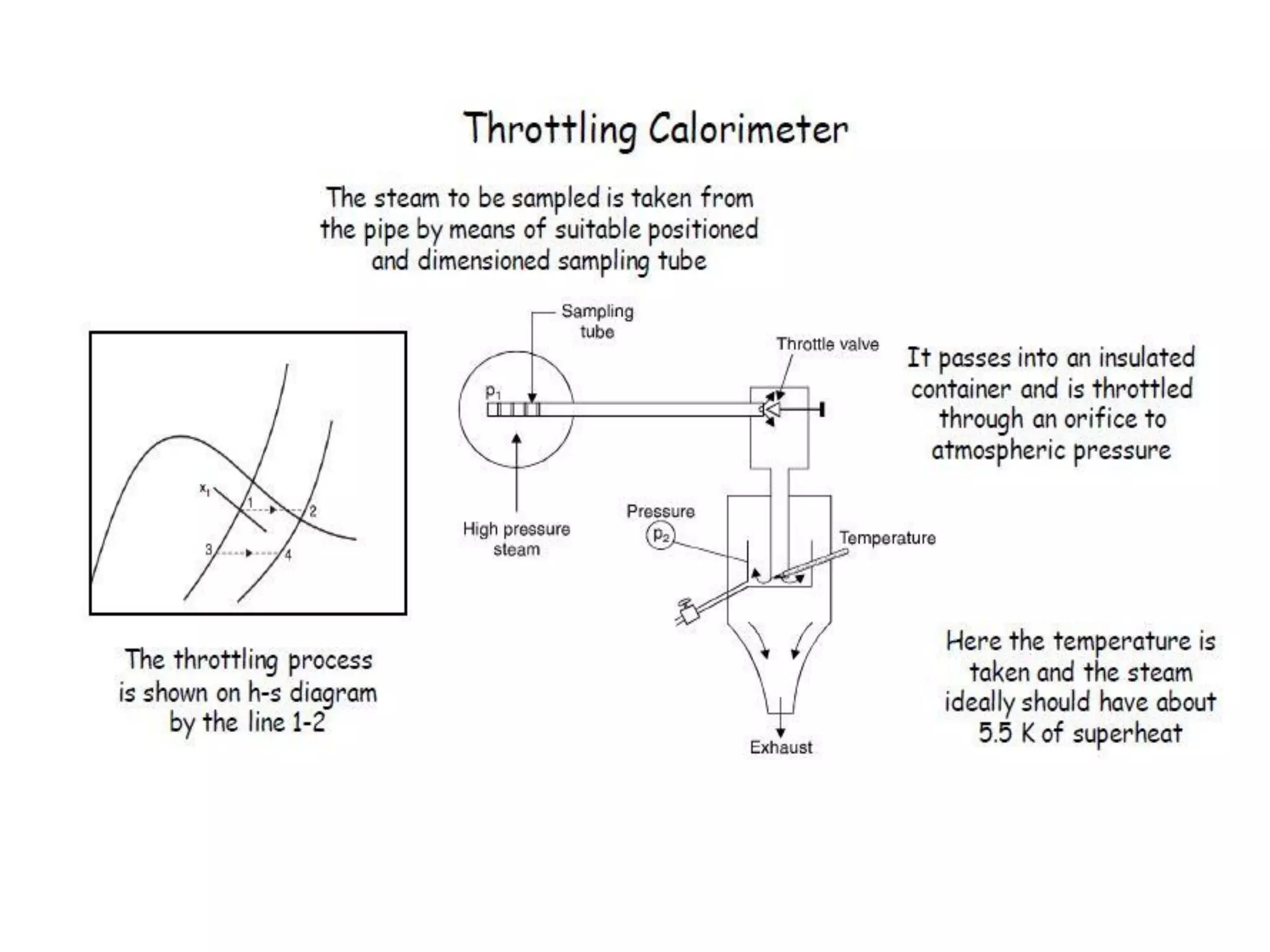
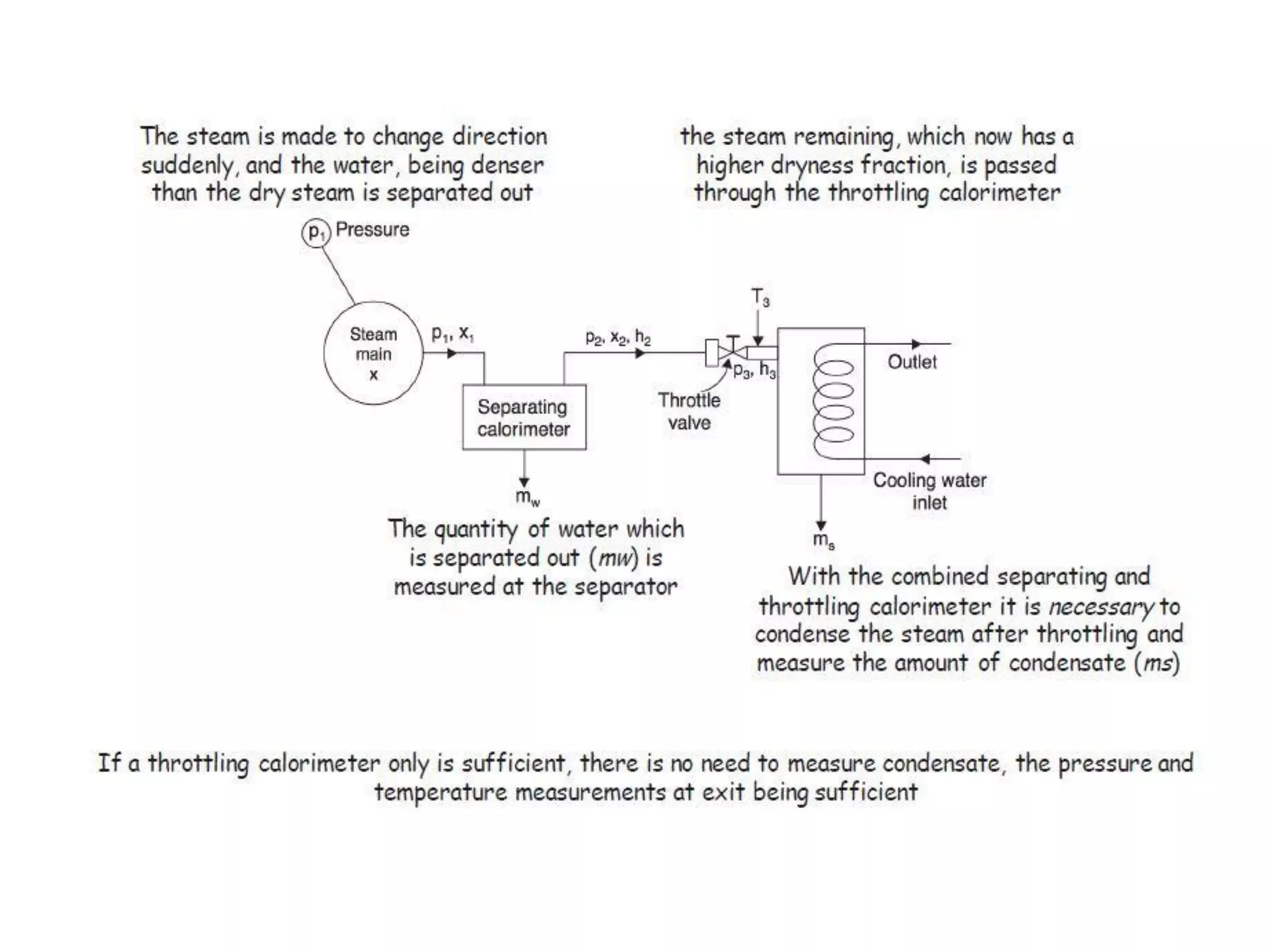
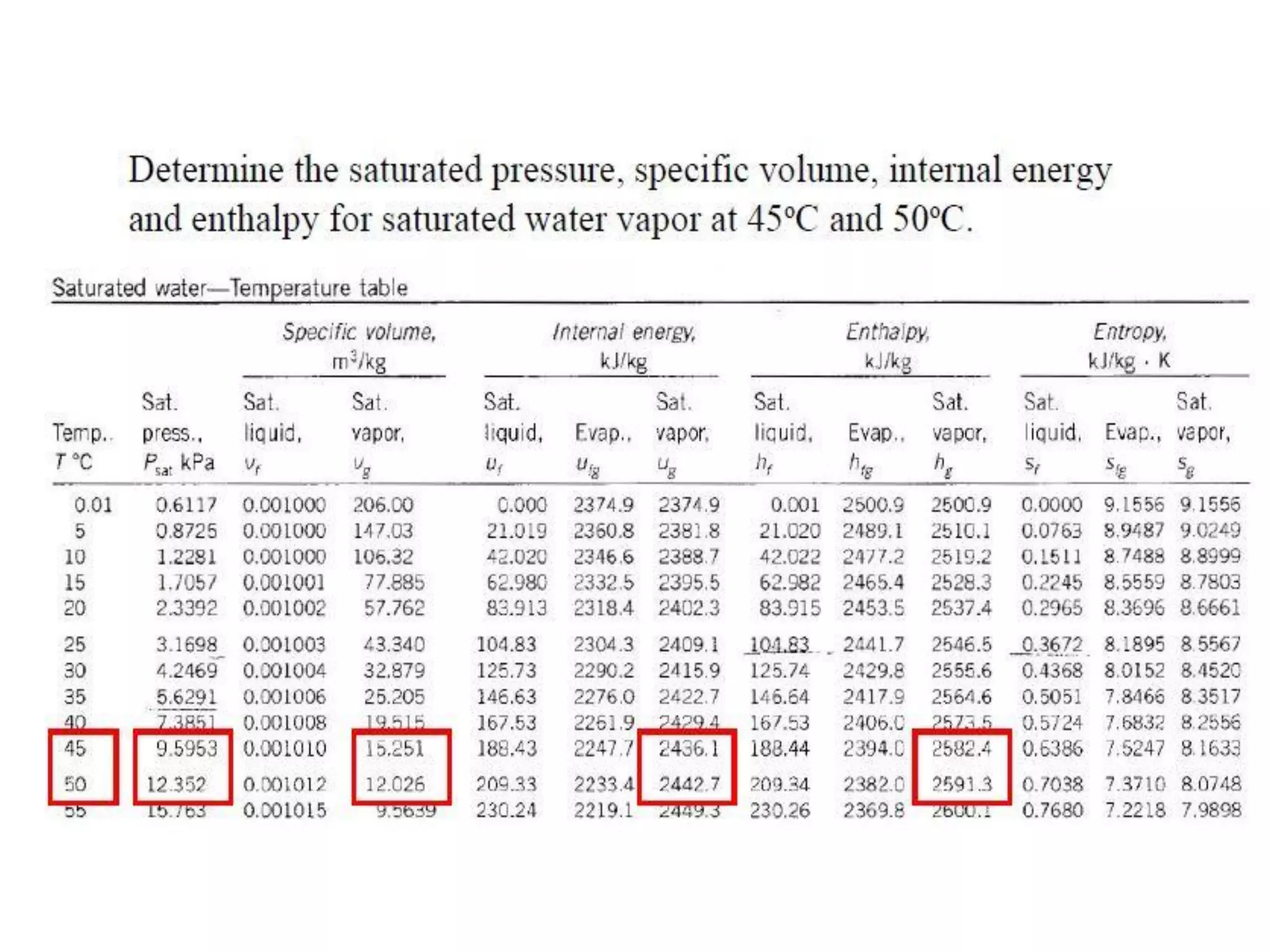
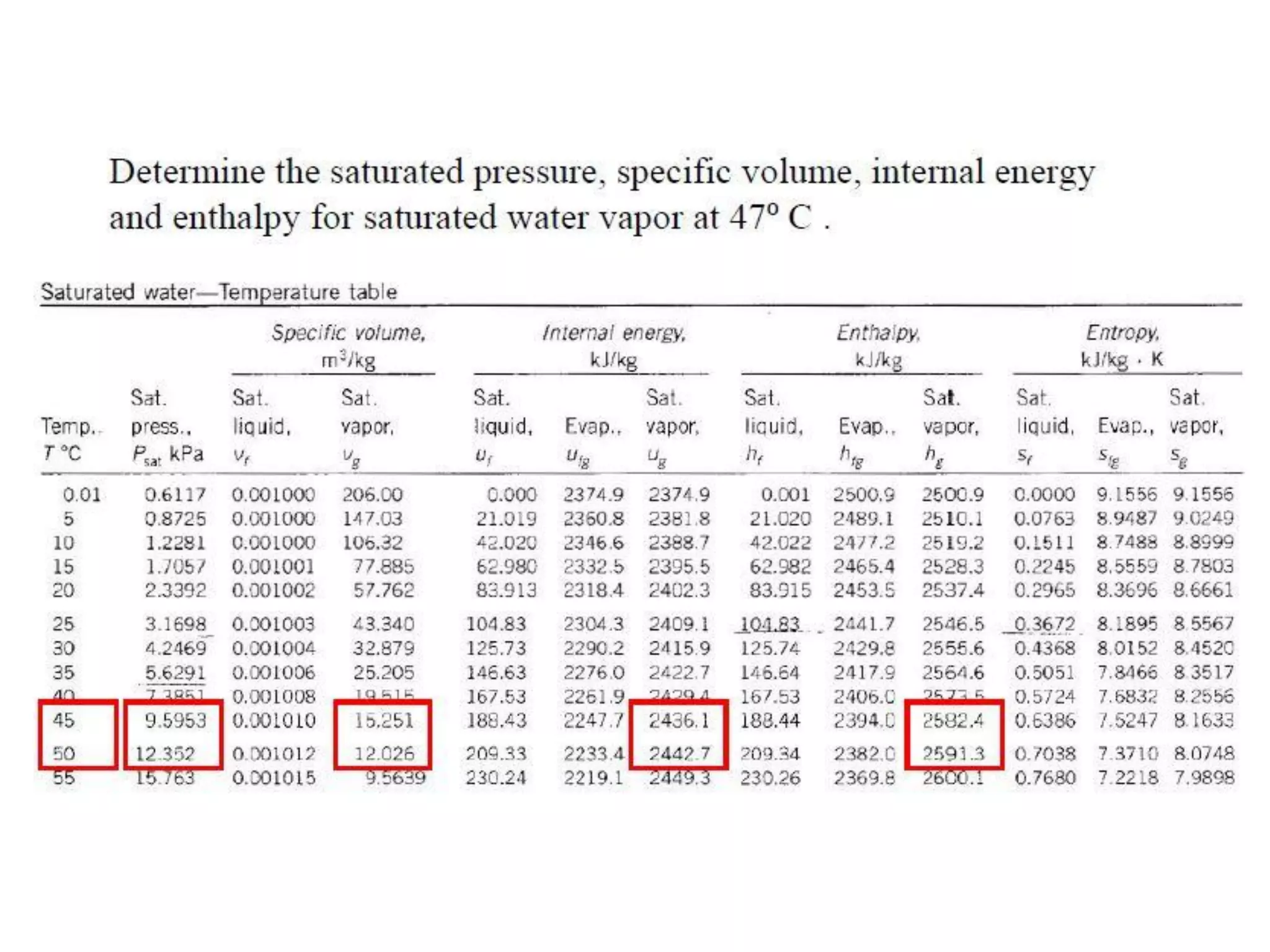
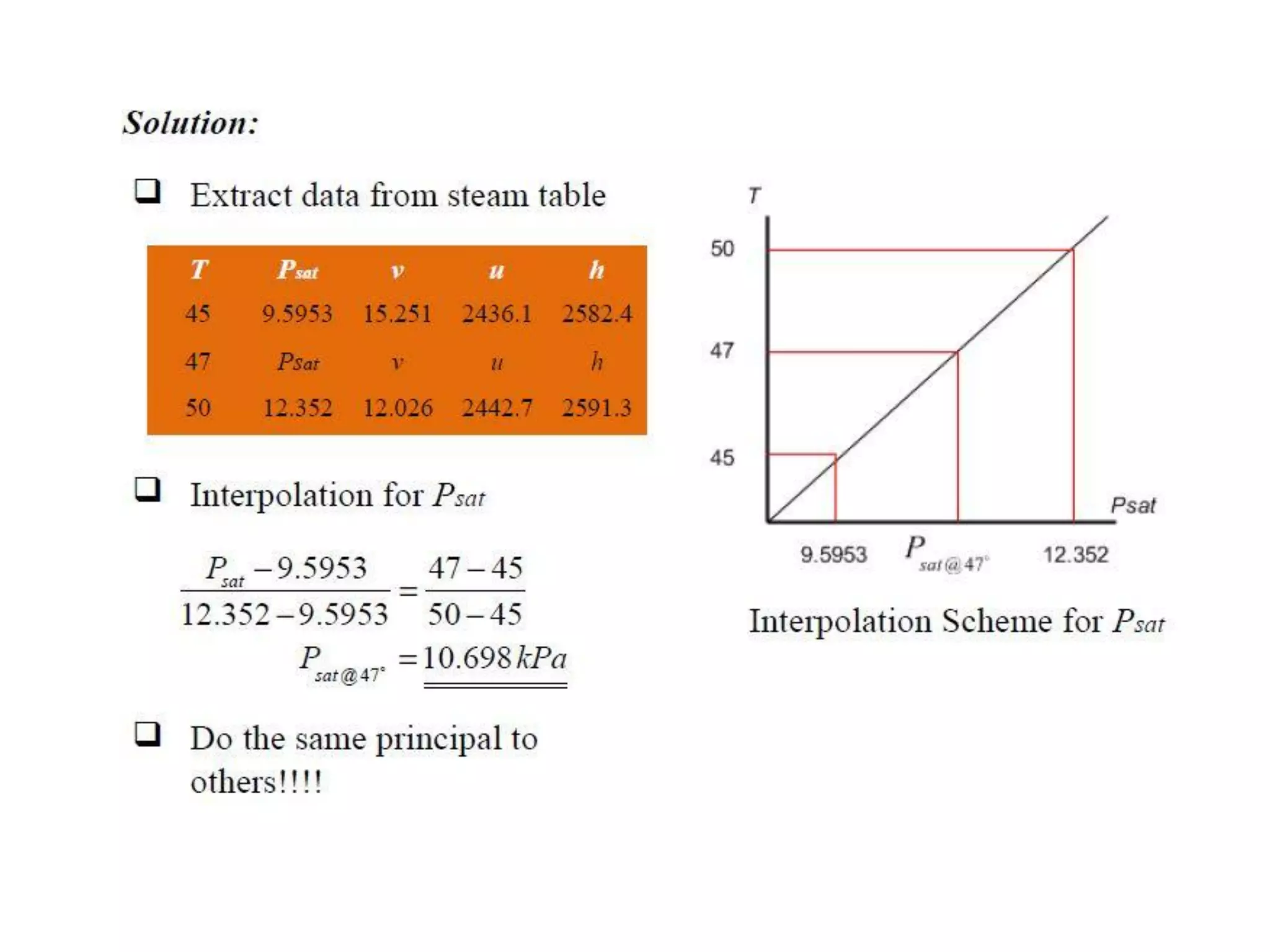
The document discusses thermodynamic properties, focusing on the Gibbs and Helmholtz functions, as well as the phases of pure substances. It explains key concepts such as saturated and superheated states, phase change processes, and the significance of latent heat in transitions between states. Additionally, it covers property diagrams for analyzing phase-change processes of pure substances, including T-v, P-v, and P-T diagrams.