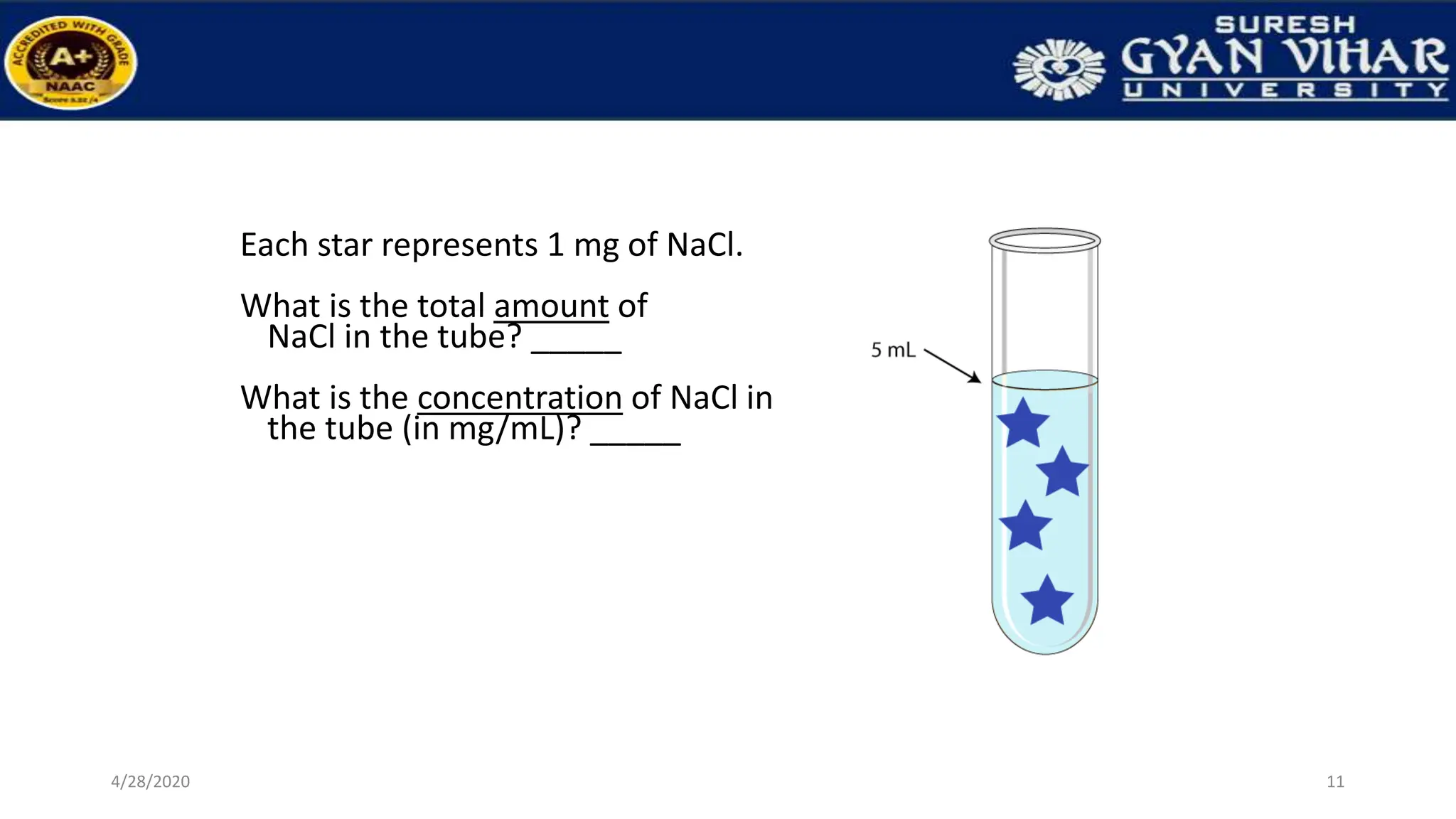
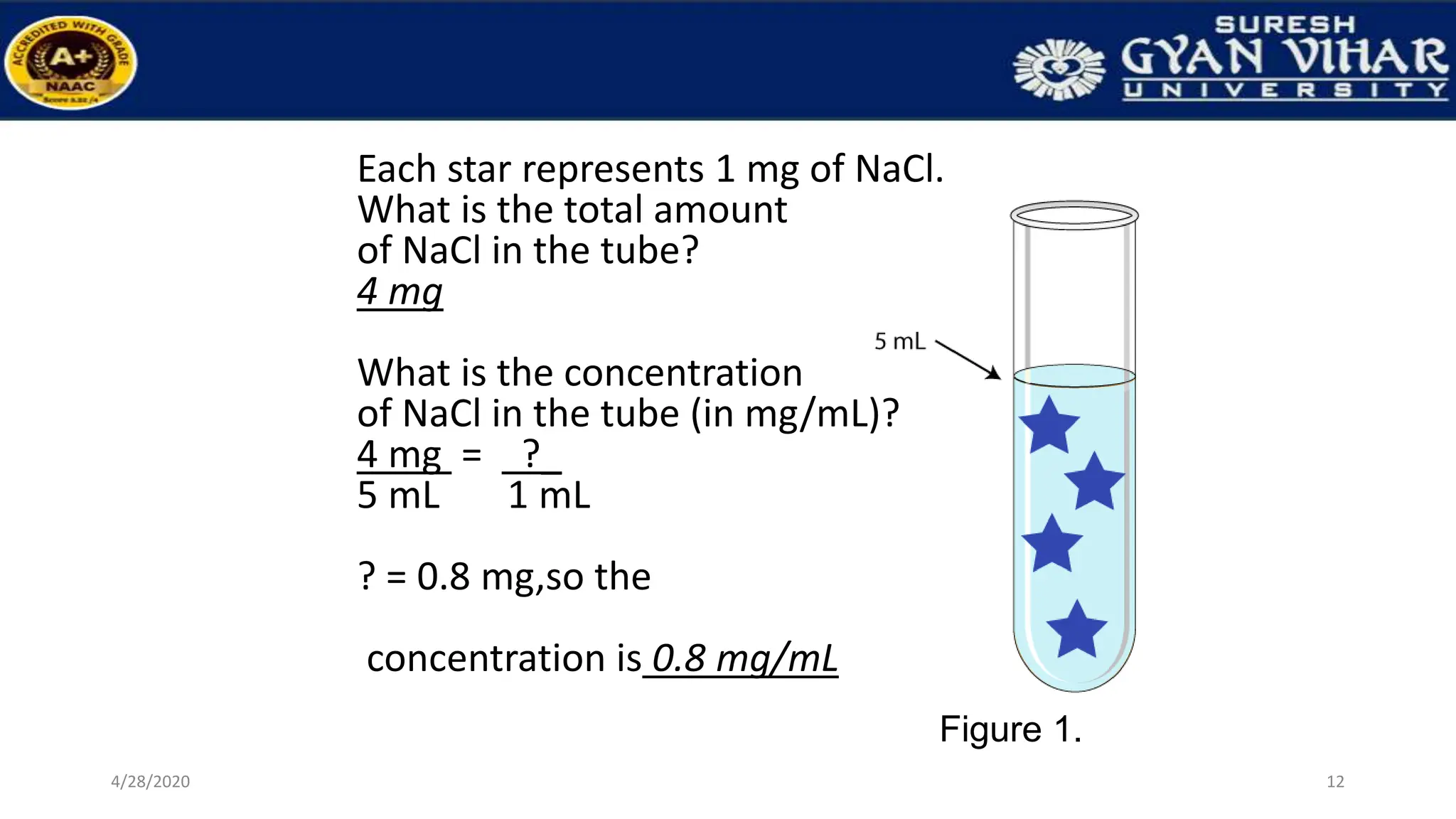
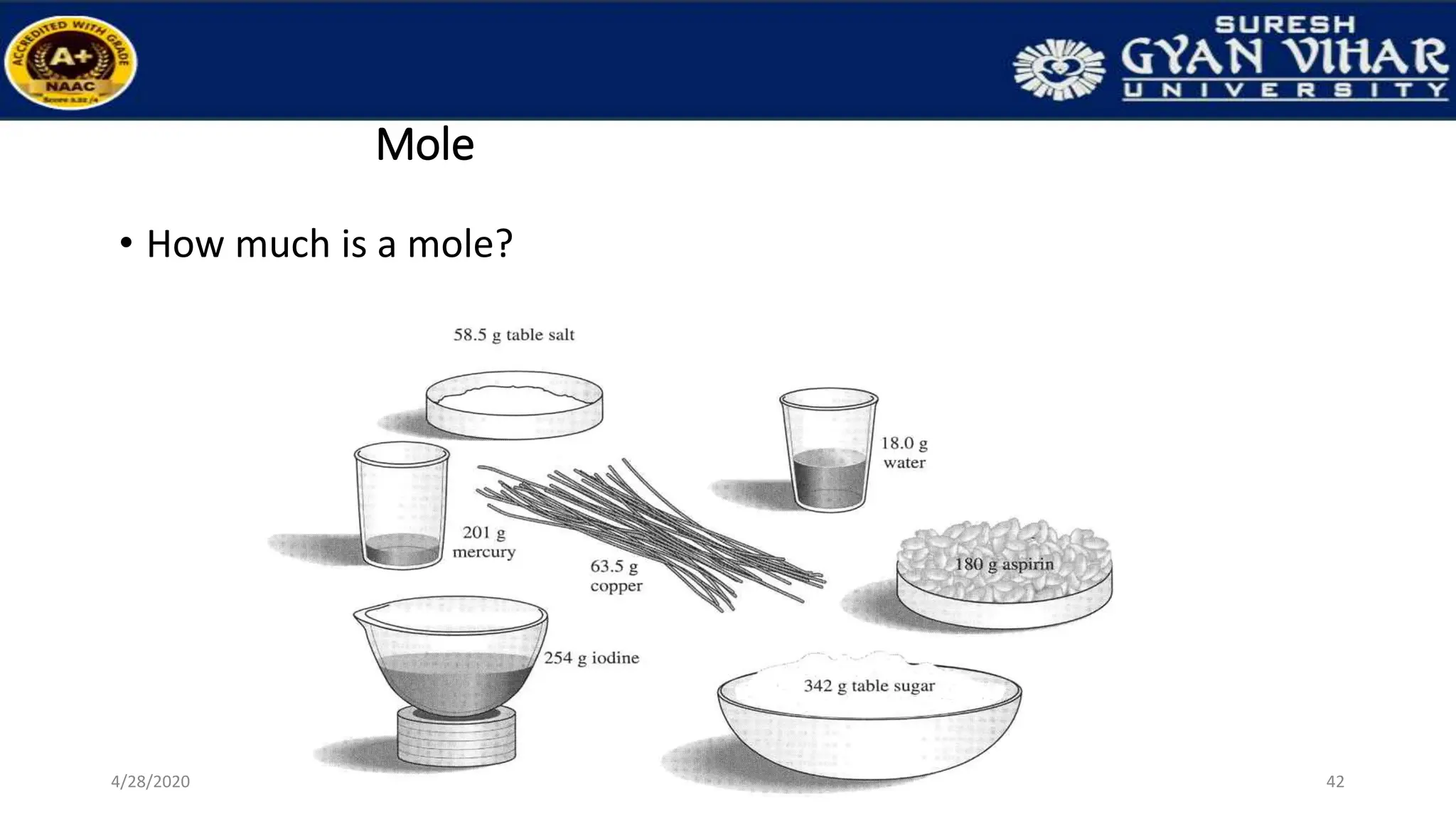
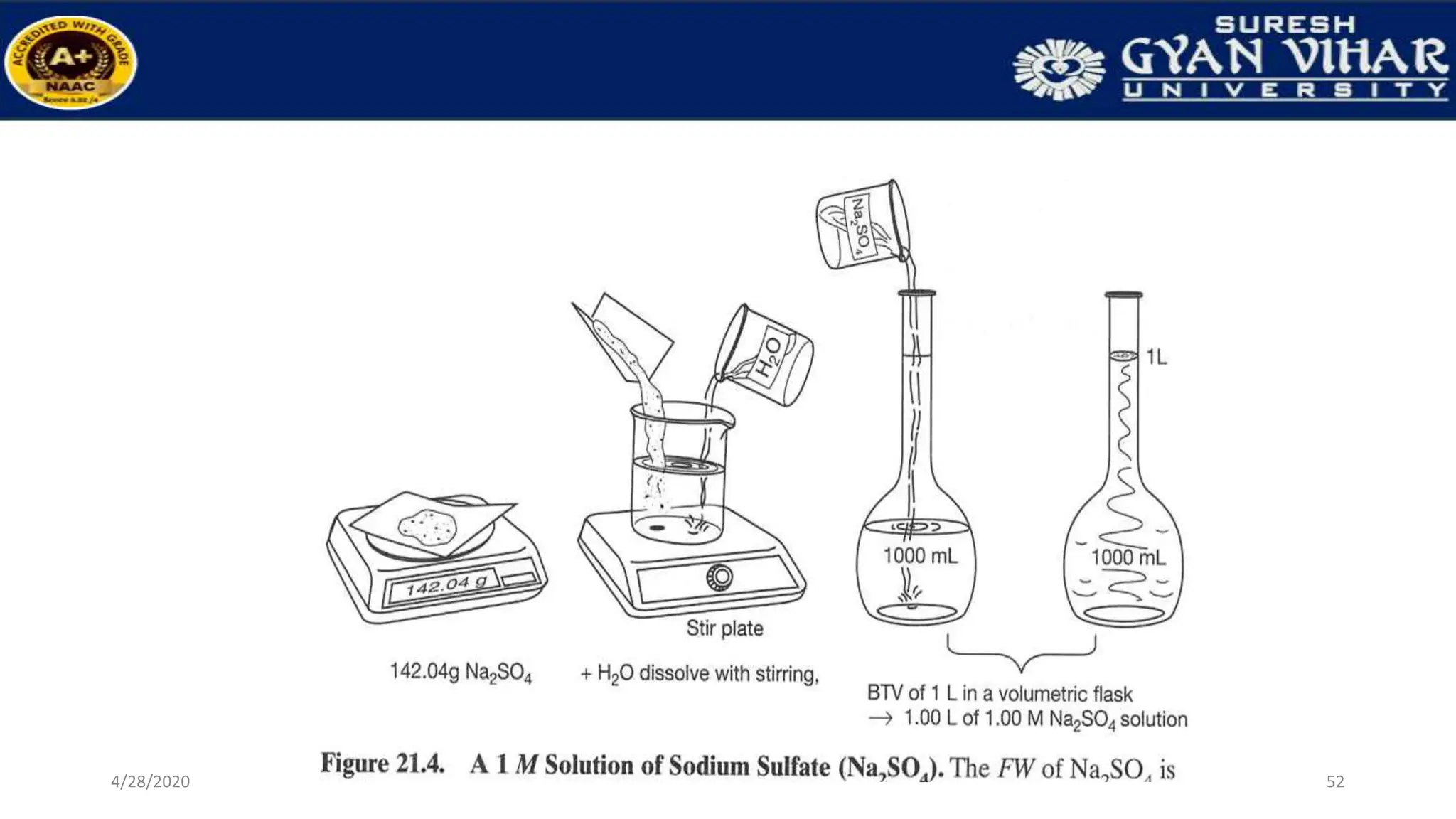
This document discusses the preparation of standard solutions, including definitions of key terms like solute, solvent, saturation, and stock solutions. It outlines methods for expressing concentrations, calculations for preparing specific concentrations, and the importance of using standardized solutions in laboratory tests. Additionally, it addresses the preparation of solutions from concentrated stock and the effects of temperature and dilution on buffer solutions.