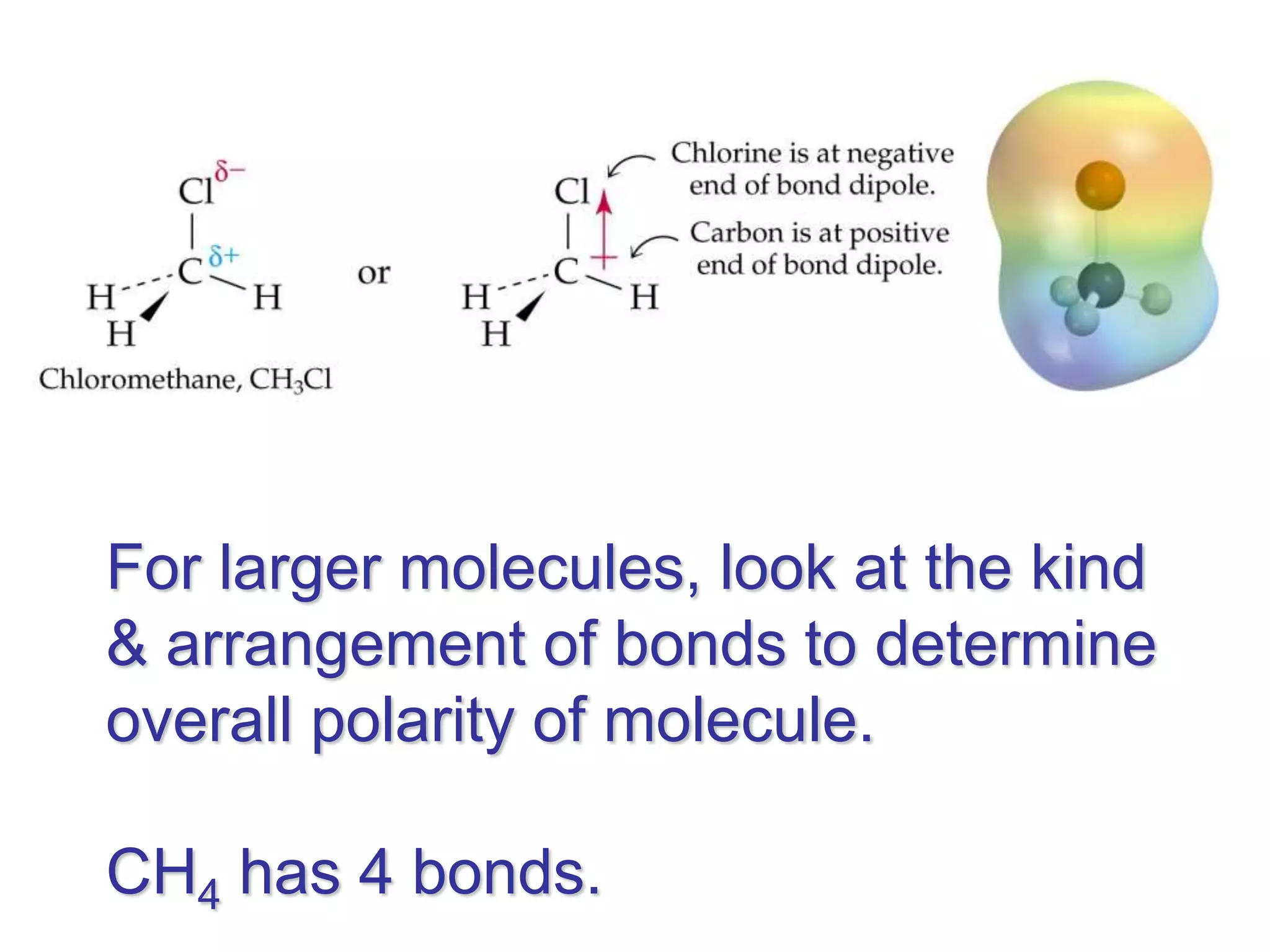
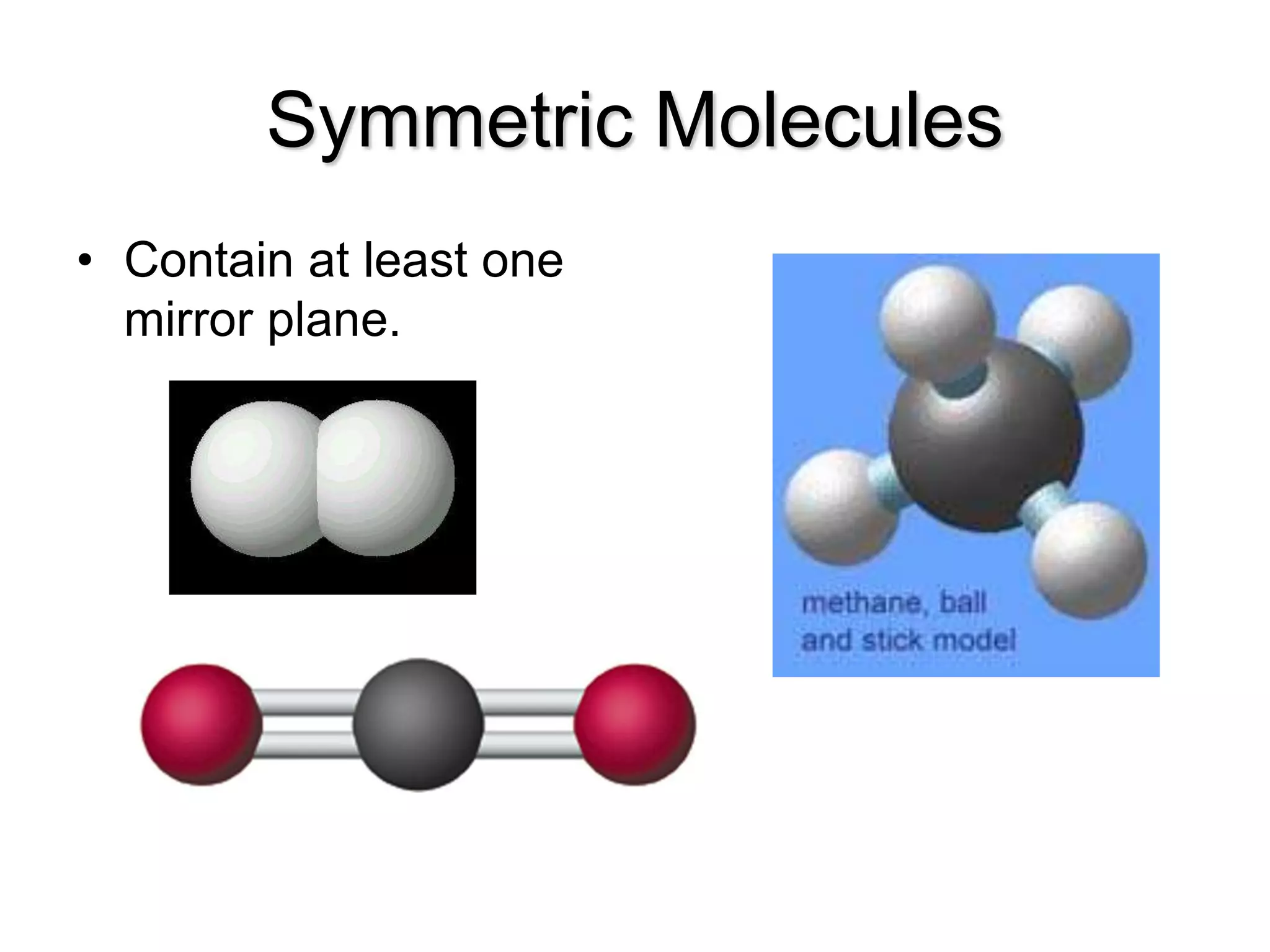
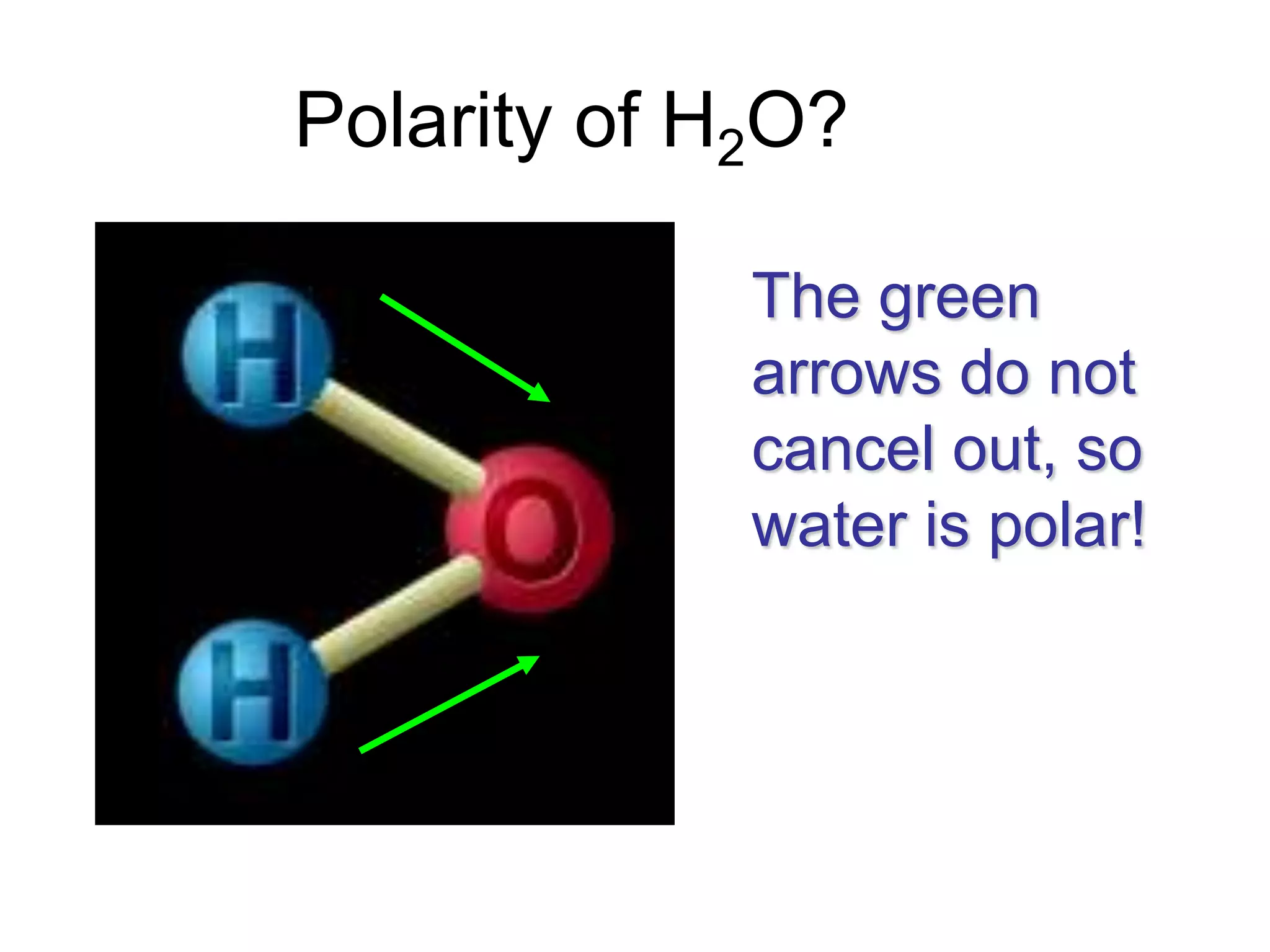
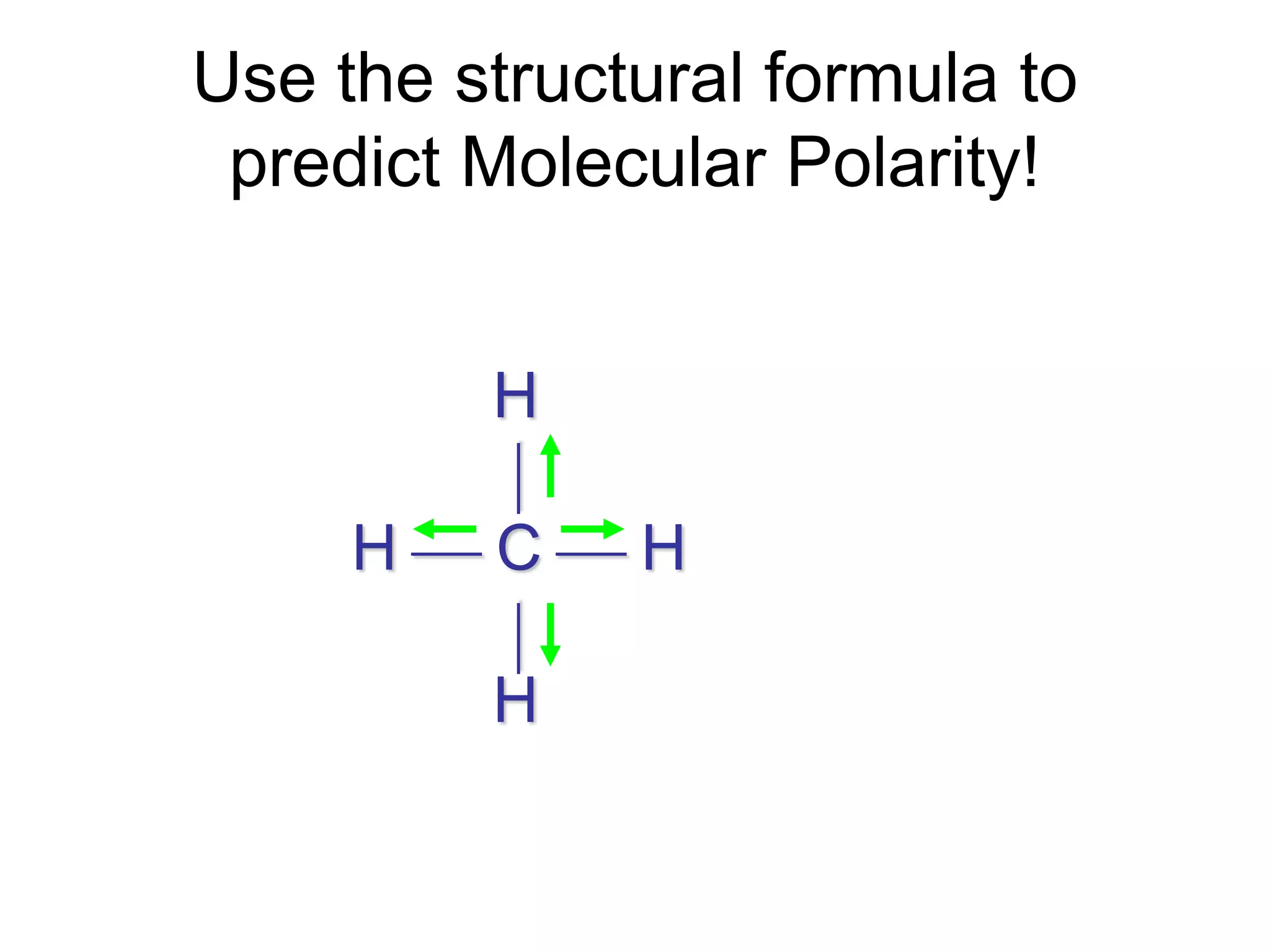
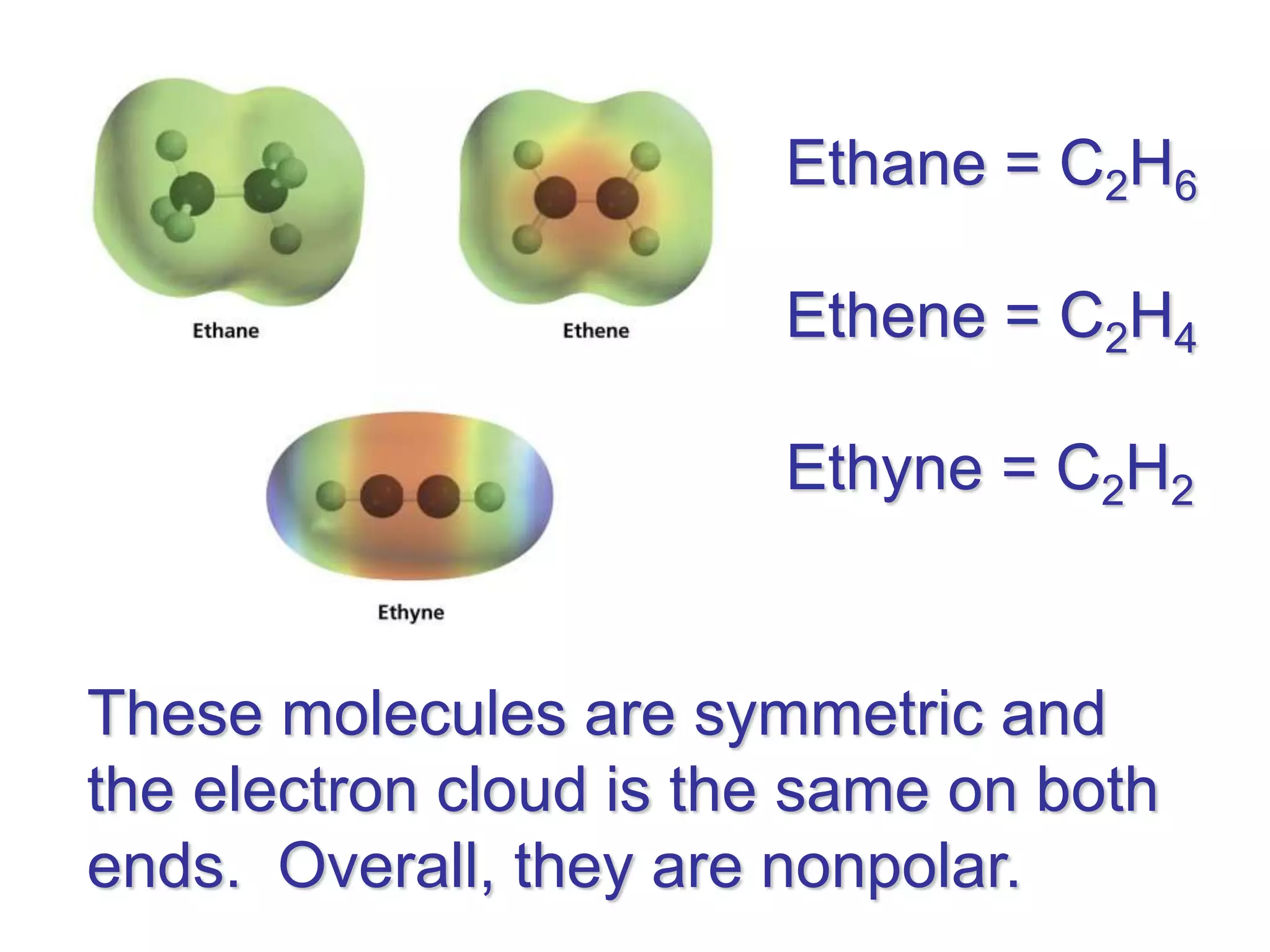
Molecular polarity depends on two factors: the type of bonds in a molecule and the arrangement of those bonds. In diatomic molecules, bond polarity and molecular polarity are the same. However, in larger molecules the arrangement of bonds must be considered. While a molecule may contain polar bonds, it is only polar at the molecular level if the bonds are arranged asymmetrically. Symmetric molecules with polar bonds can be nonpolar overall if the polar bonds cancel each other out. Molecular shape provides clues about polarity, with linear, tetrahedral and bent molecules often being polar and trigonal pyramids always polar.