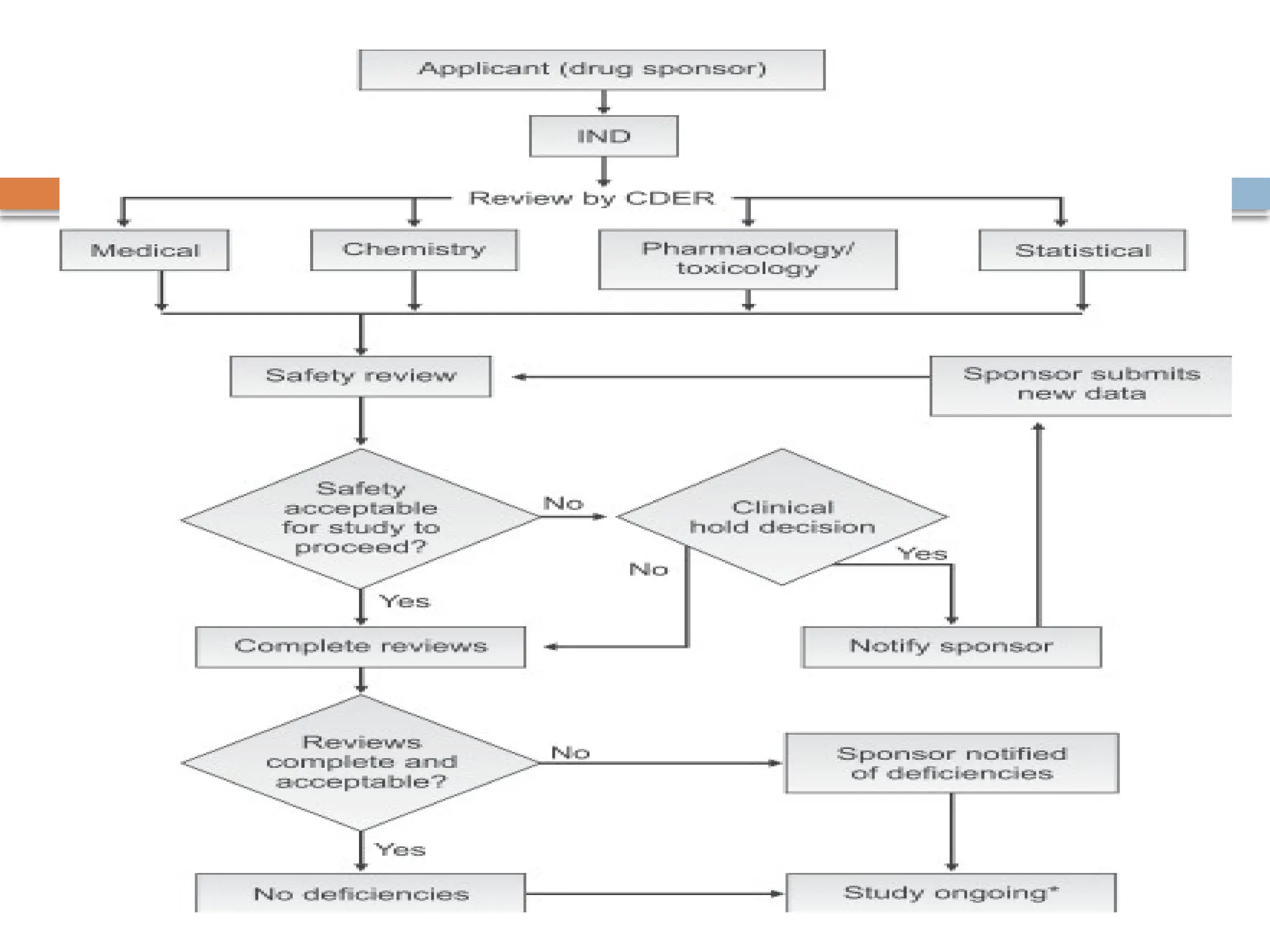
The document details the process of submitting an Investigational New Drug (IND) application, which allows pharmaceutical companies to obtain permission for human clinical trials and shipment of experimental drugs. It encompasses various types of IND applications including investigator, emergency use, treatment, and screening INDs, along with the essential contents needed for submission such as safety studies, manufacturing information, and detailed clinical protocols. The review process by the FDA is outlined, indicating the necessary evaluations to ensure that trials do not expose subjects to unreasonable risks.


























![21CFR Part 312 Investigational New Drug Application
21CFR Part 314
INDA and NDA Applications for FDA Approval to Market a
New Drug (New Drug Approval)
21CFR Part 316 Orphan Drugs
21CFR Part 58 Good Lab Practice for Nonclinical Laboratory [Animal] Studies
21CFR Part 50 Protection of Human Subjects
21CFR Part 56 Institutional Review Boards
21CFR Part 201 Drug Labeling
21CFR Part 54 Financial Disclosure by Clinical Investigators](https://image.slidesharecdn.com/ind-240808153818-41cbe43c/75/Investigational-new-drug-application-pptx-27-2048.jpg)