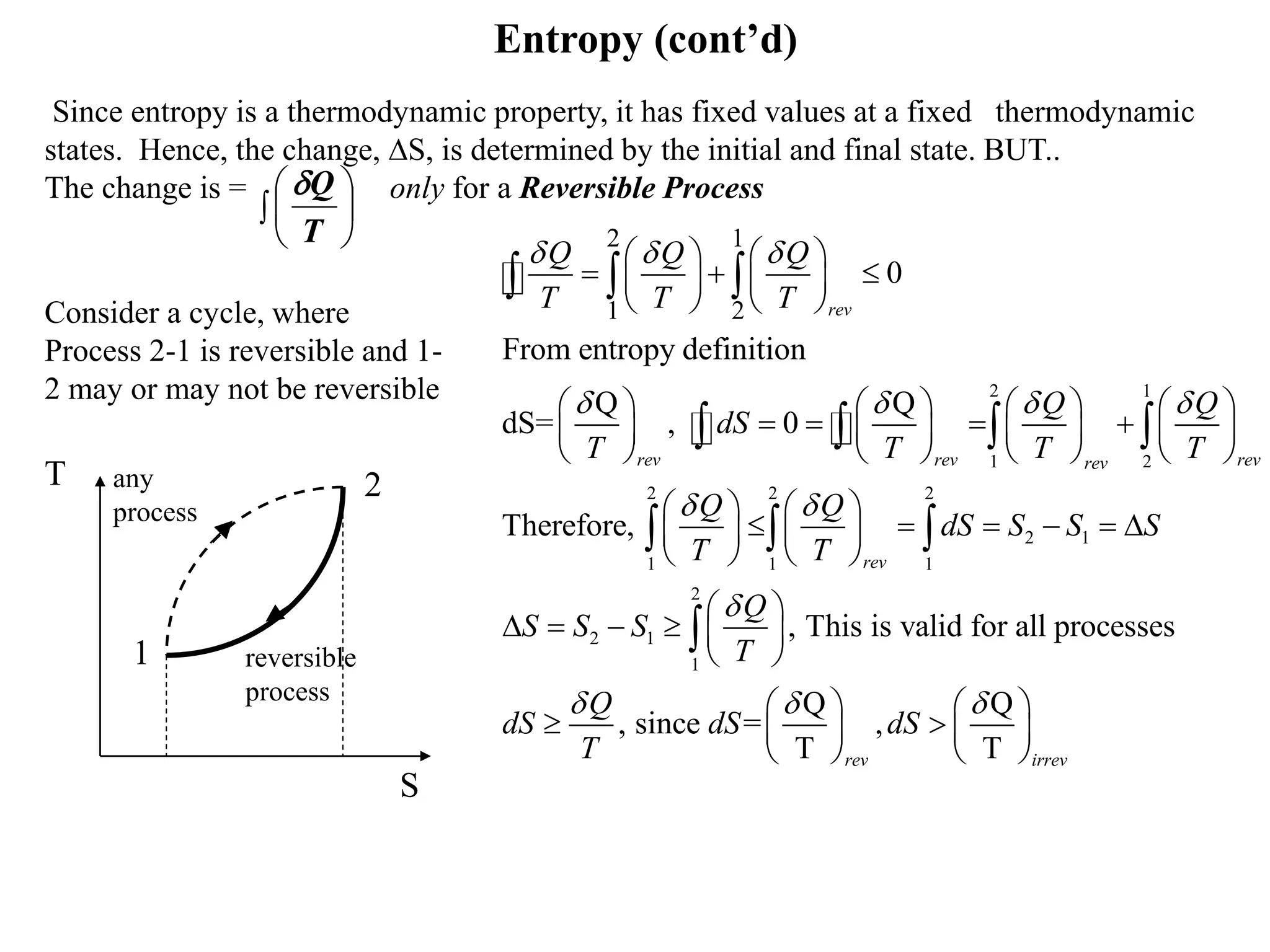
This document discusses entropy, a thermodynamic property that represents disorder in a system. It defines entropy using the Clausius inequality and introduces the increase of entropy principle, which states that entropy always increases or stays the same for isolated systems. The key points made are:
- Entropy is defined using the Clausius inequality and can be viewed as a measure of disorder.
- The increase of entropy principle is another formulation of the second law of thermodynamics and means entropy can increase but not decrease.
- The entropy balance equation accounts for entropy changes, transfers, and generation in thermodynamic processes. Entropy generation must be greater than or equal to zero.