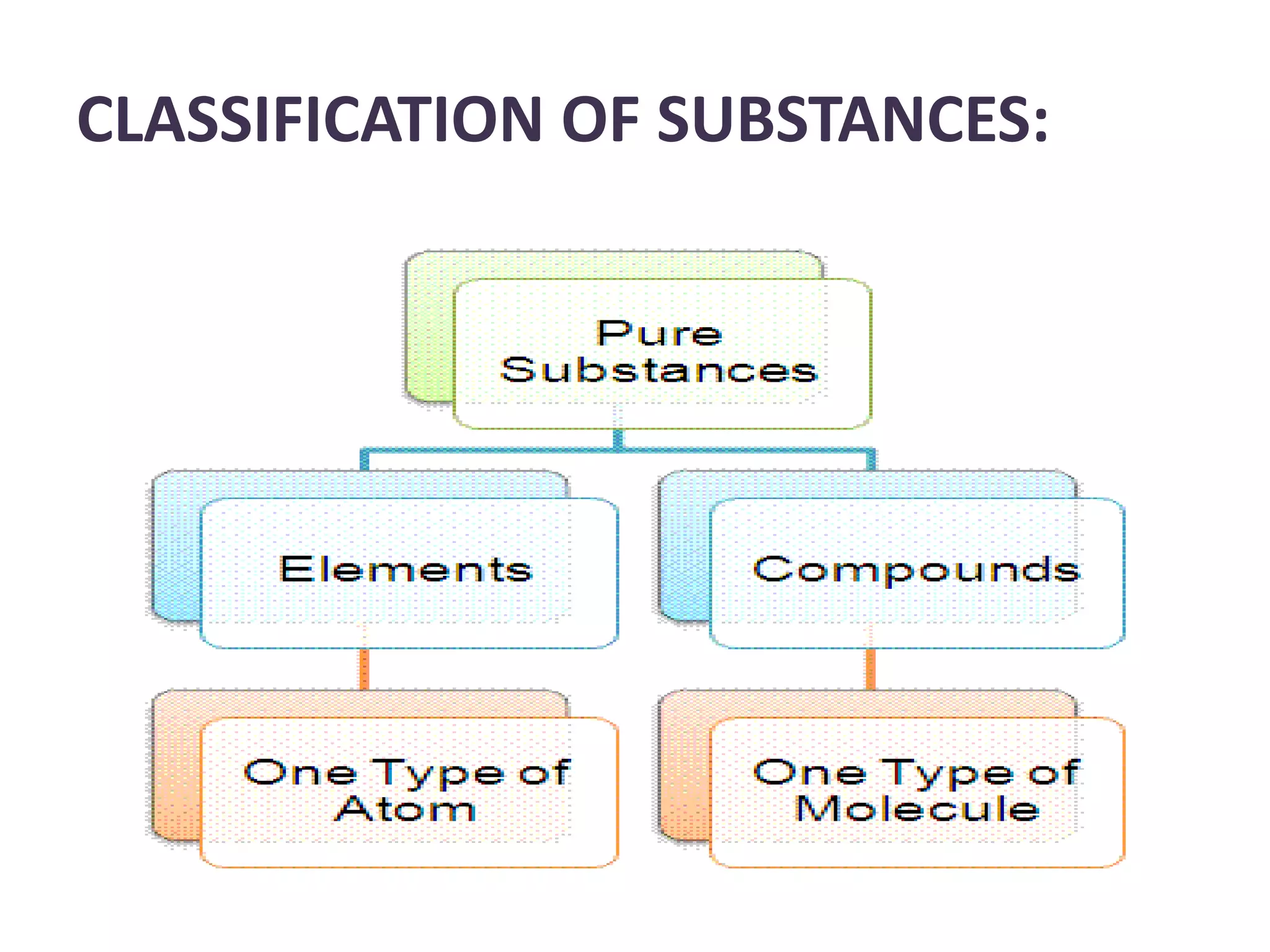
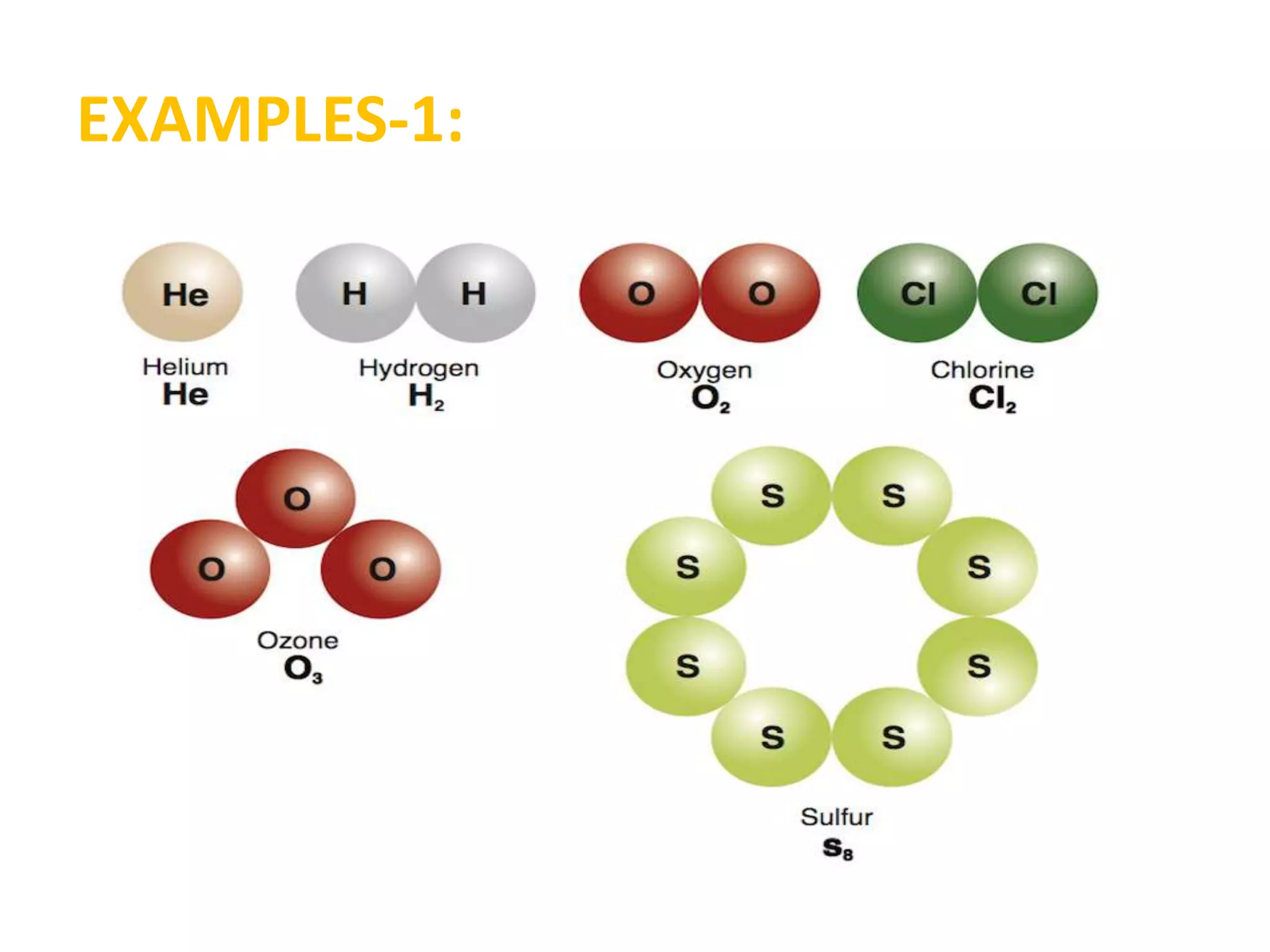
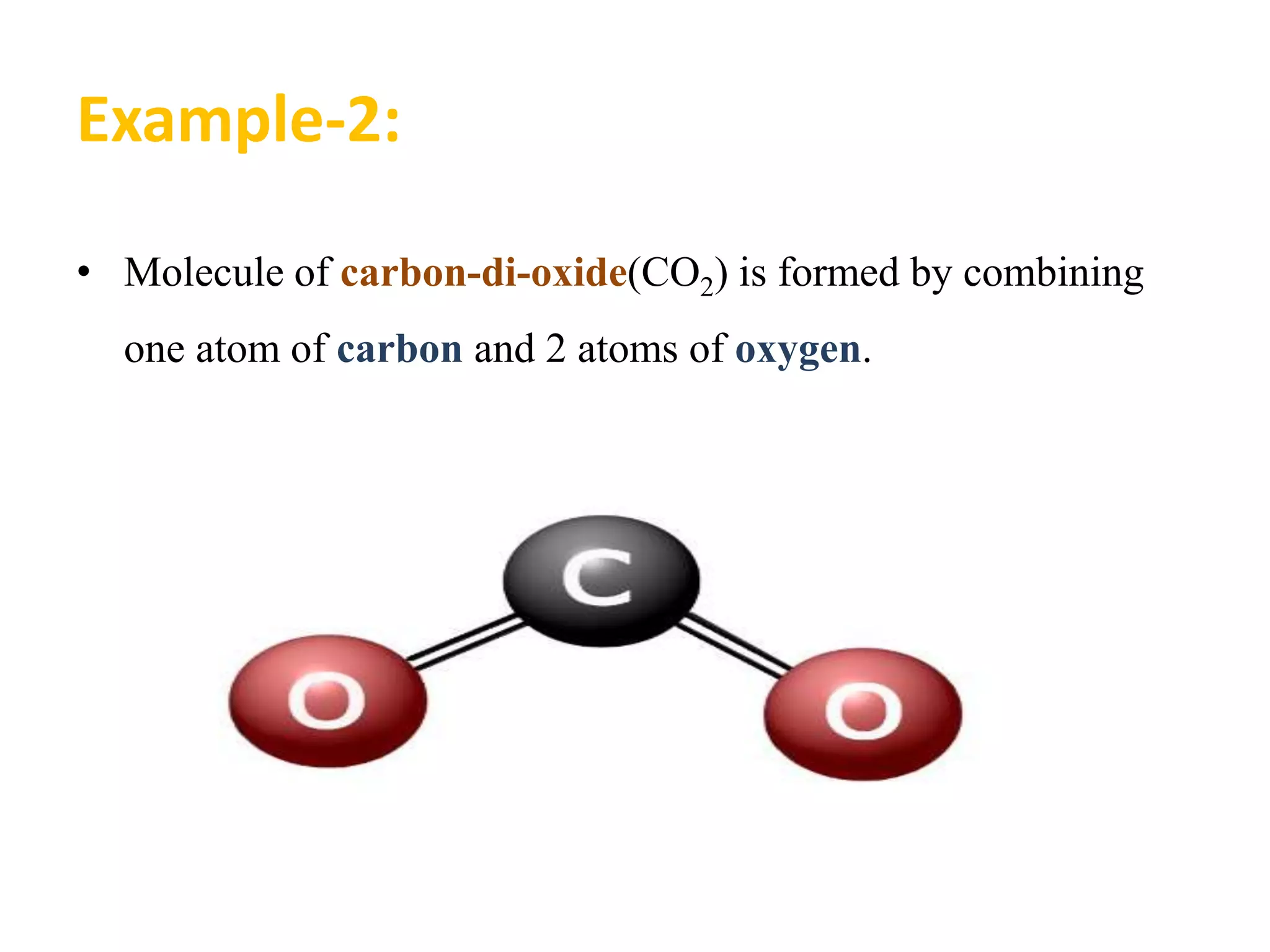
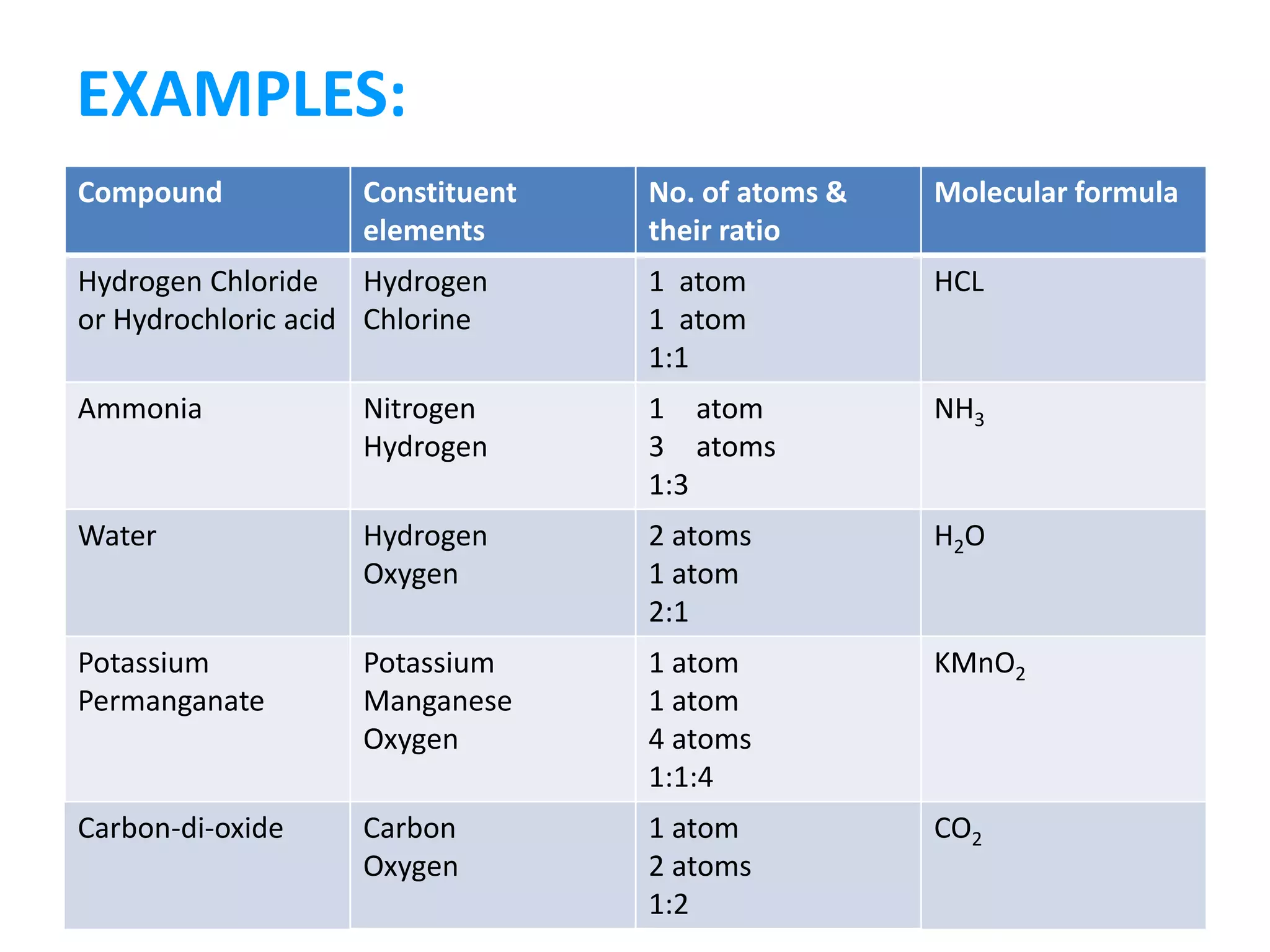
A substance is composed of only one type of atom or molecule. Elements are substances made of the same kind of atom and cannot be broken down further. Examples of elements include gold, silver, and mercury in their solid and liquid states, as well as helium in its gaseous state. Chemical symbols represent elements using letters, often the first one or two letters of their names. Compounds are made of two or more elements combined in a definite ratio, such as water (H2O) which contains hydrogen and oxygen. Molecules form when two or more atoms of elements or compounds combine. Molecular formulas use symbols to represent the elements and numbers of atoms in a molecule, such as CO2 for carbon dioxide.