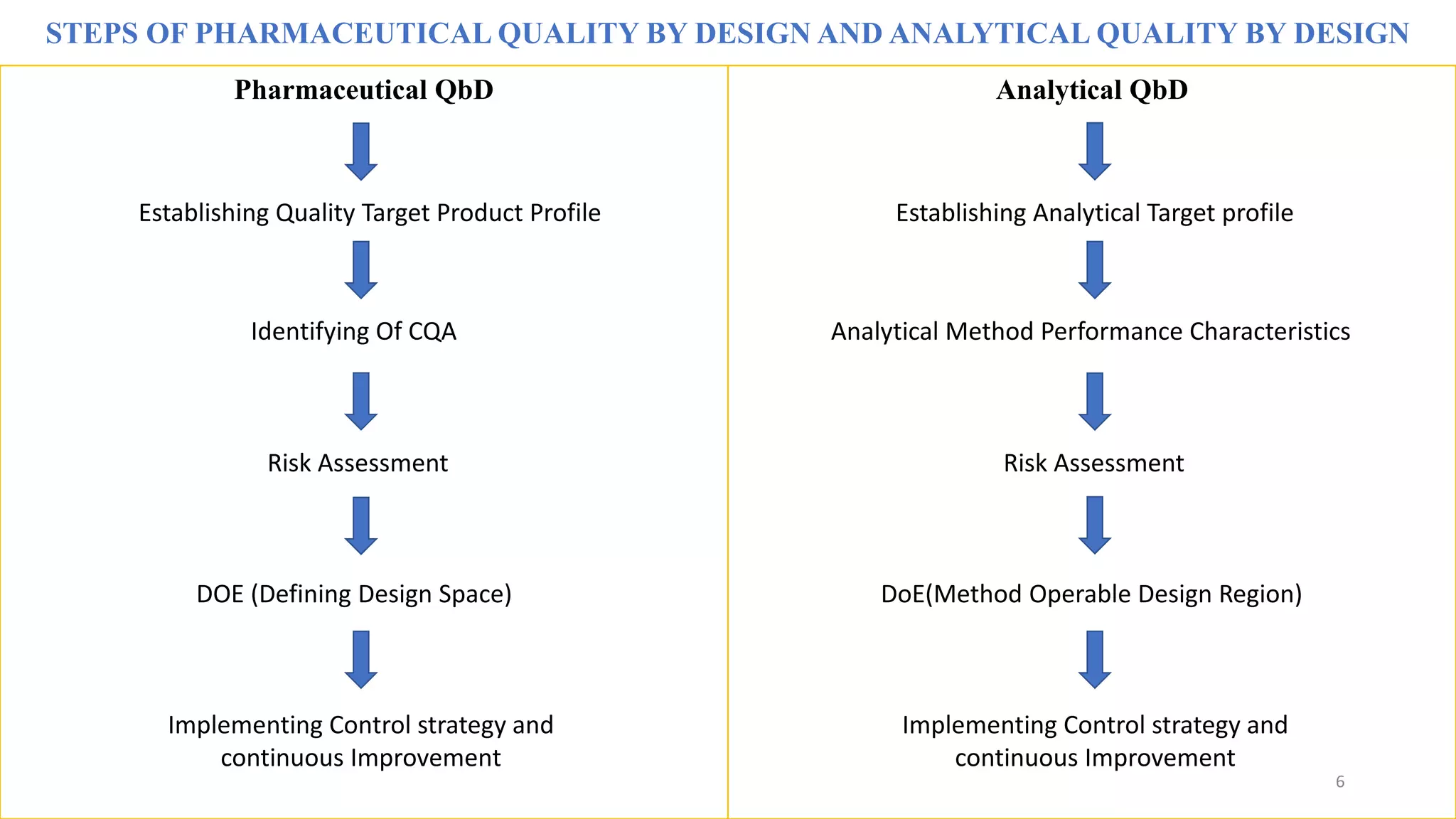
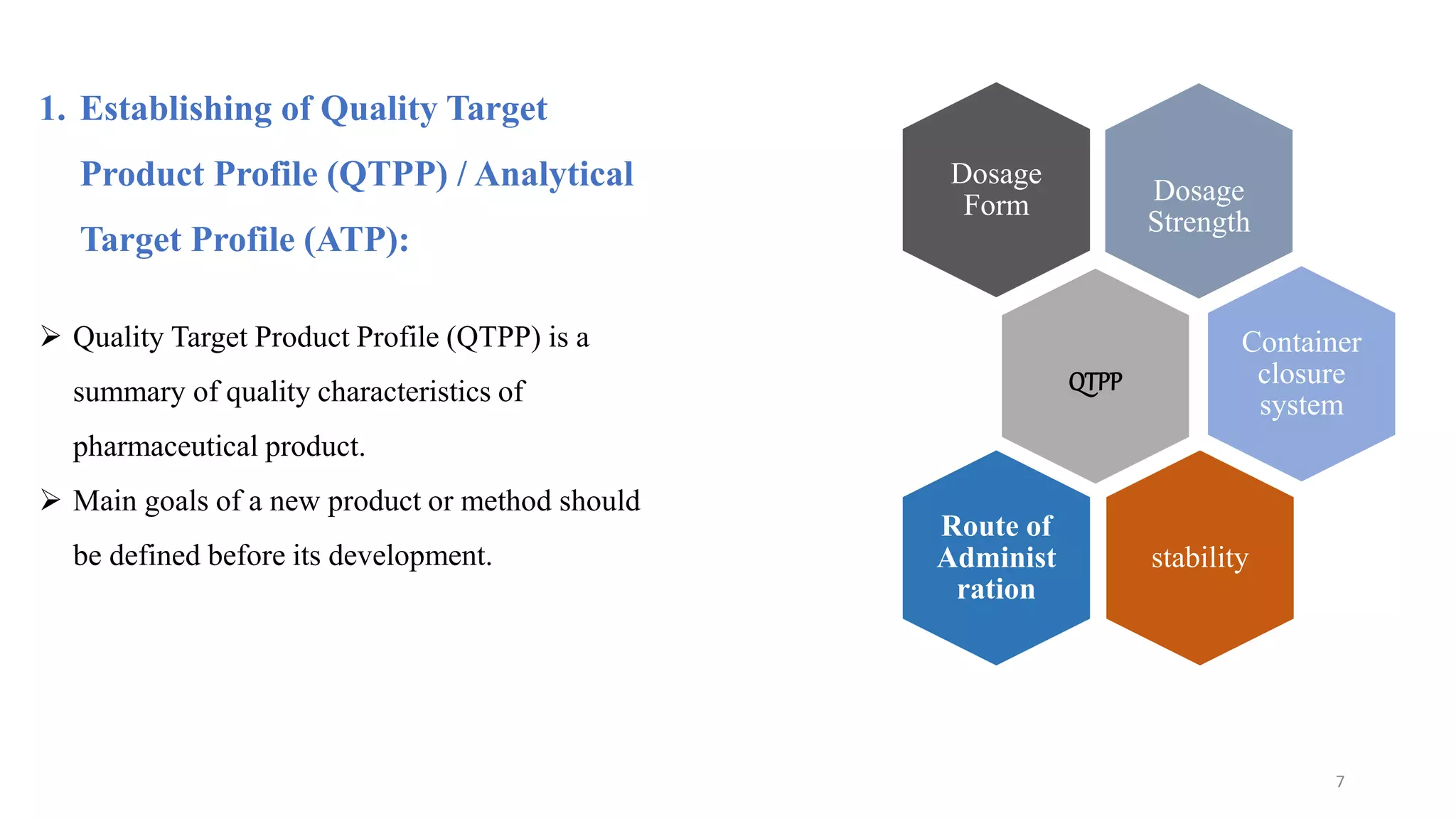
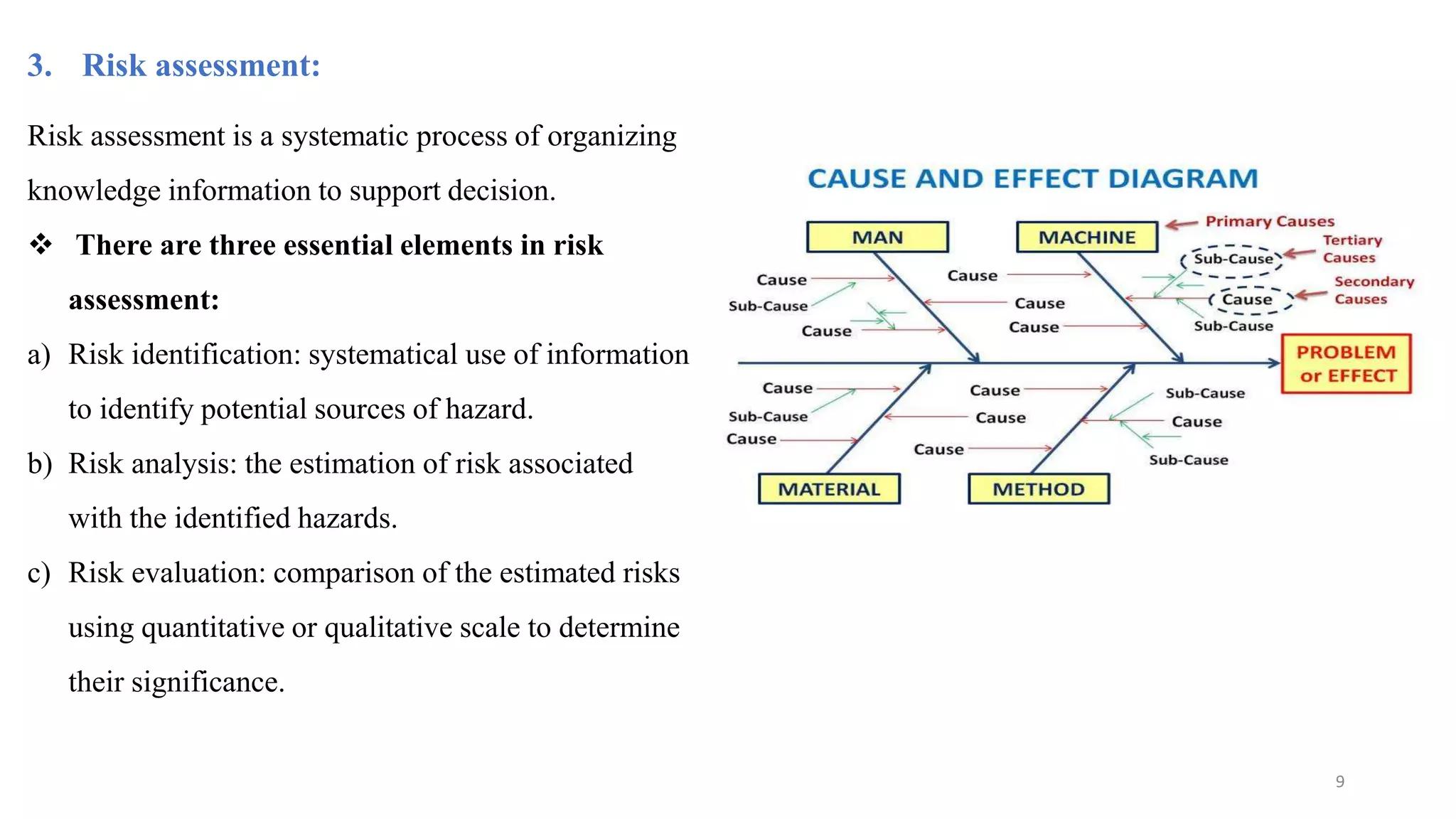
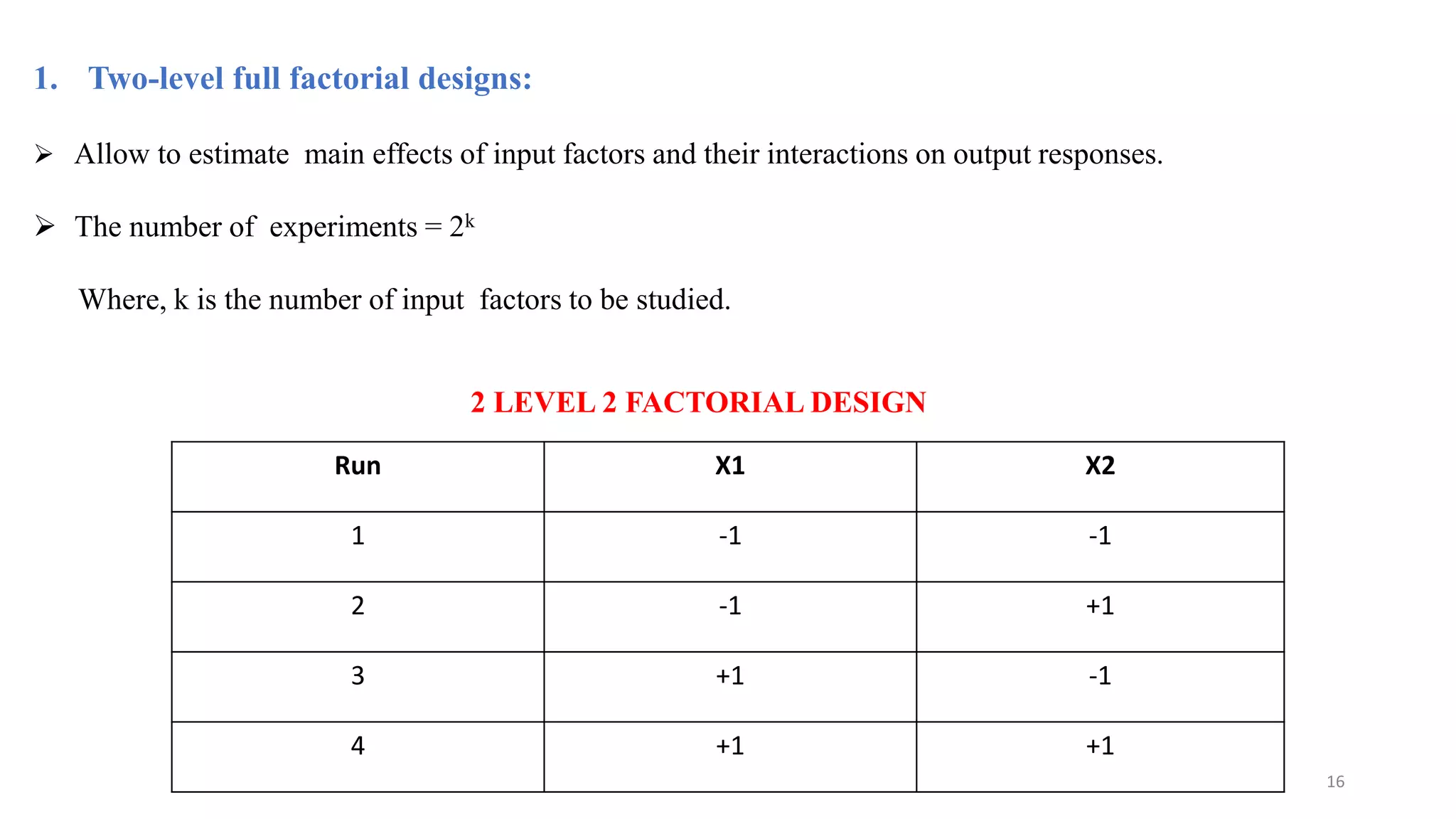
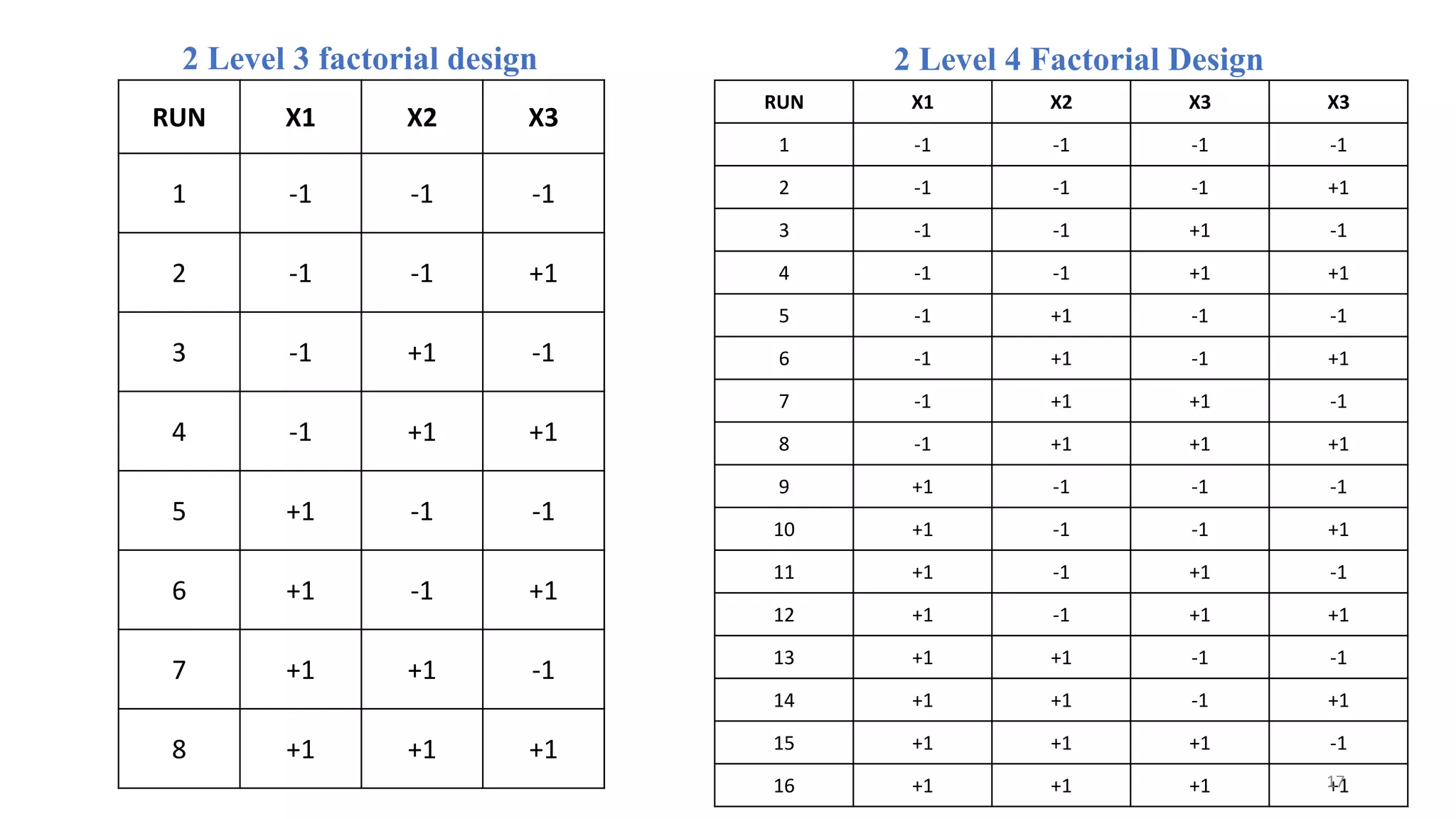
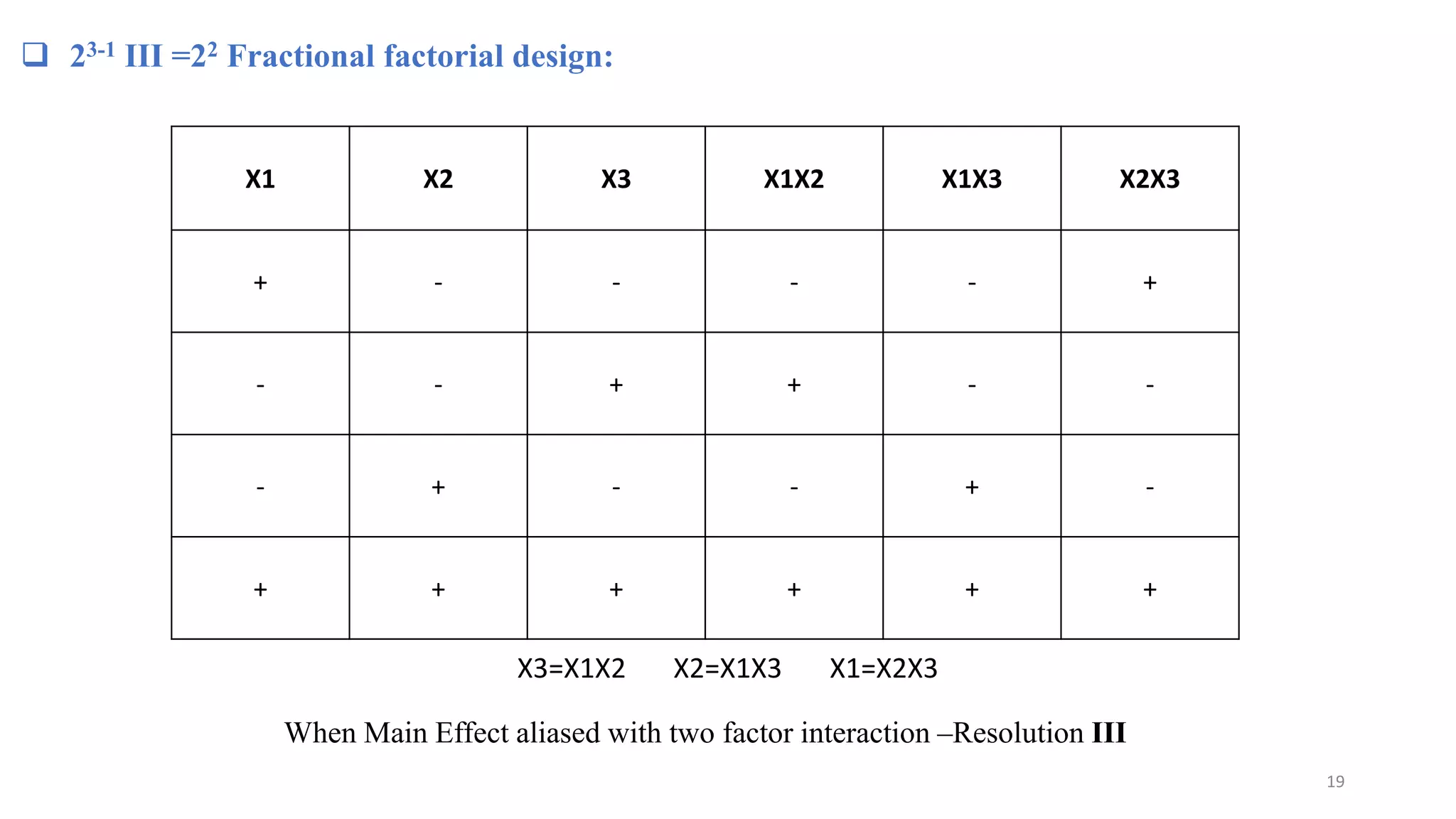
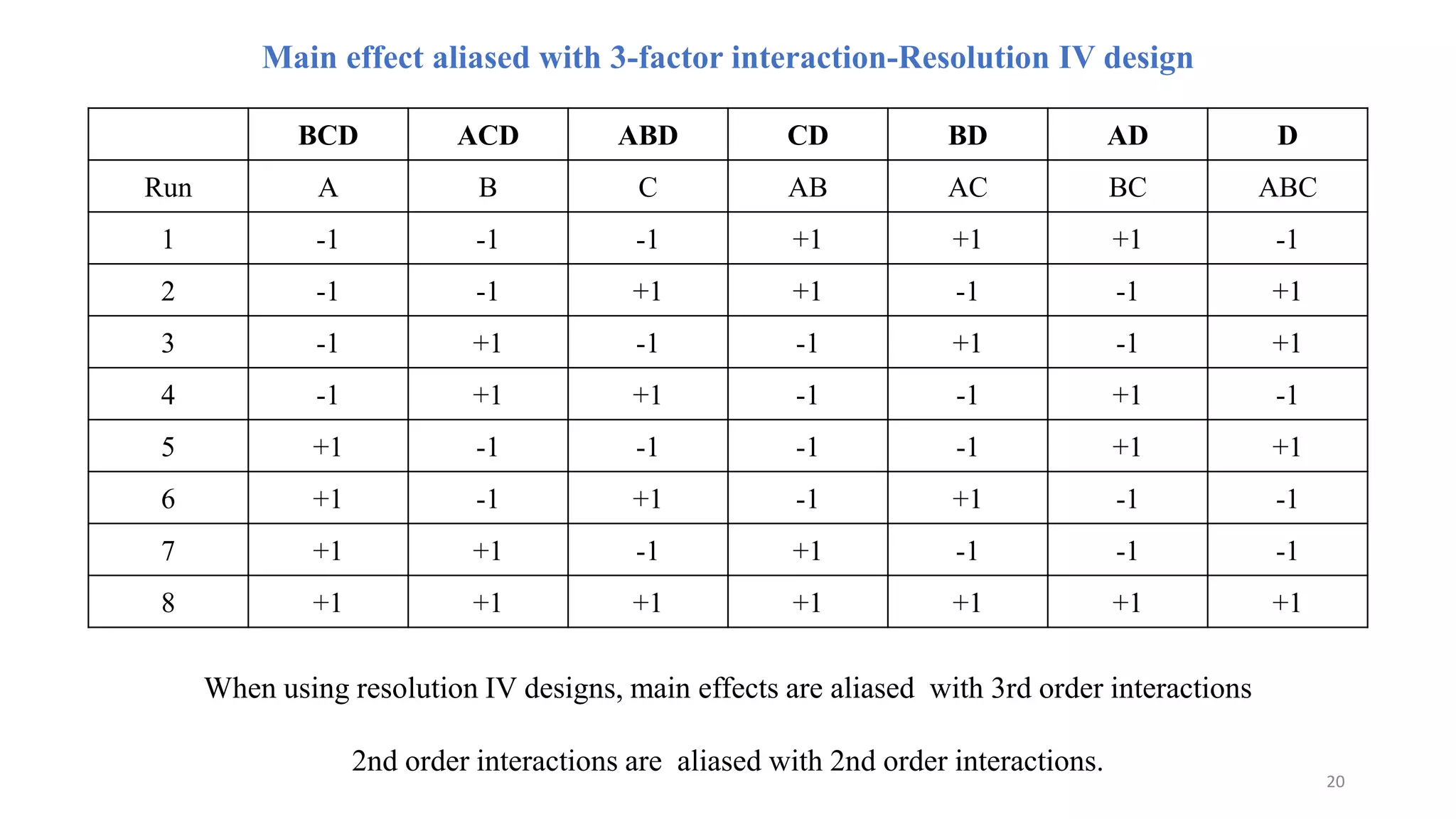
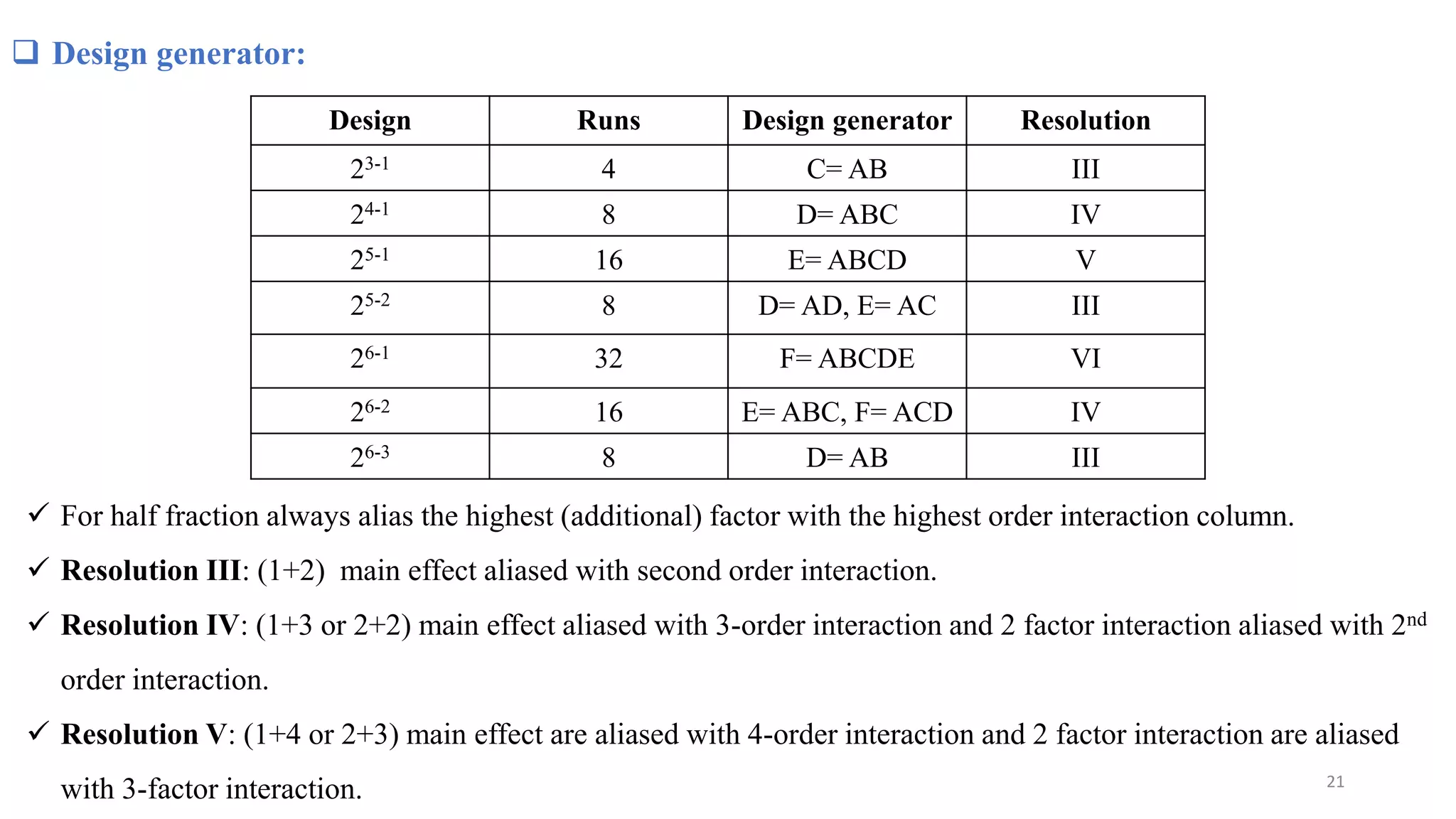
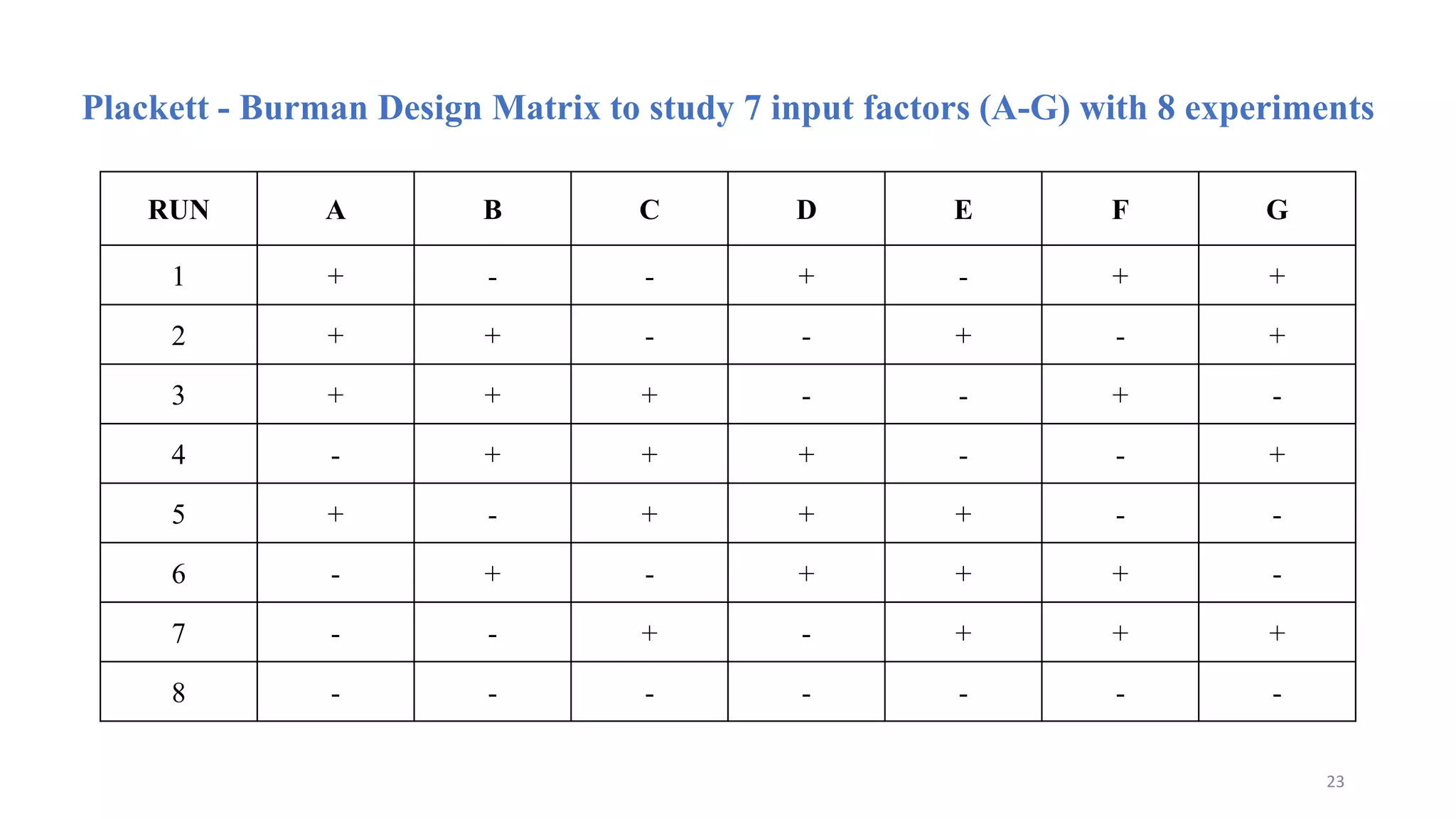
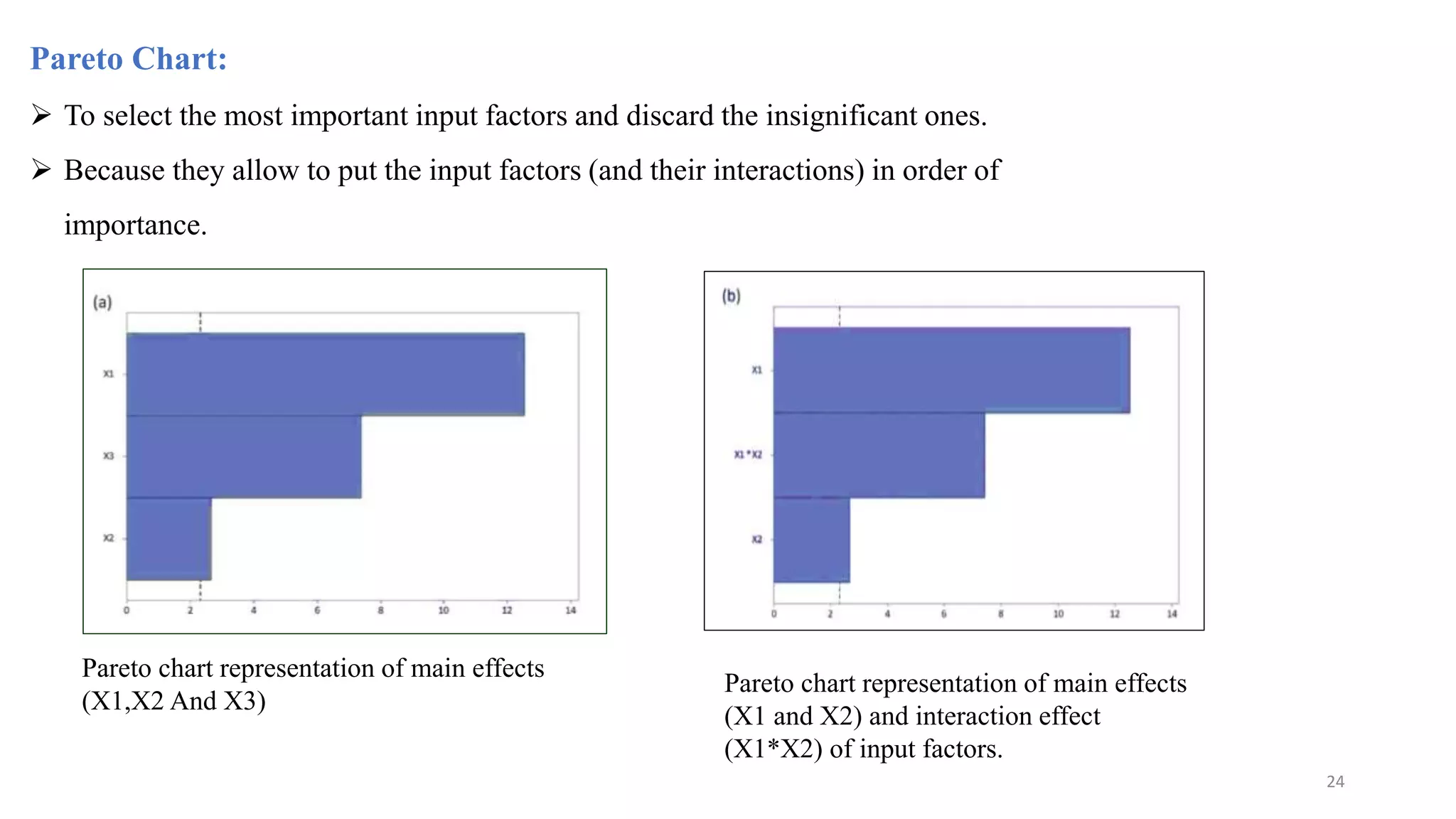
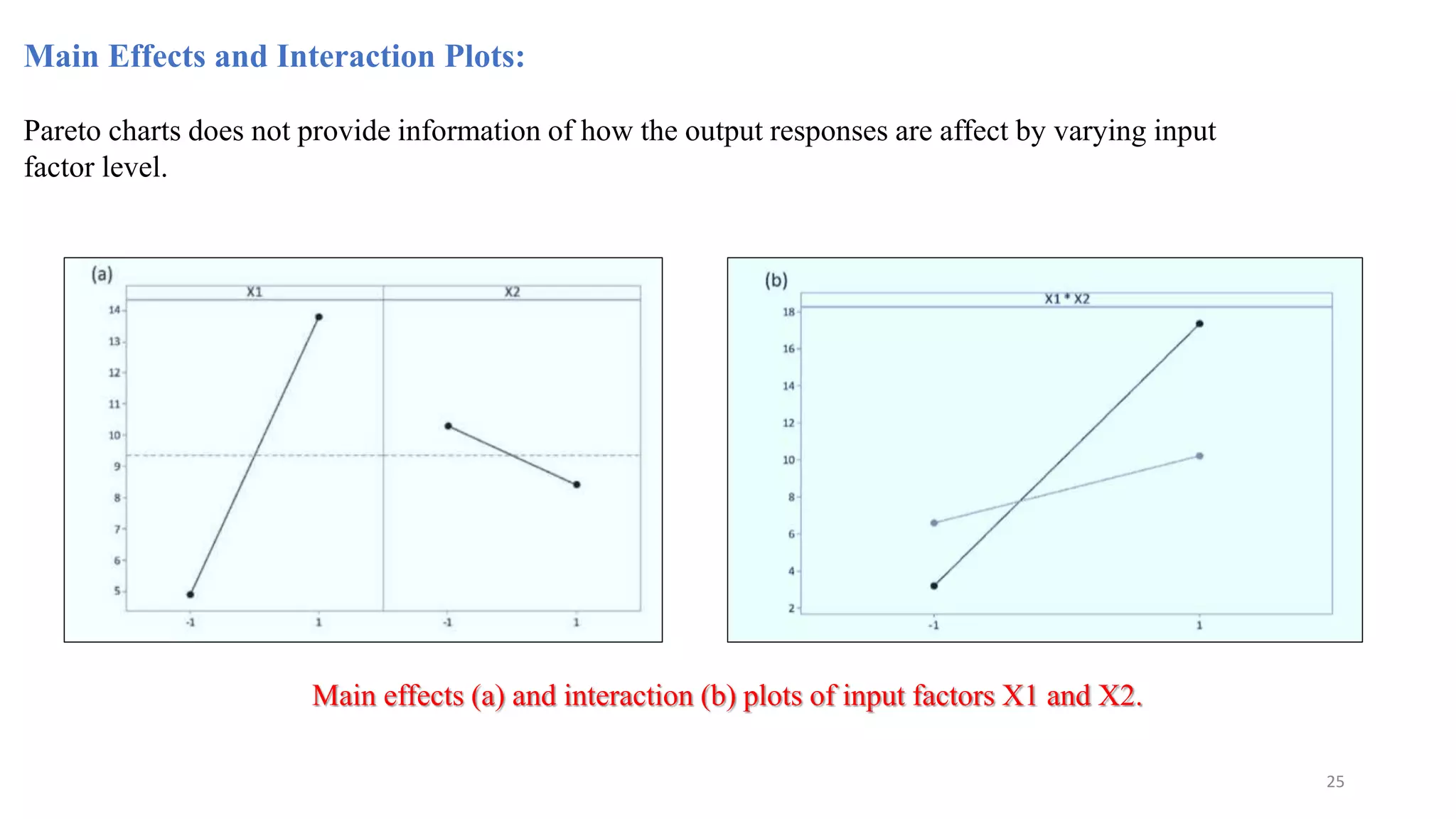
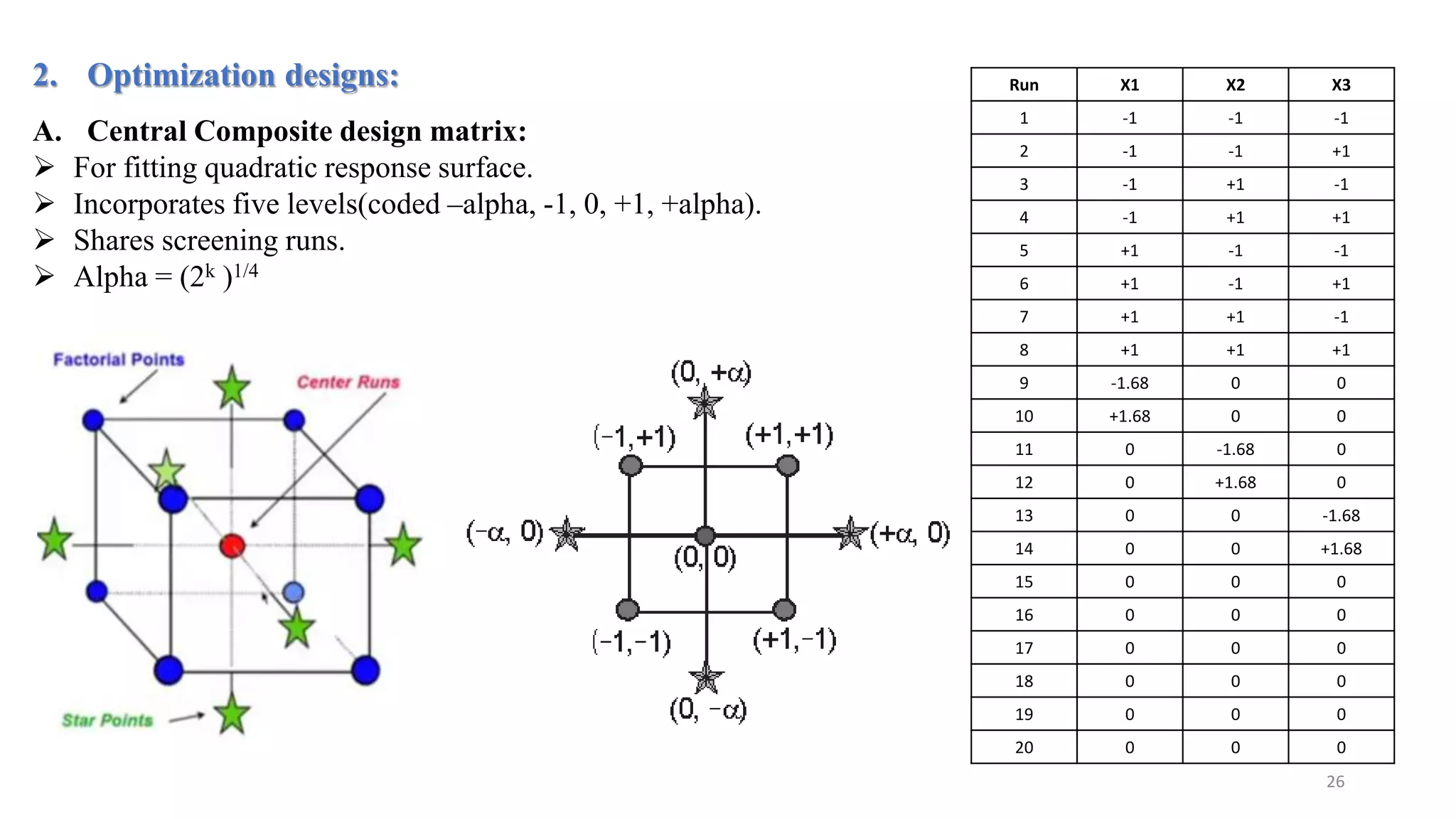
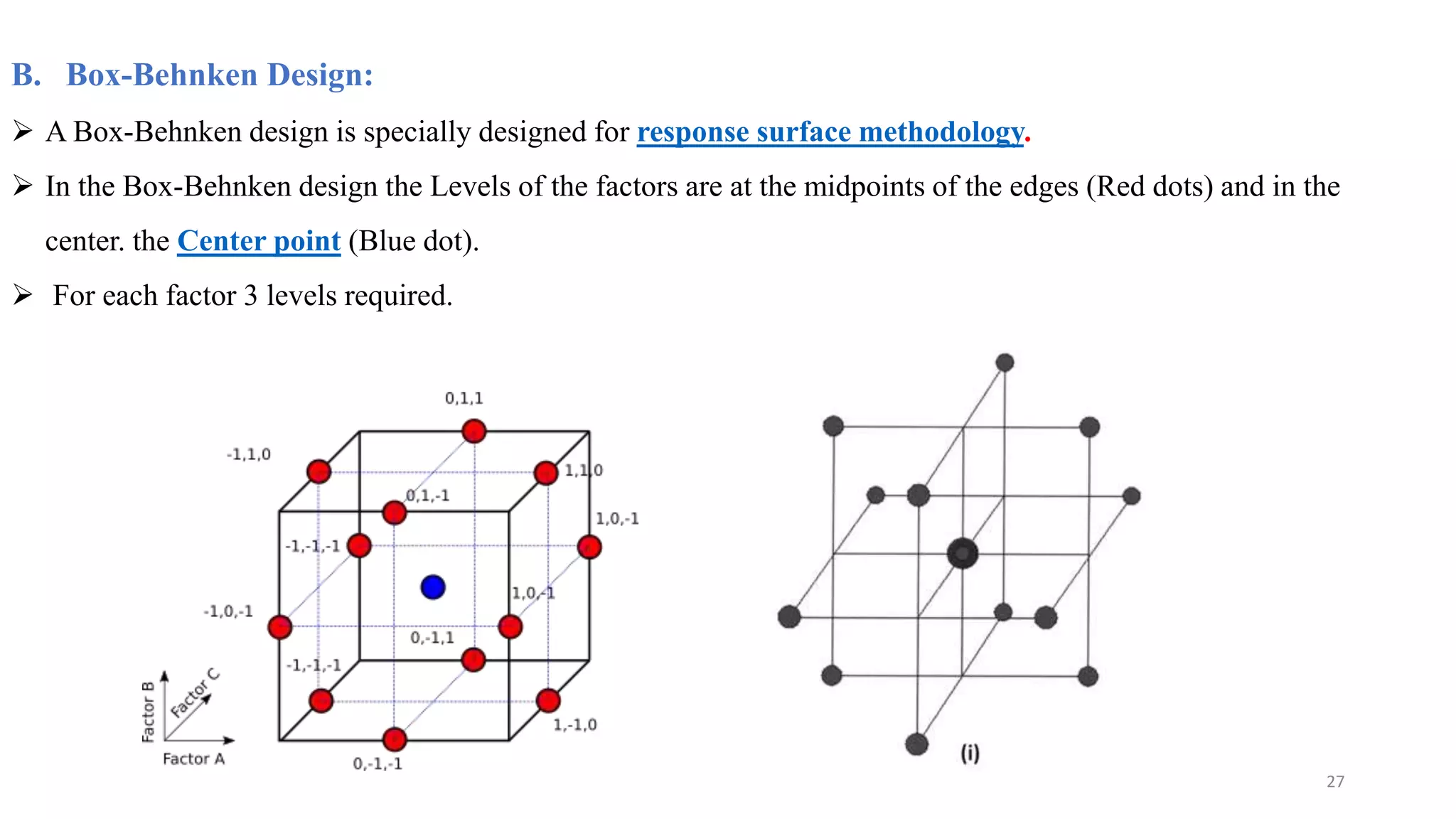
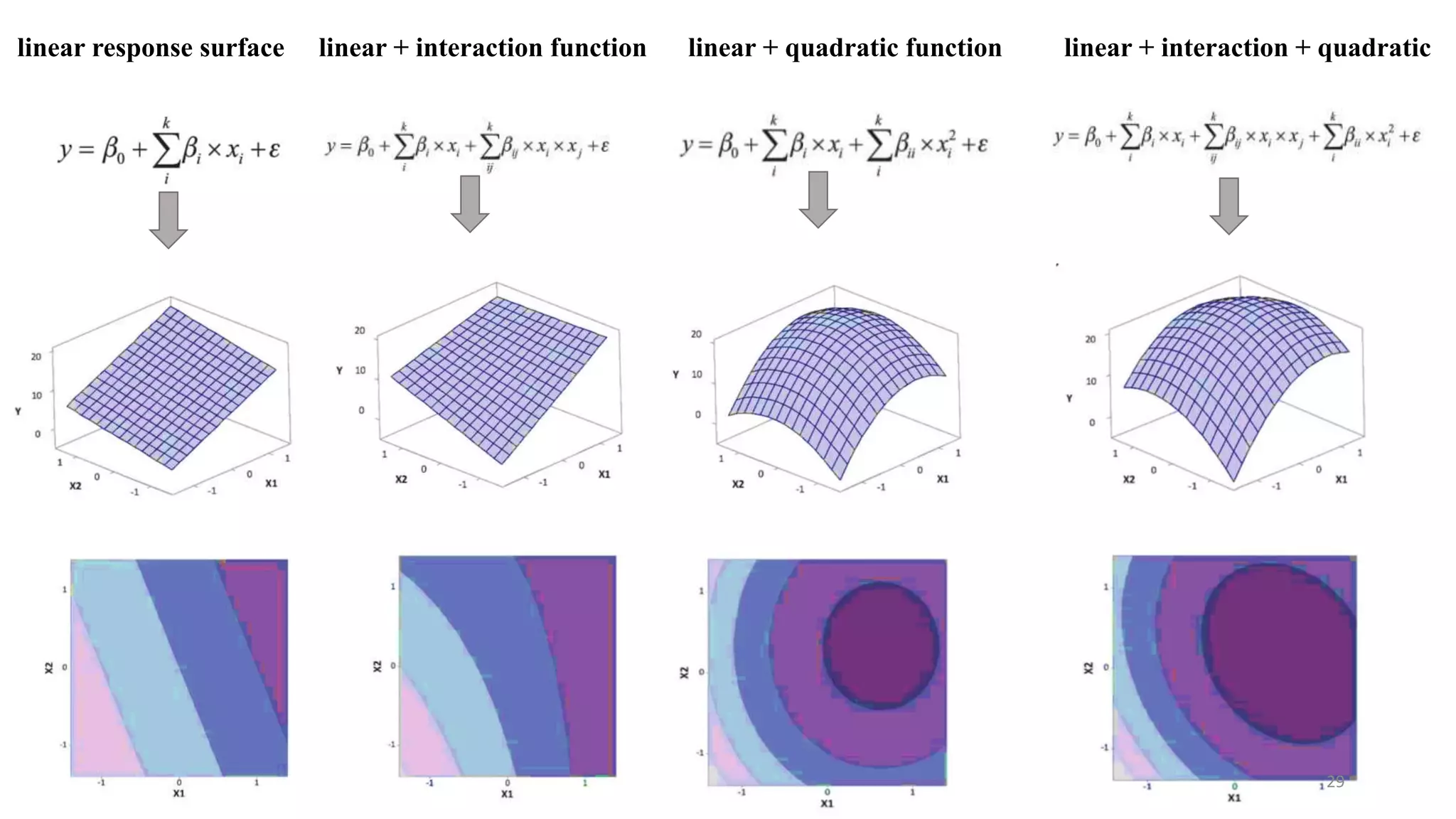
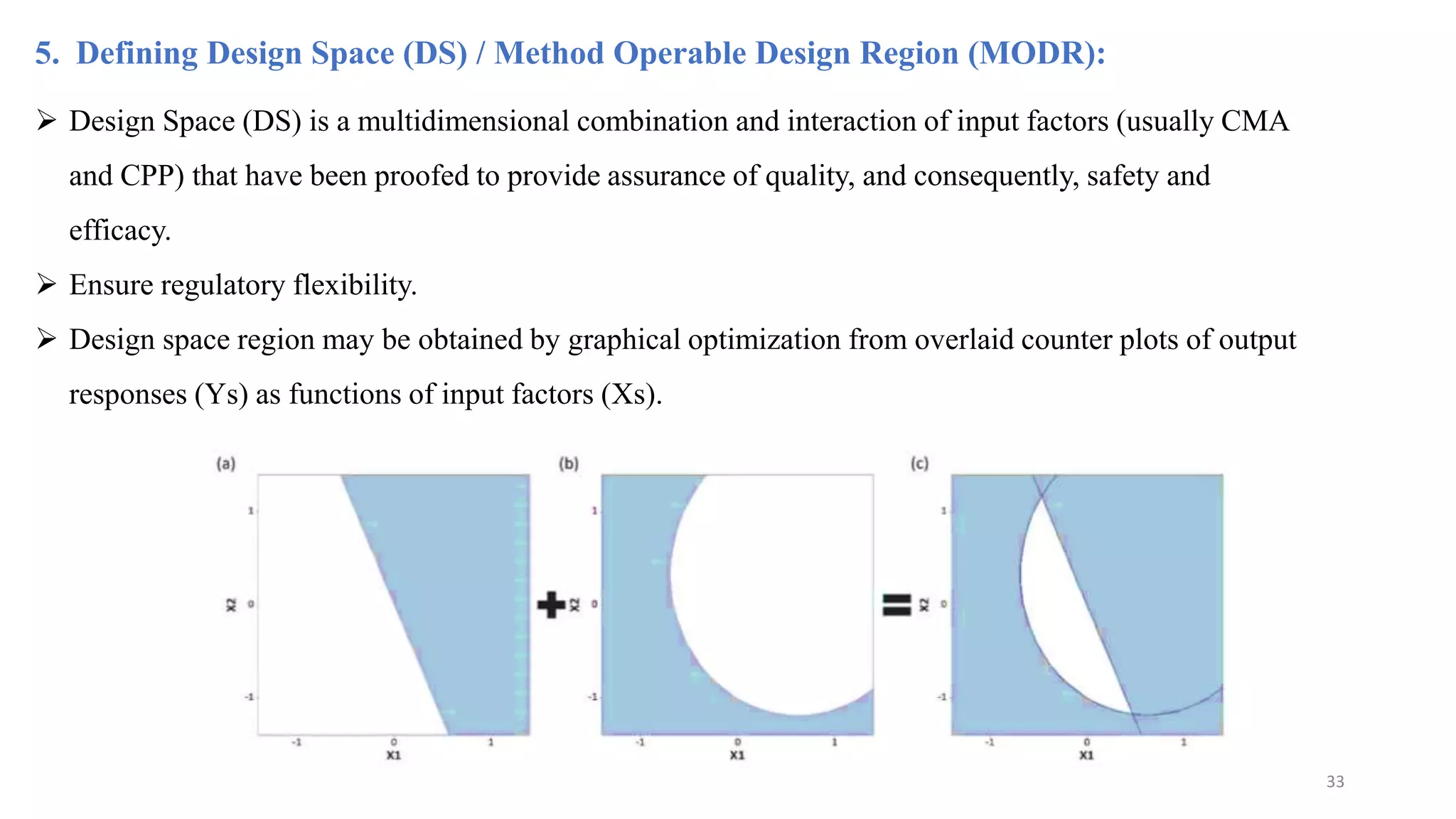
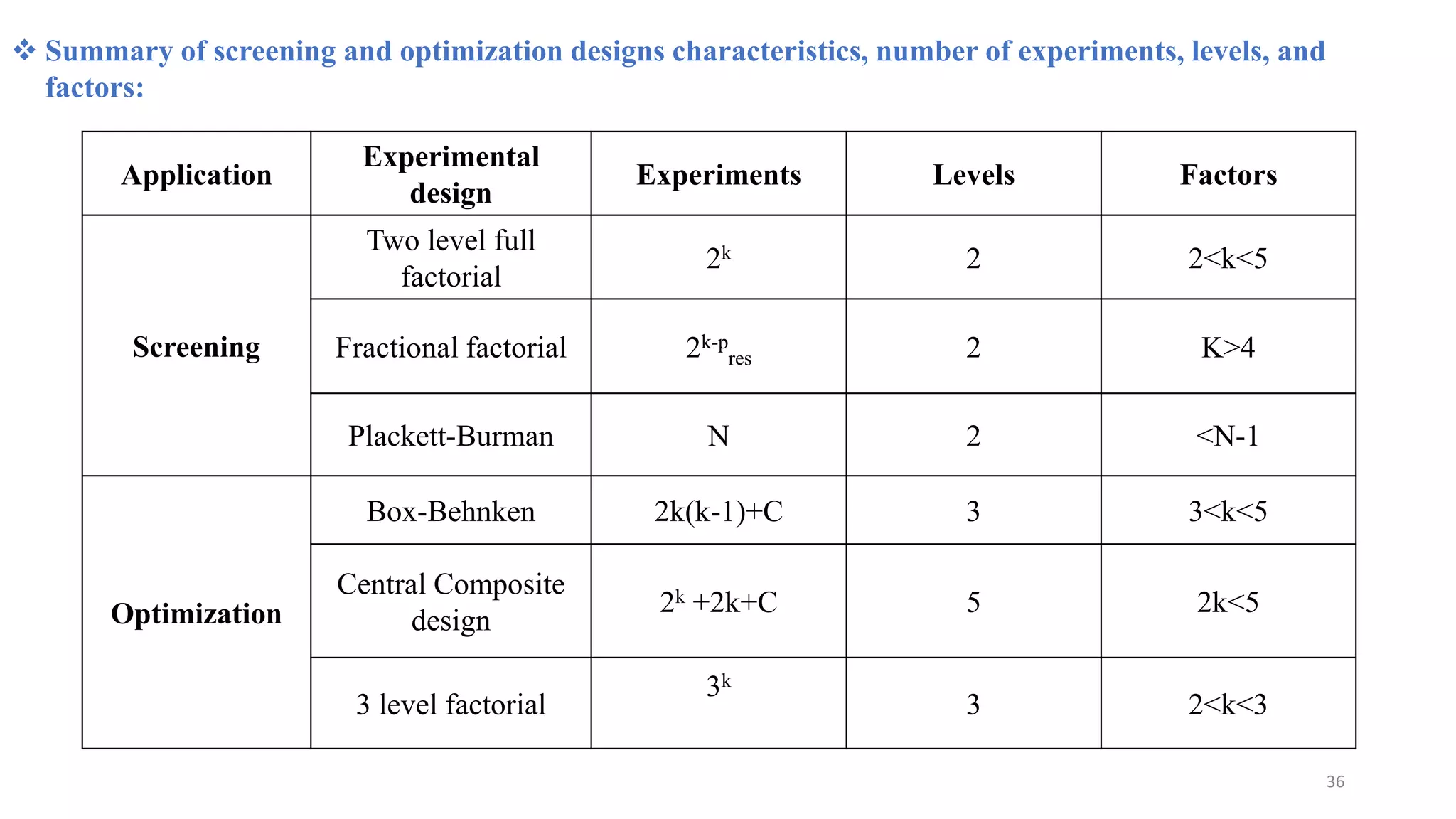
The document discusses the application of Design of Experiments (DOE) in pharmaceutical and analytical Quality by Design (QbD), emphasizing the structured approach to optimizing product quality through systematic experimentation. It covers various experimental designs, including screening and optimization designs, and details the steps necessary to implement QbD, risk assessment, and the statistical methods used to analyze results. Key topics include critical quality attributes, establishing a design space, and the importance of employing DOE to improve product performance and development efficiency.