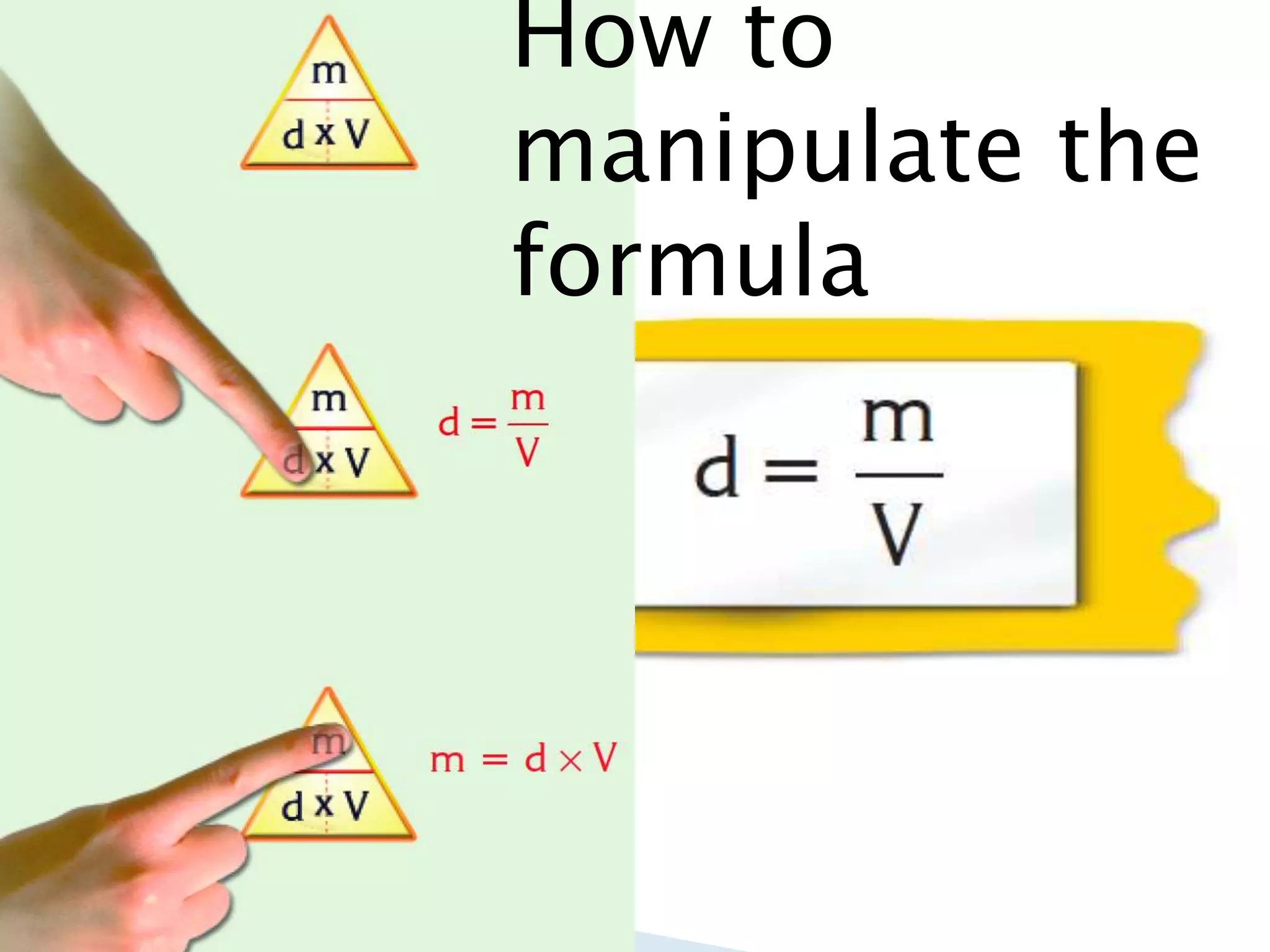
Density is defined as mass per unit volume. The SI unit for density is kg/m3, but g/cm3 is more commonly used. To calculate density, mass is measured using a scale and volume is measured using a ruler or graduated cylinder for irregular objects. Density can be used to differentiate between substances and is a characteristic property of matter. The density of a mixture is calculated by dividing the total mass of the mixture by the total volume.