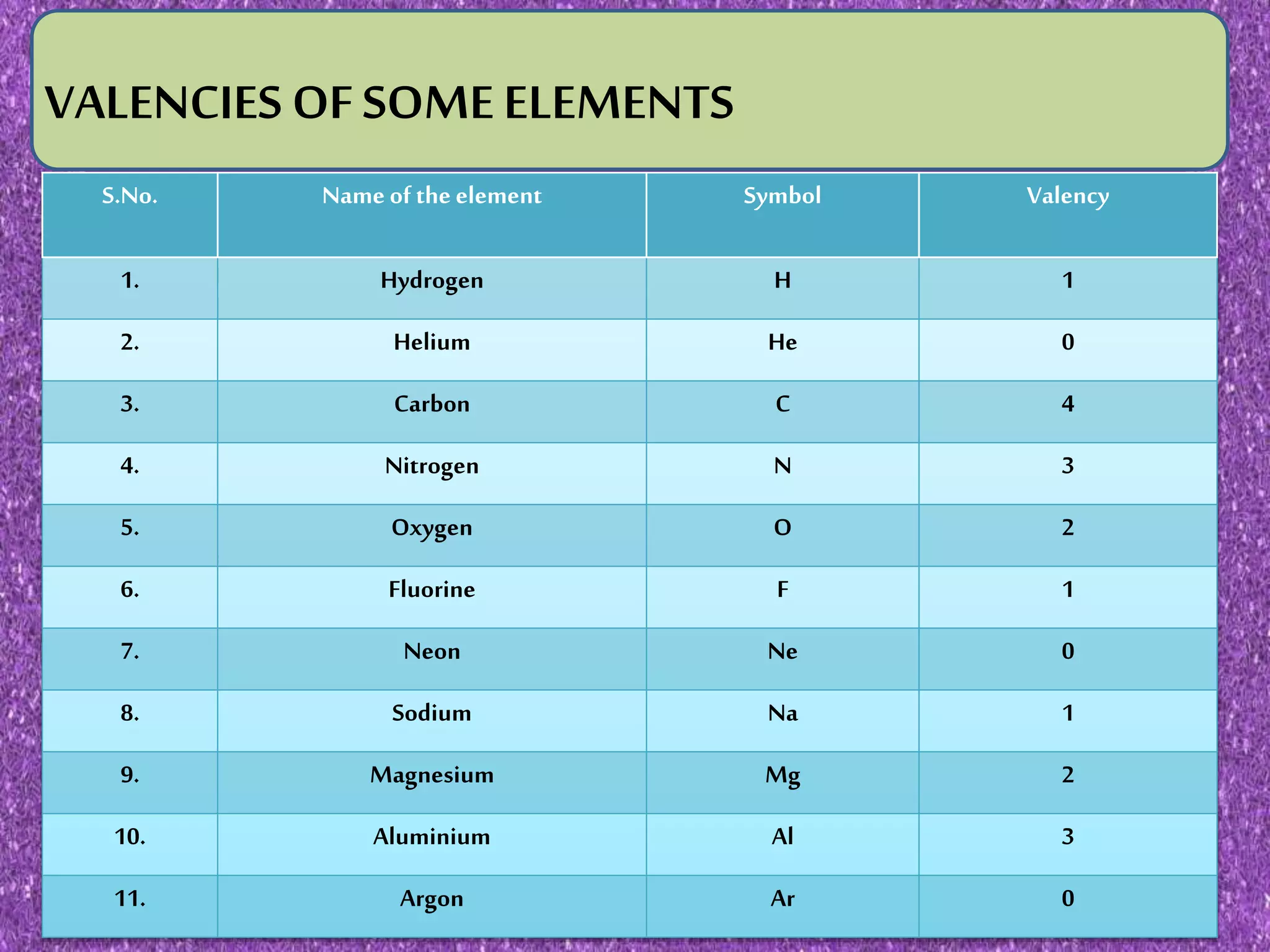
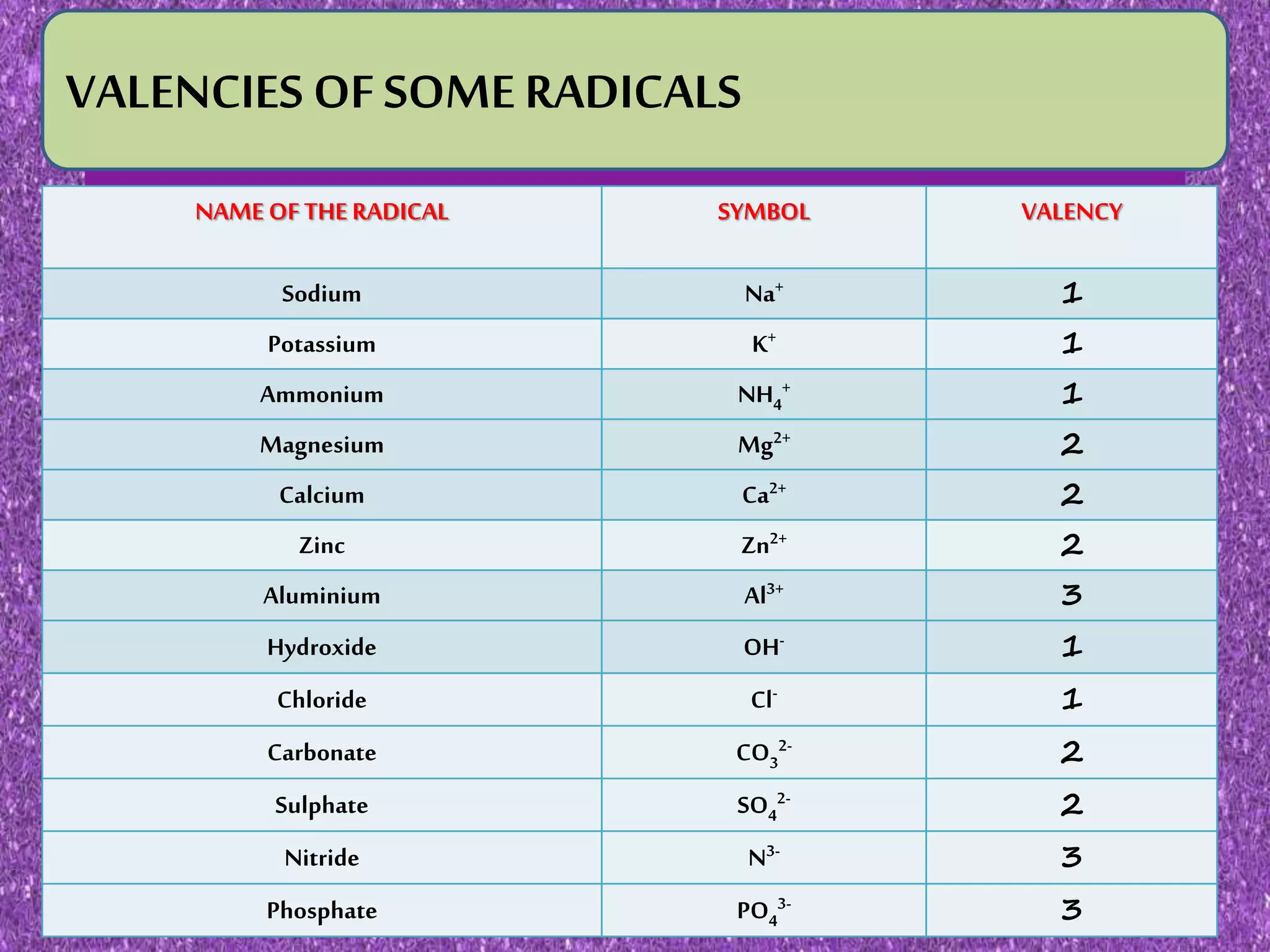
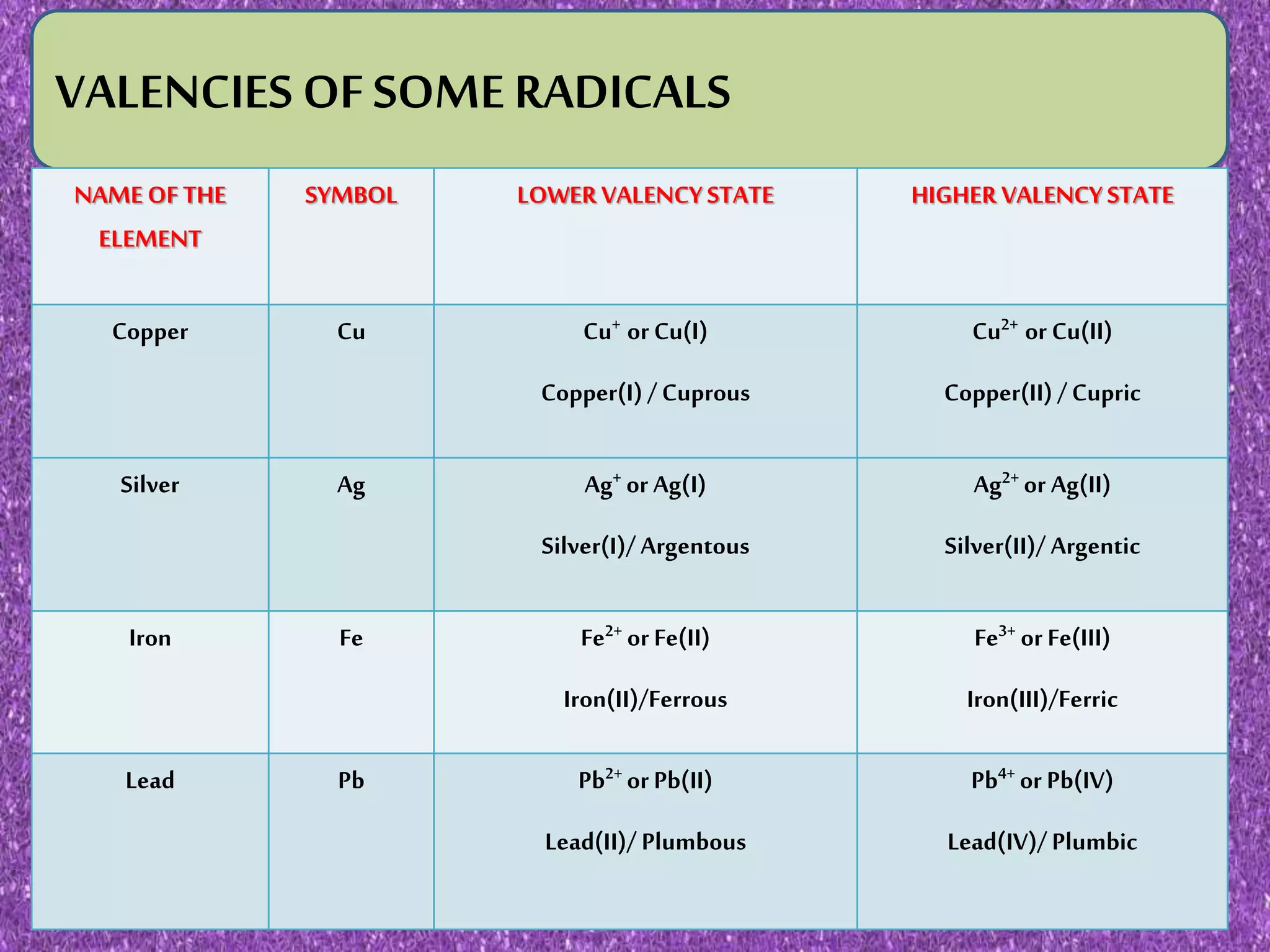
This document discusses valency and variable valency in chemistry. It defines valency as the combining capacity of an element or radical. It then lists the valencies of some common elements and radicals. The document explains that elements showing more than one valency exhibit variable valency, like iron which can be +2 or +3. It describes how lower and higher valencies are denoted, either by adding suffixes like "-ous" and "-ic", or using roman numerals in brackets. Finally, it gives examples of common elements that exhibit variable valency, such as copper, silver, and iron.