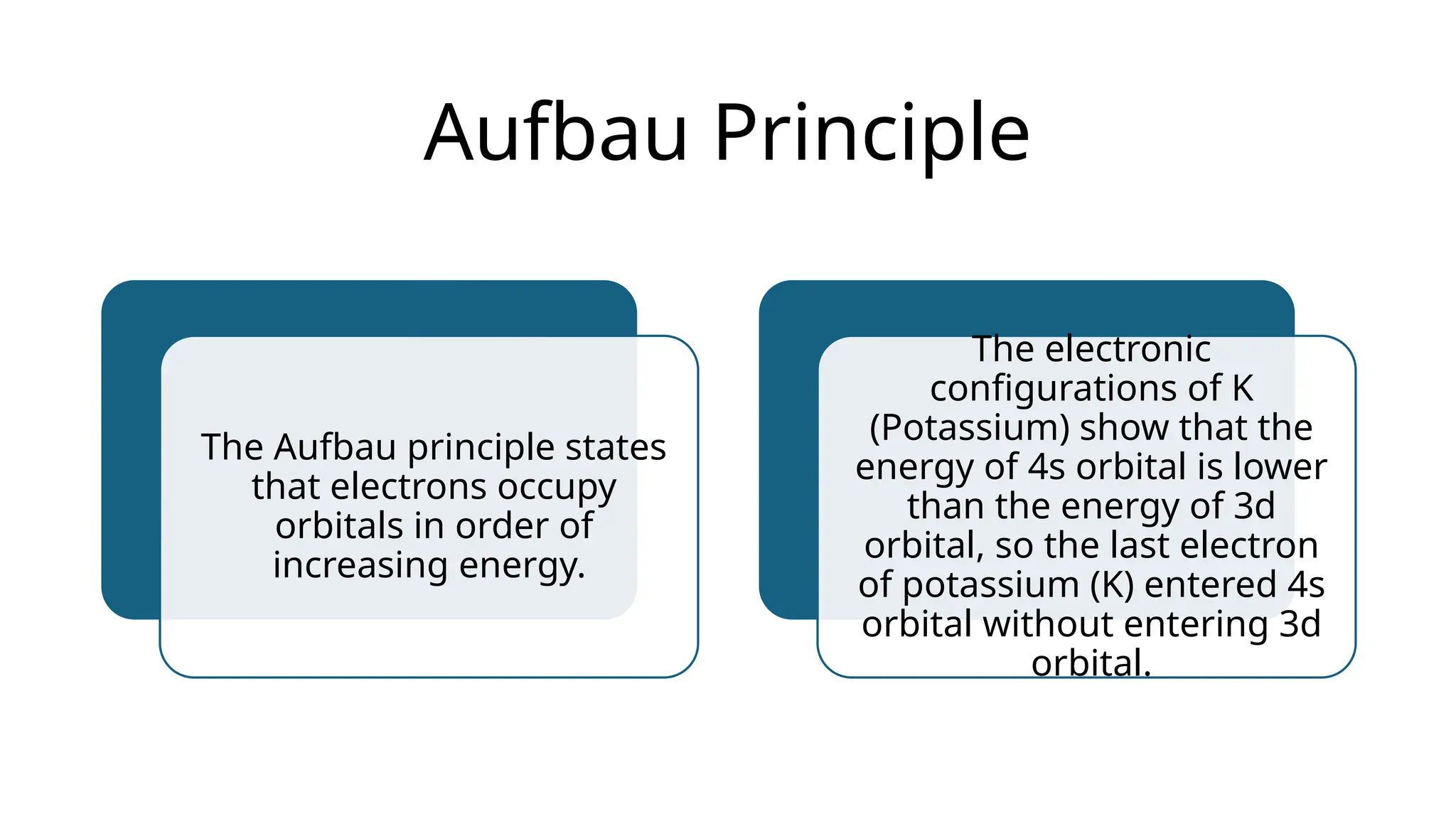
The document outlines key concepts in atomic structure, focusing on atomic models developed by Rutherford and Bohr, along with fundamental particles and atomic properties such as atomic number, isotopes, and quantum numbers. It discusses Rutherford's conclusion of a dense, positively charged nucleus and the limitations of his model, which Bohr addressed by introducing stable electron orbits and energy levels. However, Bohr's model also has limitations, particularly in explaining multi-electron atoms and spectral lines.