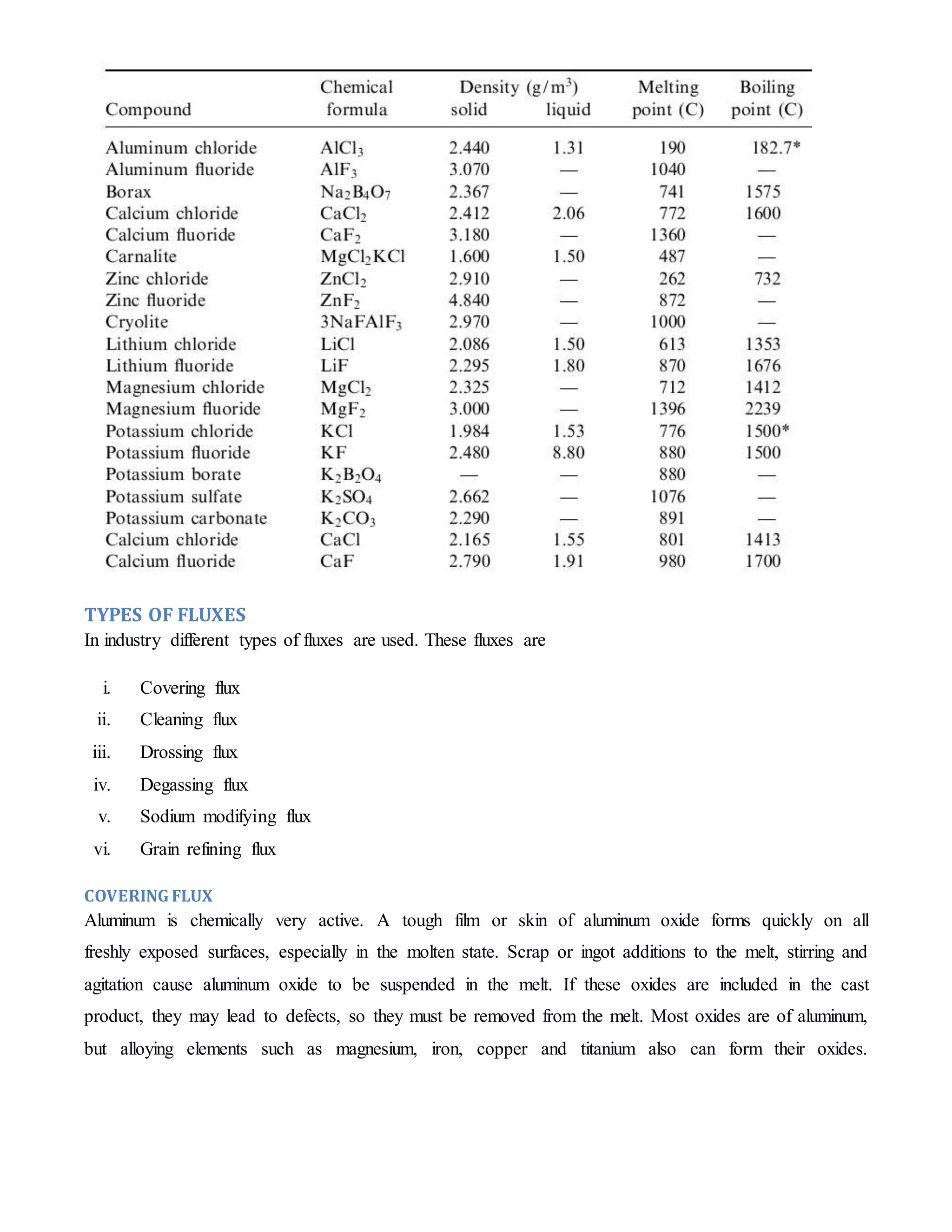
This document discusses different types of fluxes used in aluminum casting processes. It describes fluxes as mixtures that facilitate removing impurities from molten aluminum alloys. The main types are covering fluxes to prevent oxidation, cleaning fluxes to remove oxides, drossing fluxes to promote separating trapped aluminum from dross, and degassing fluxes containing chlorine and fluorine salts to remove hydrogen by forming gas bubbles. Each flux is designed for a specific purpose based on its chemical composition and reactions with impurities in the molten alloy.