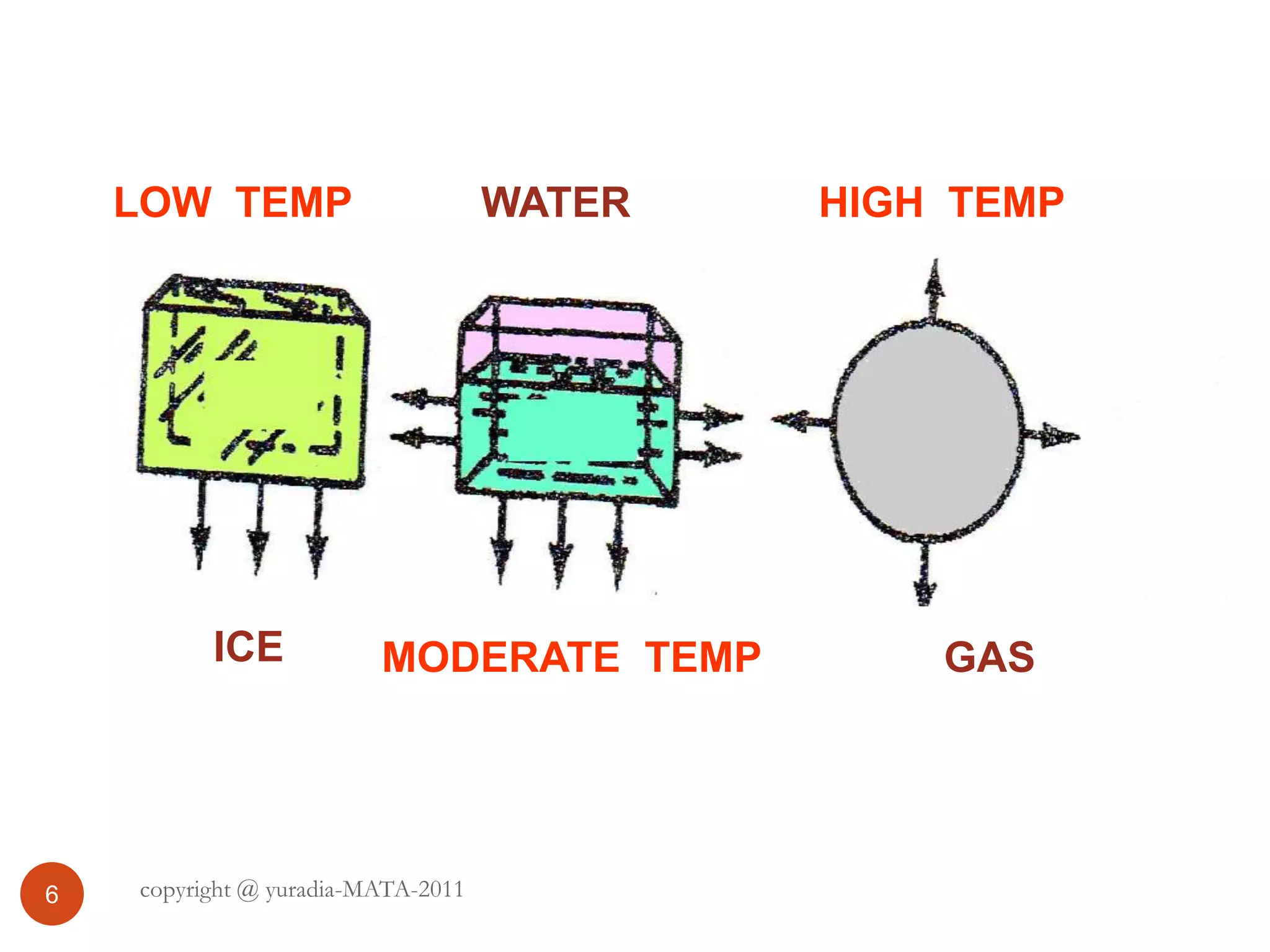
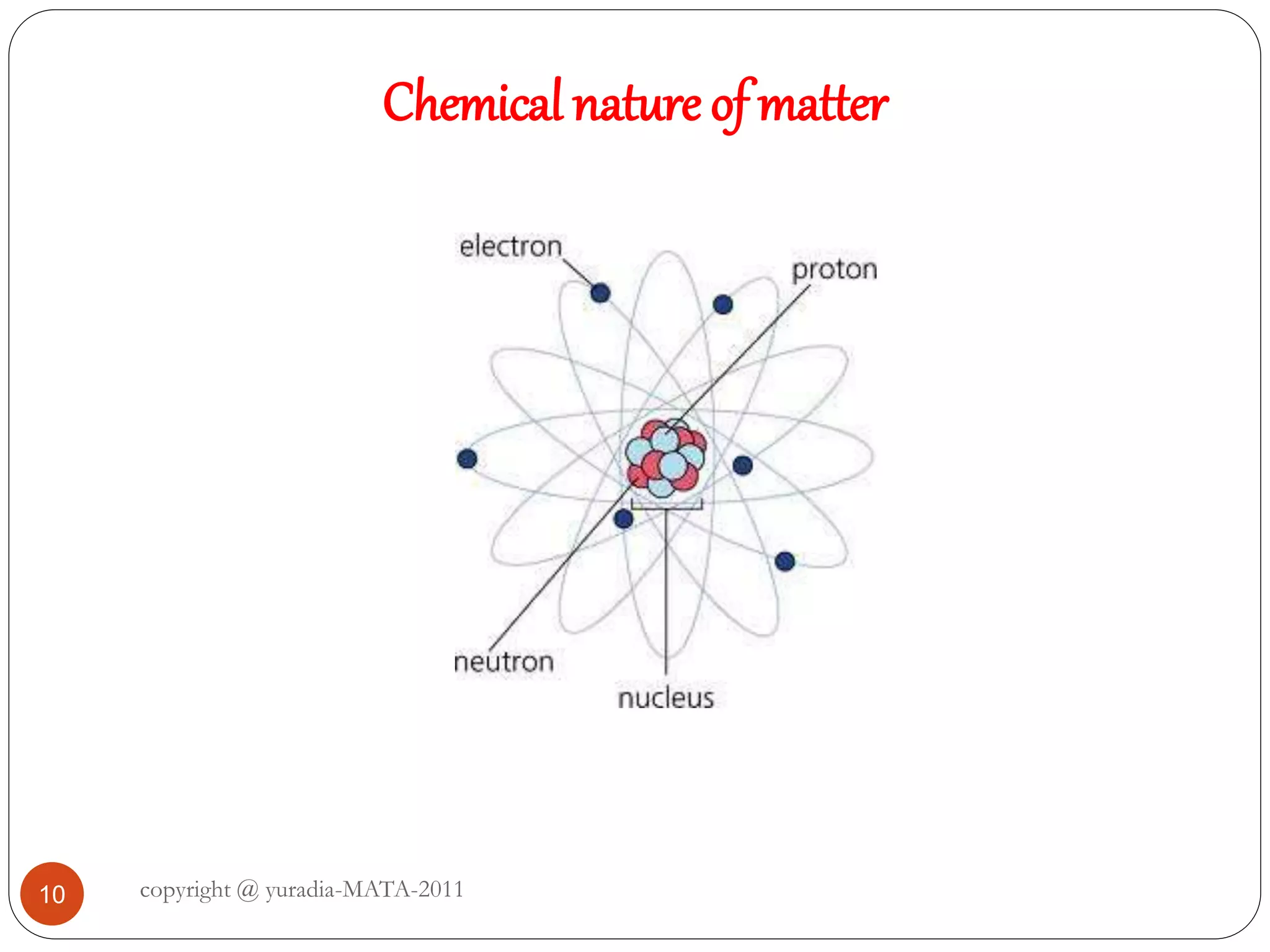
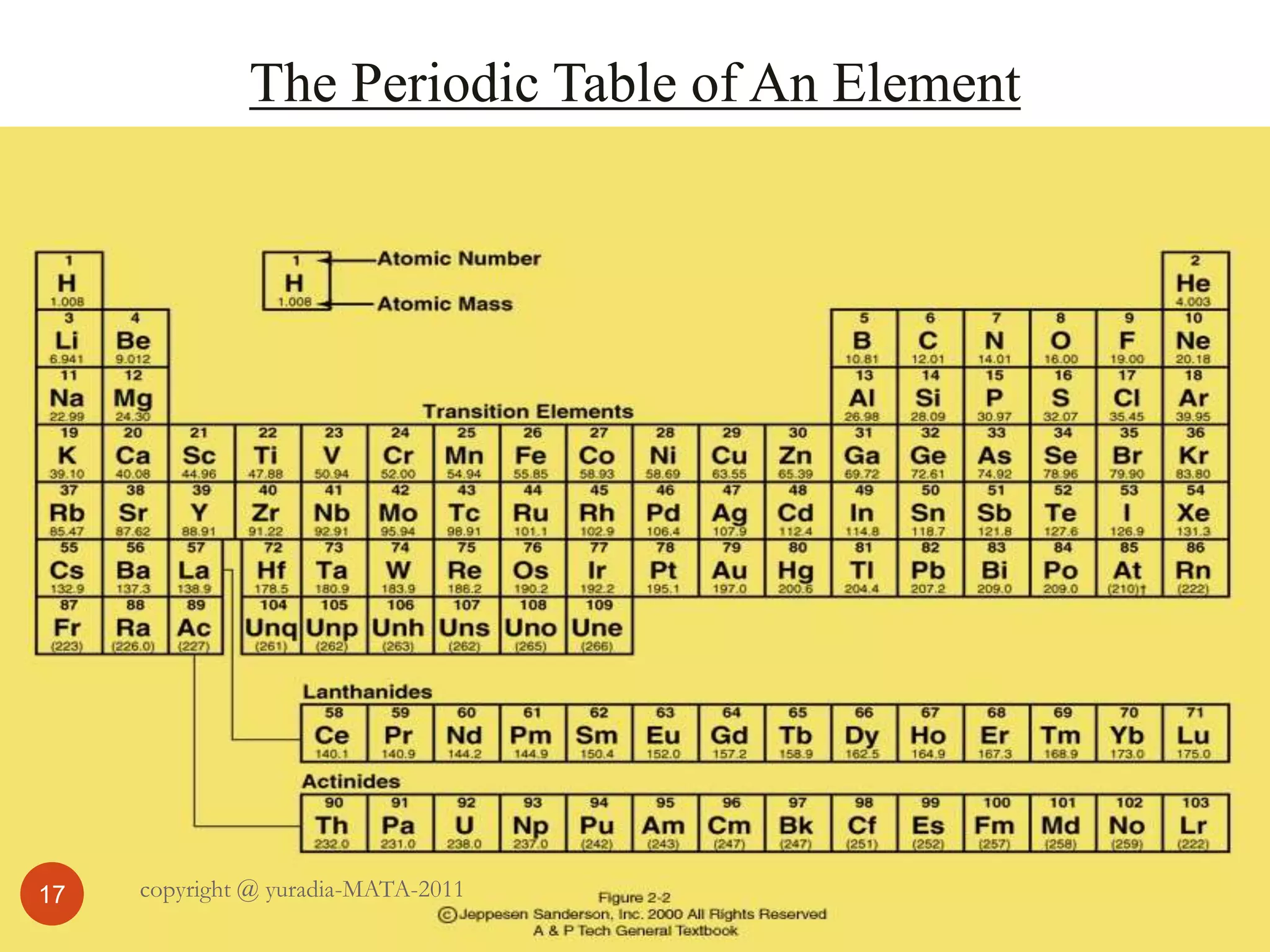
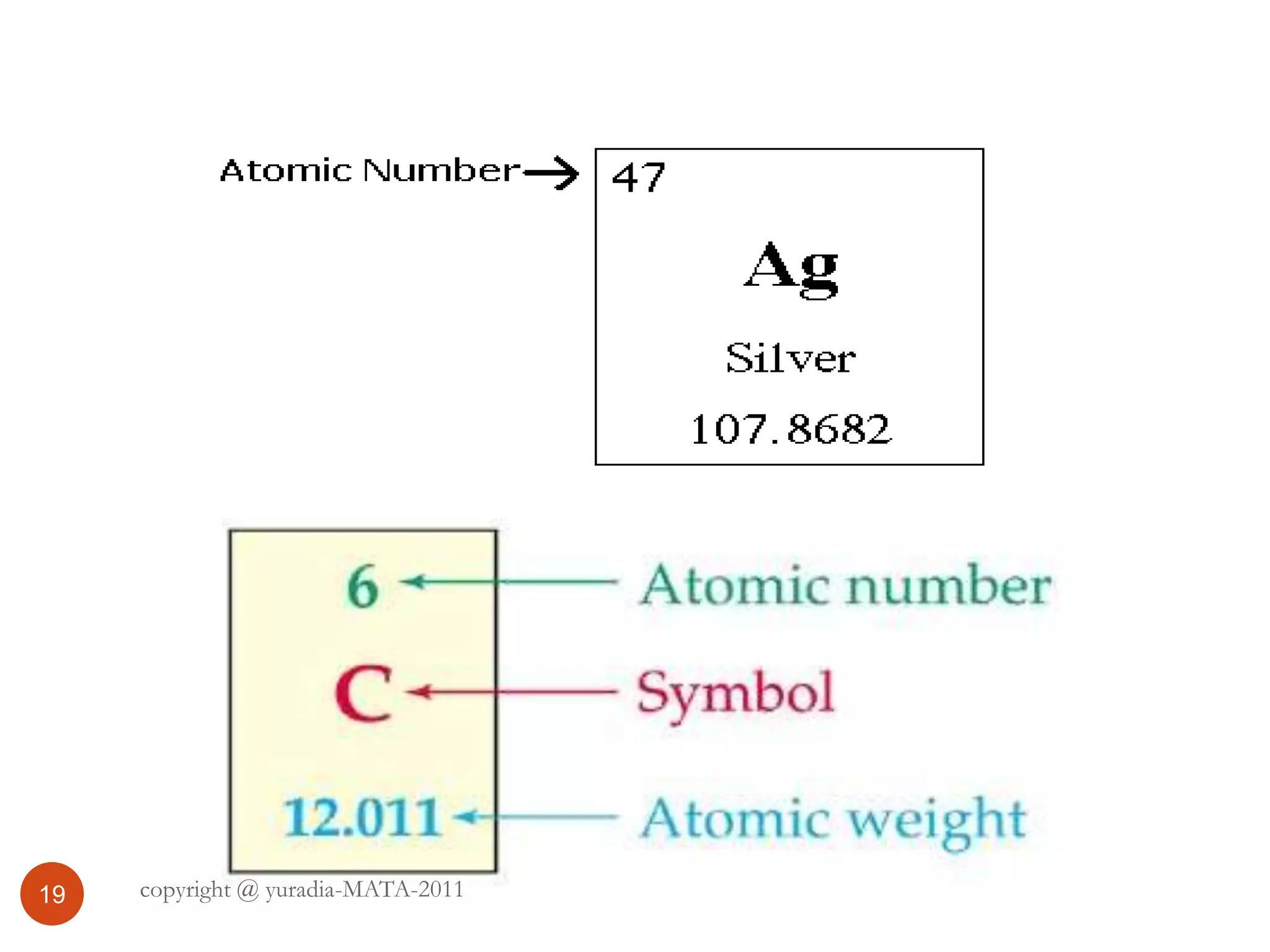
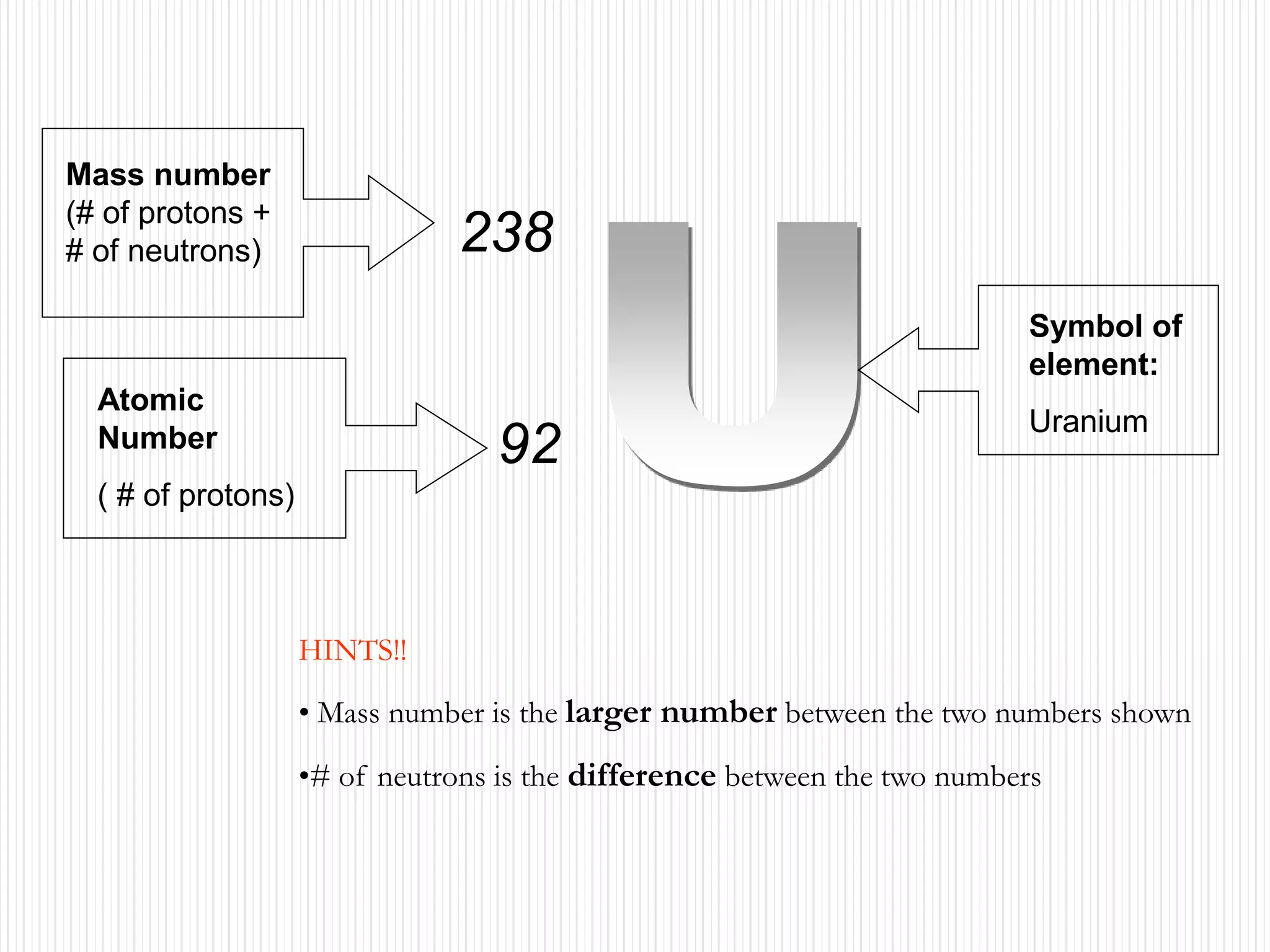
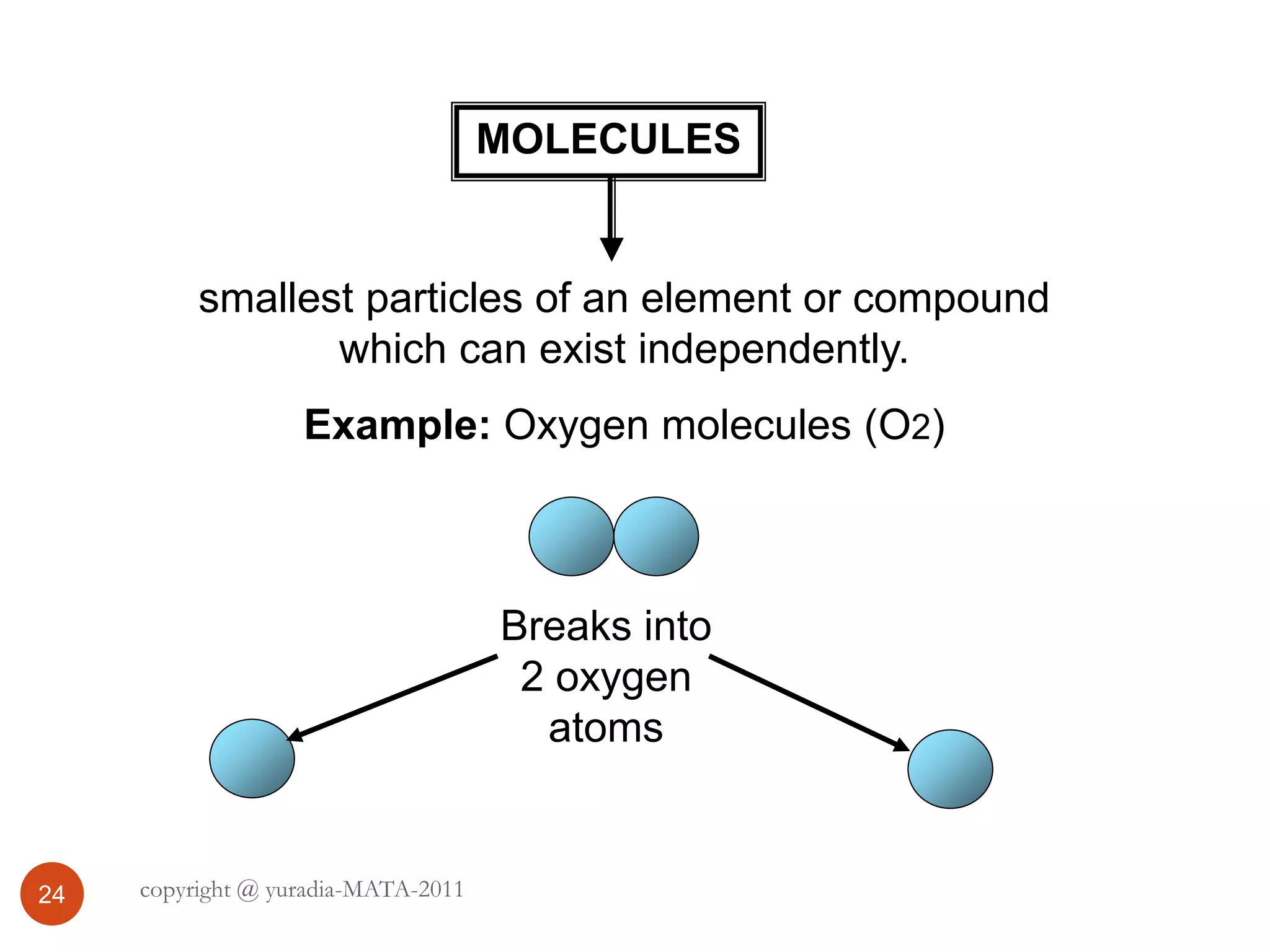
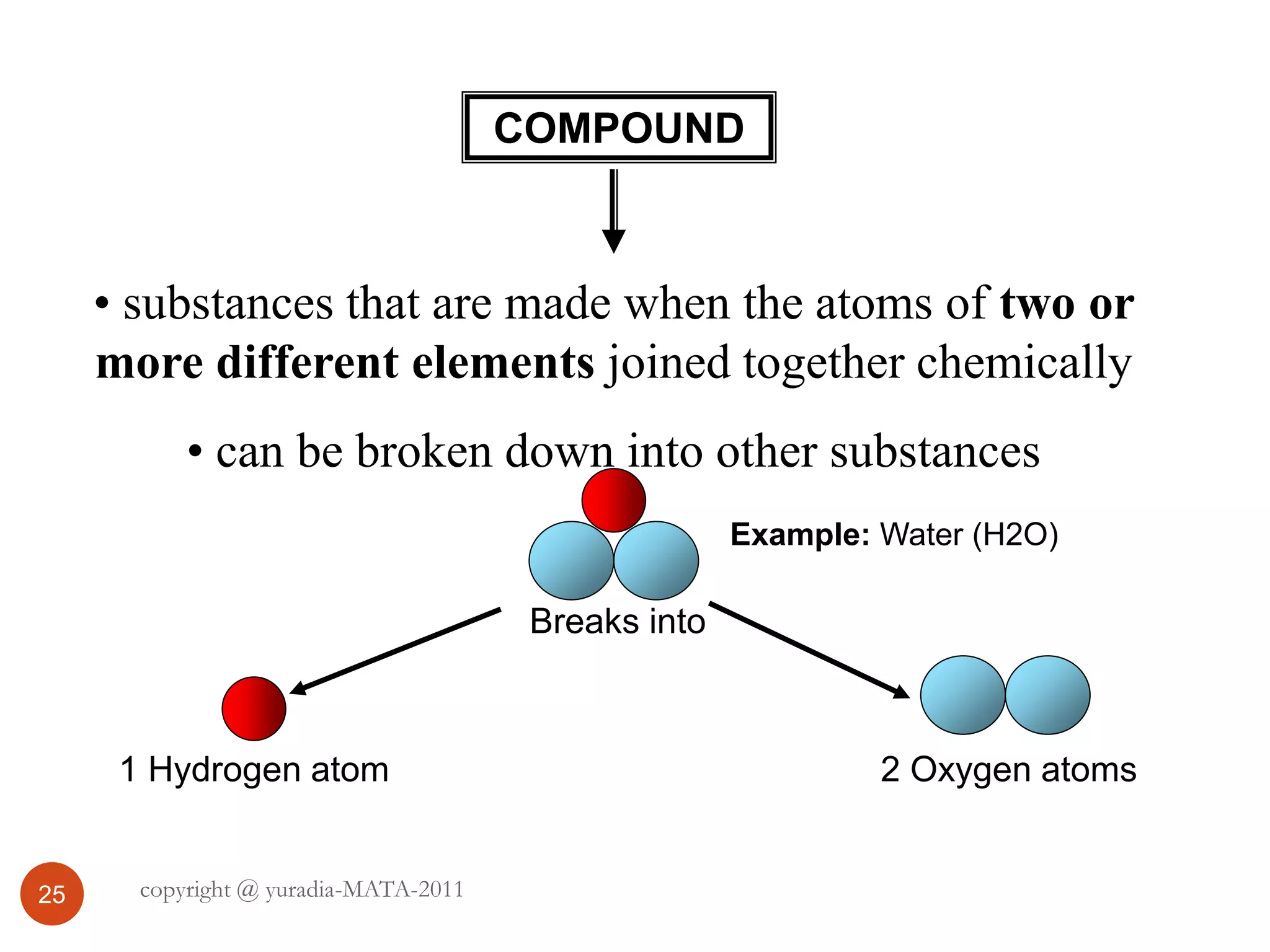
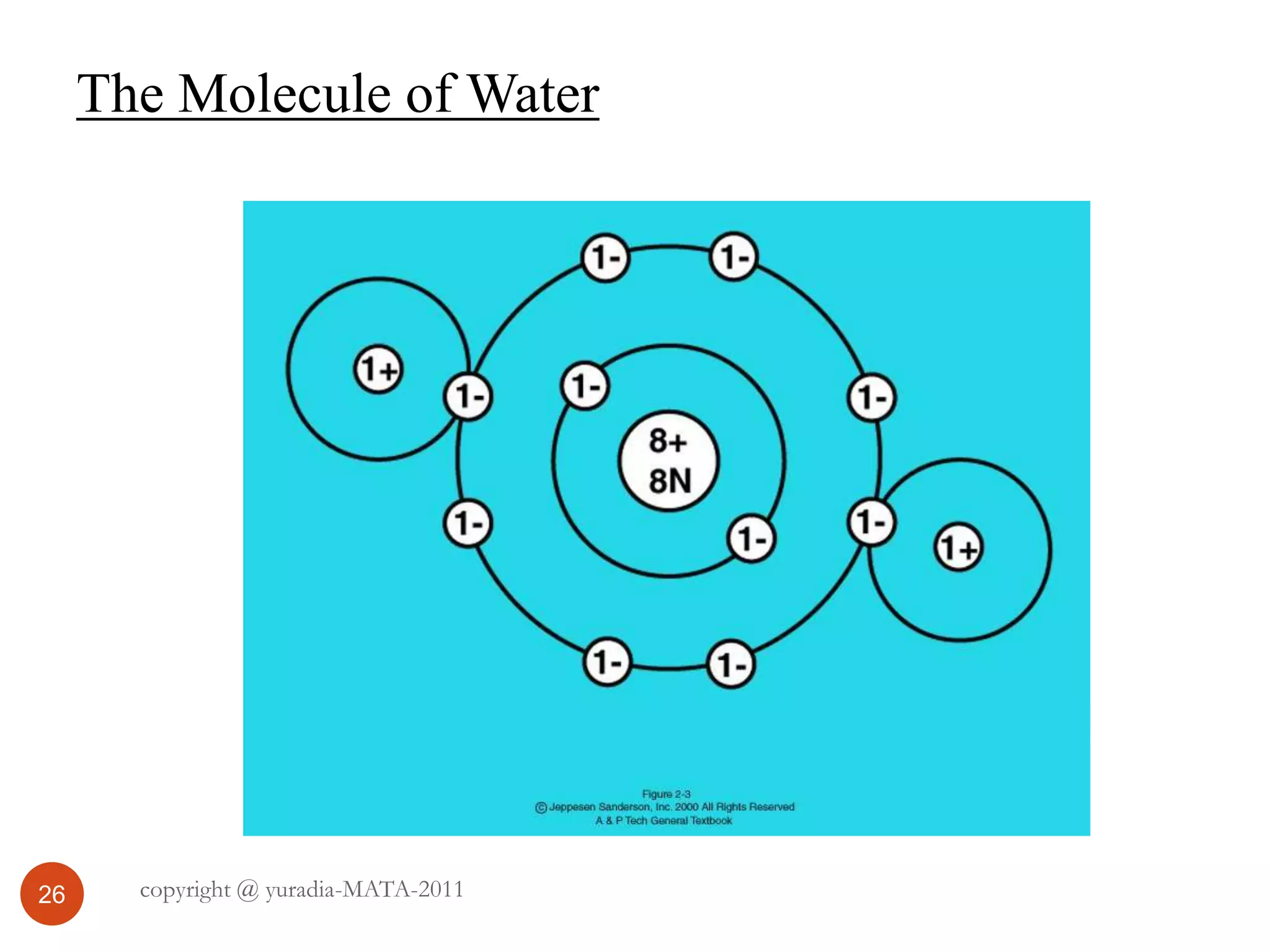
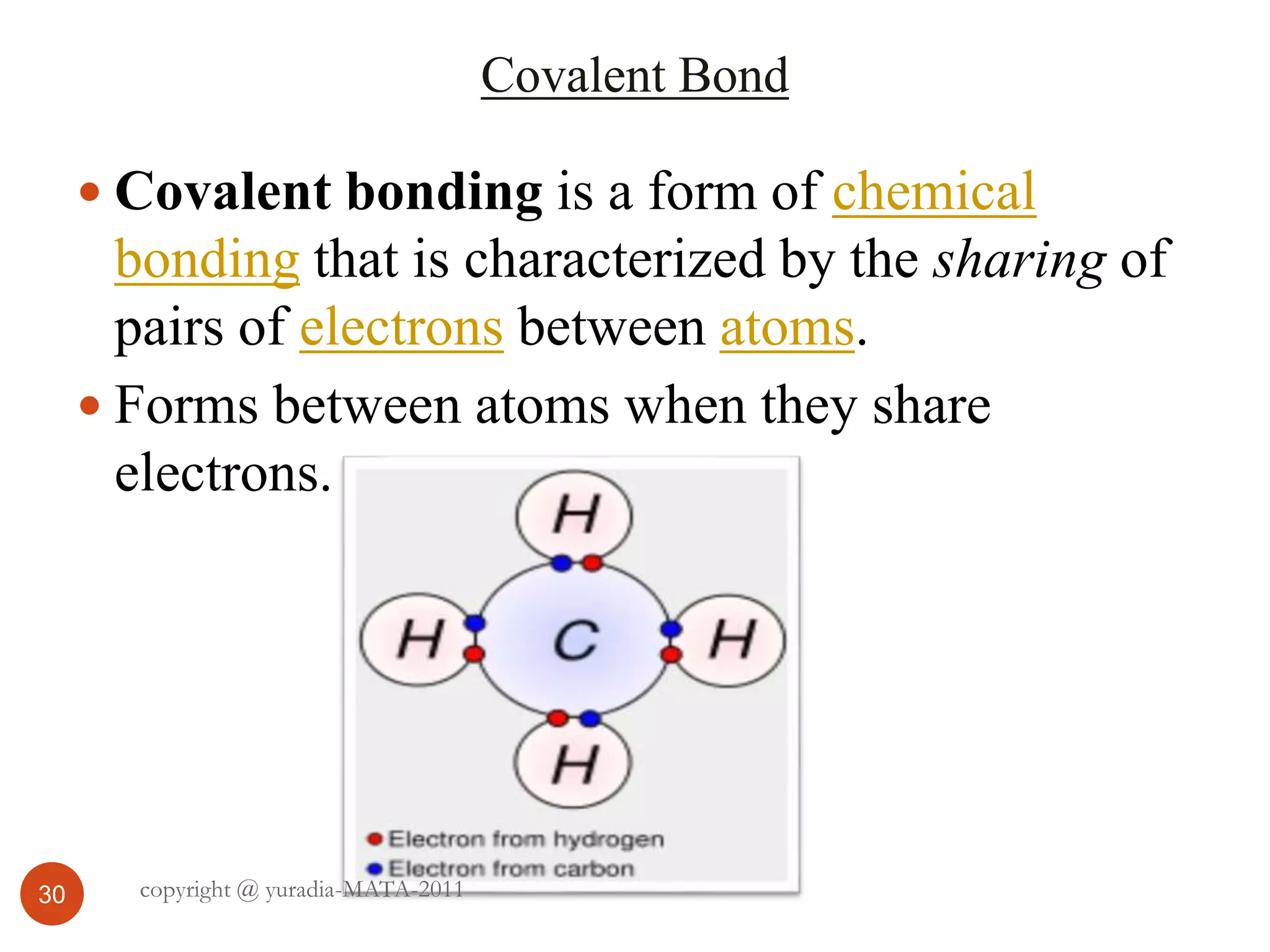
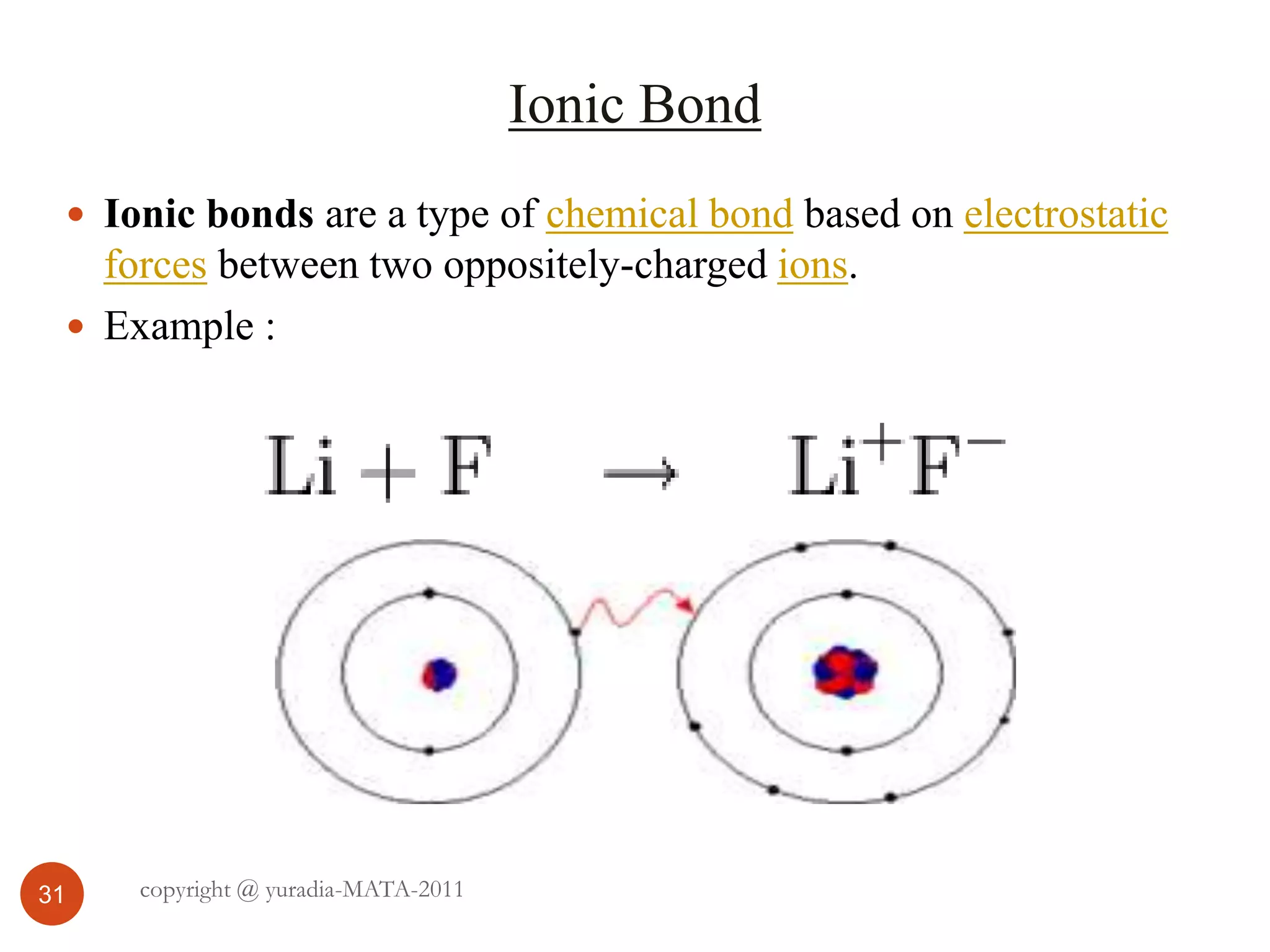
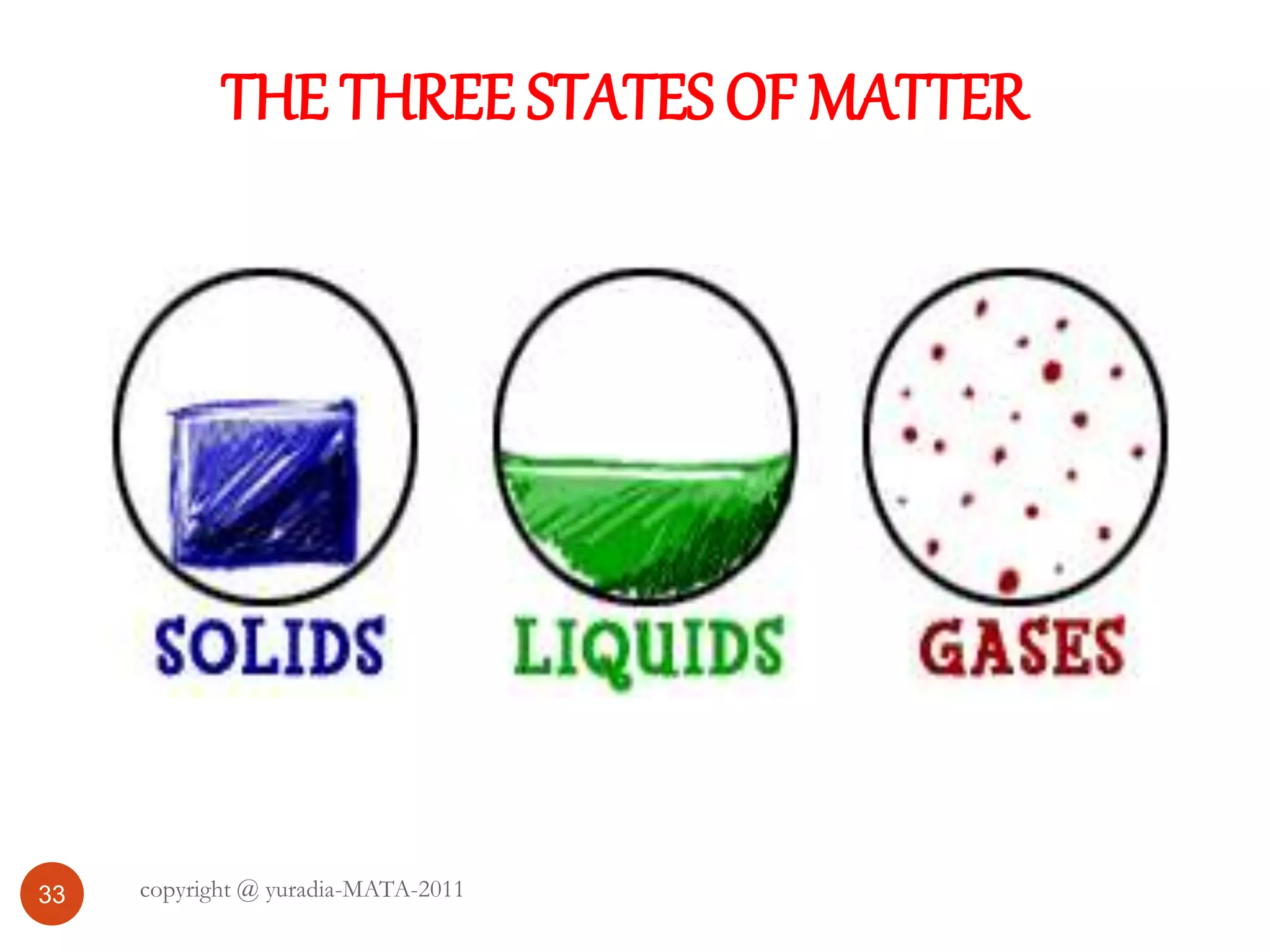
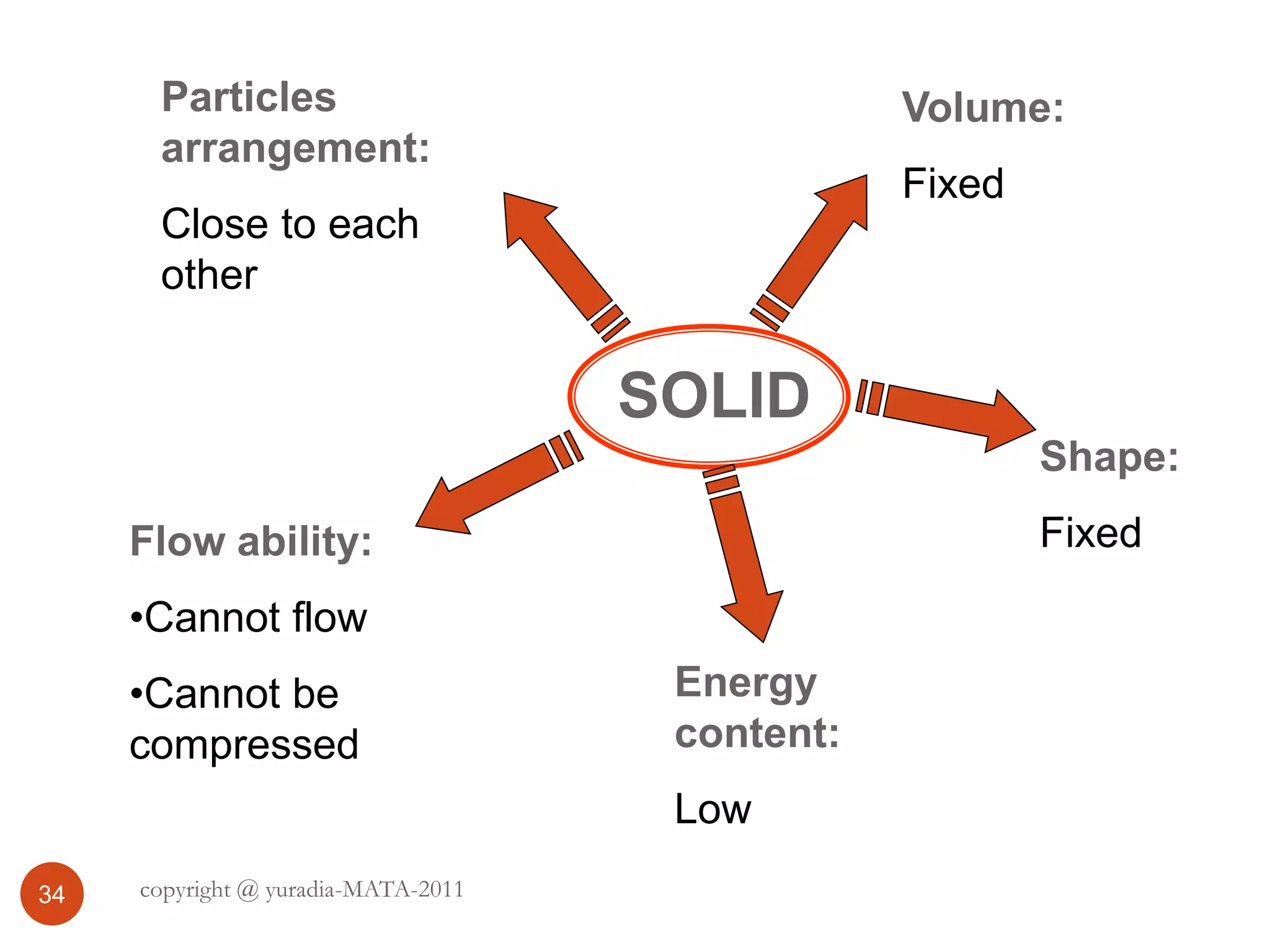
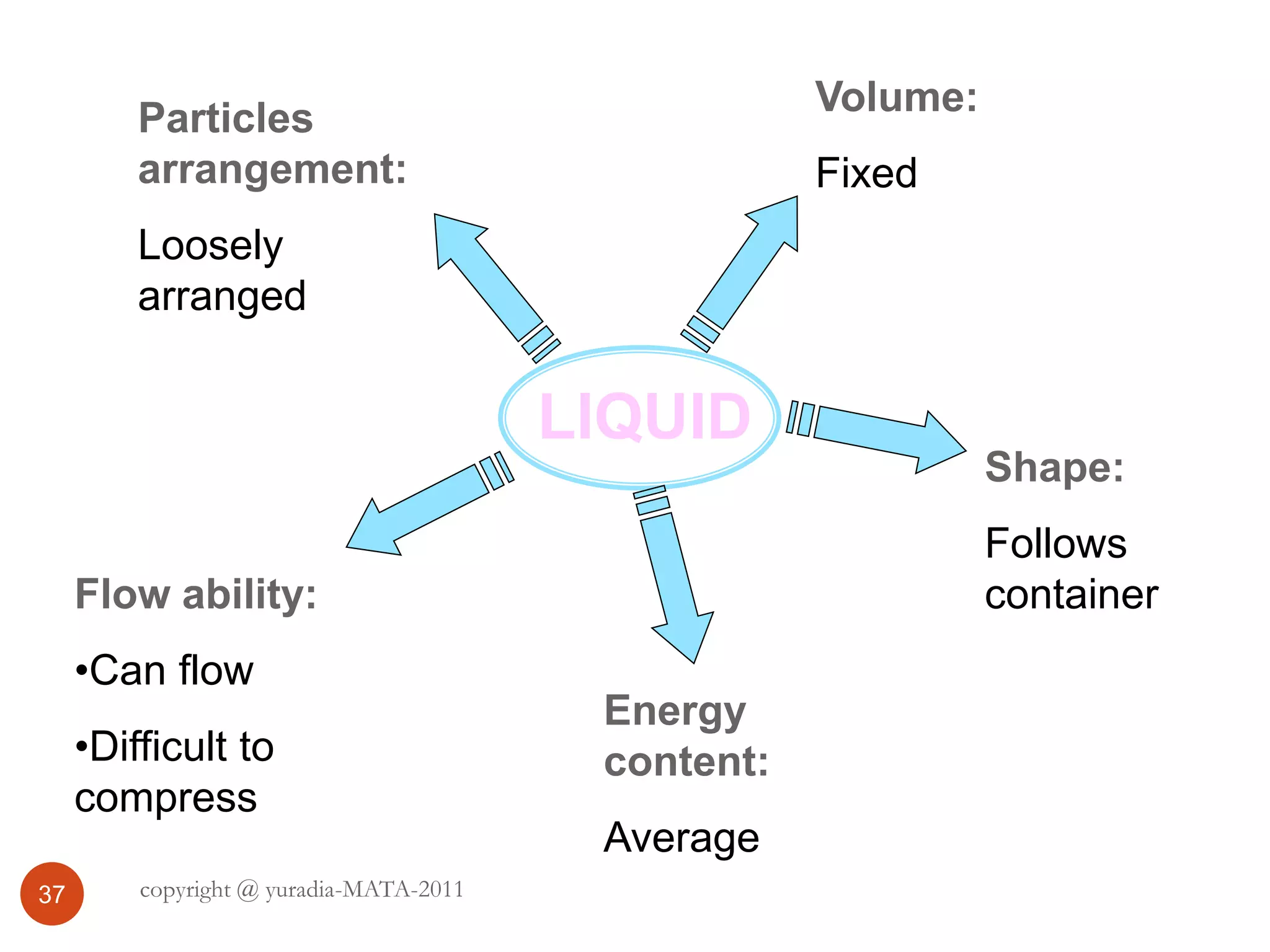
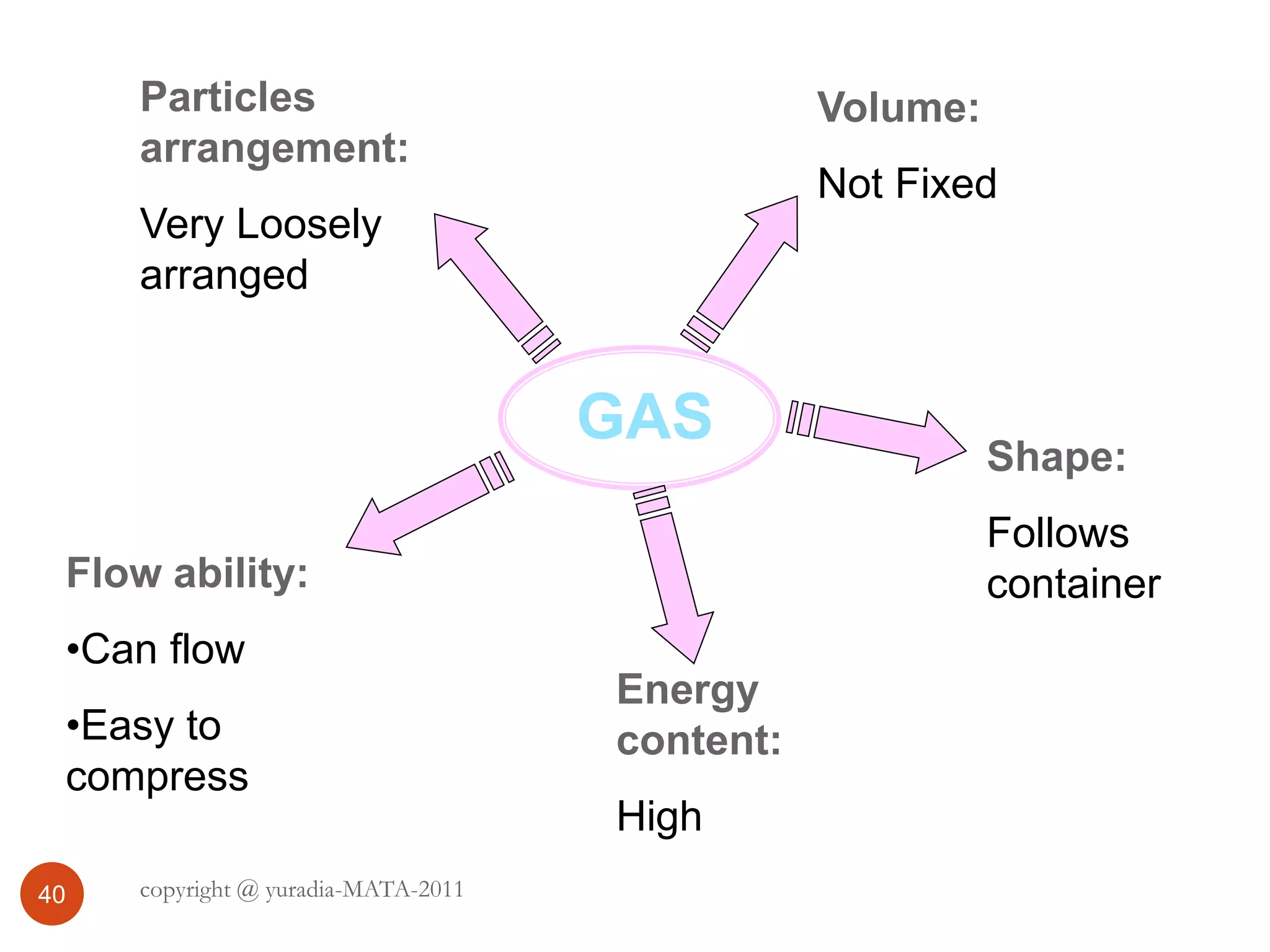
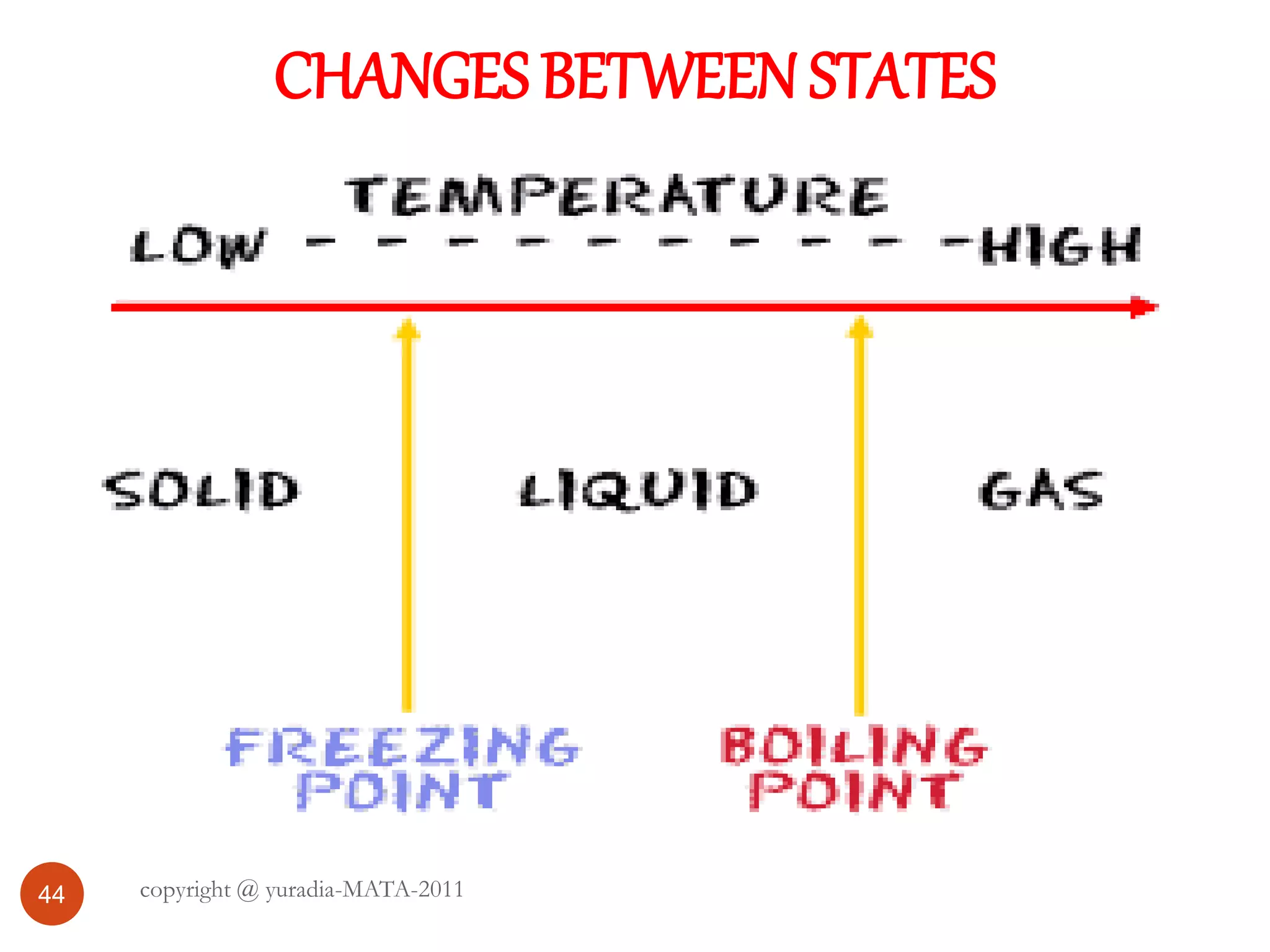
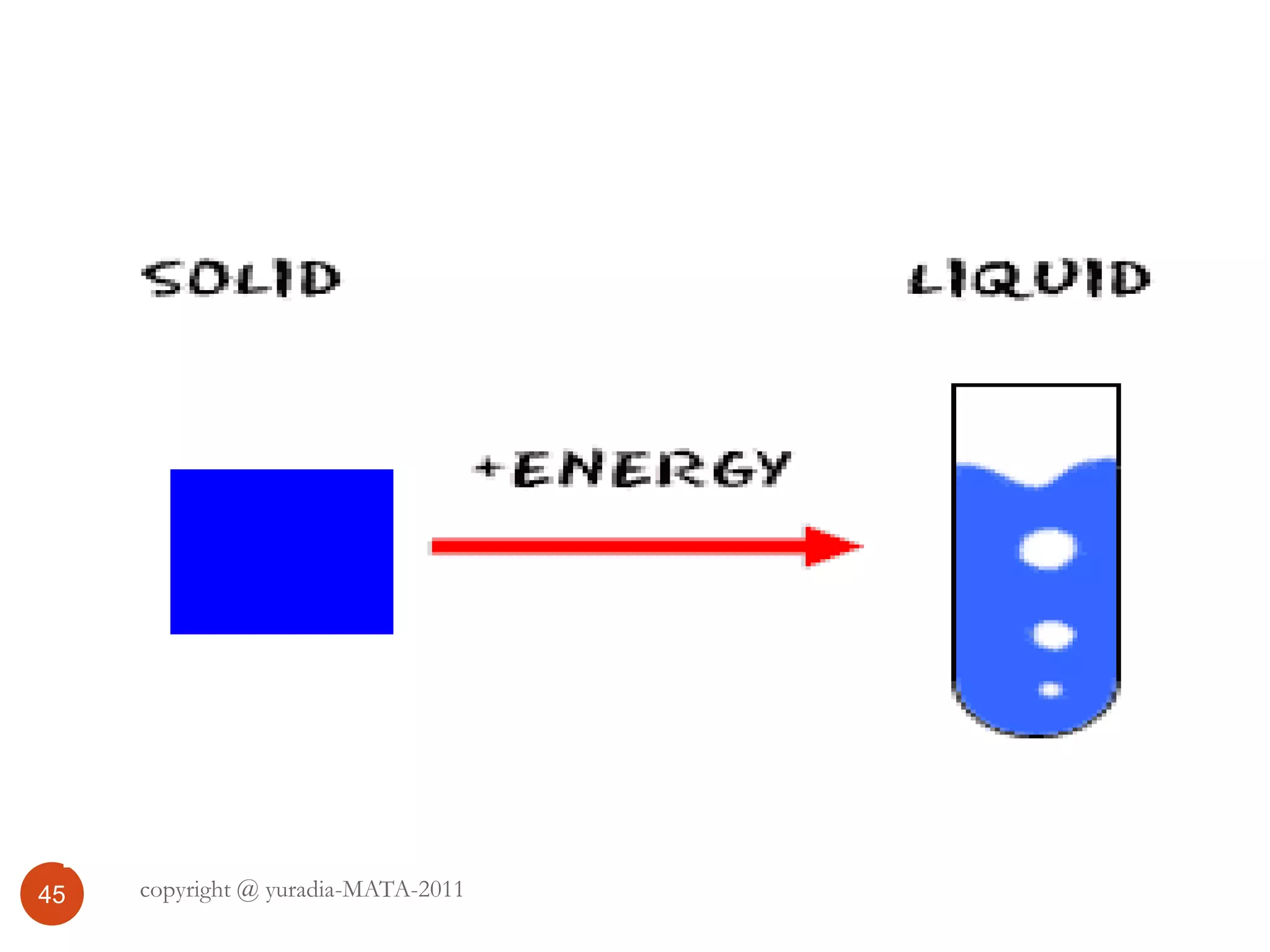
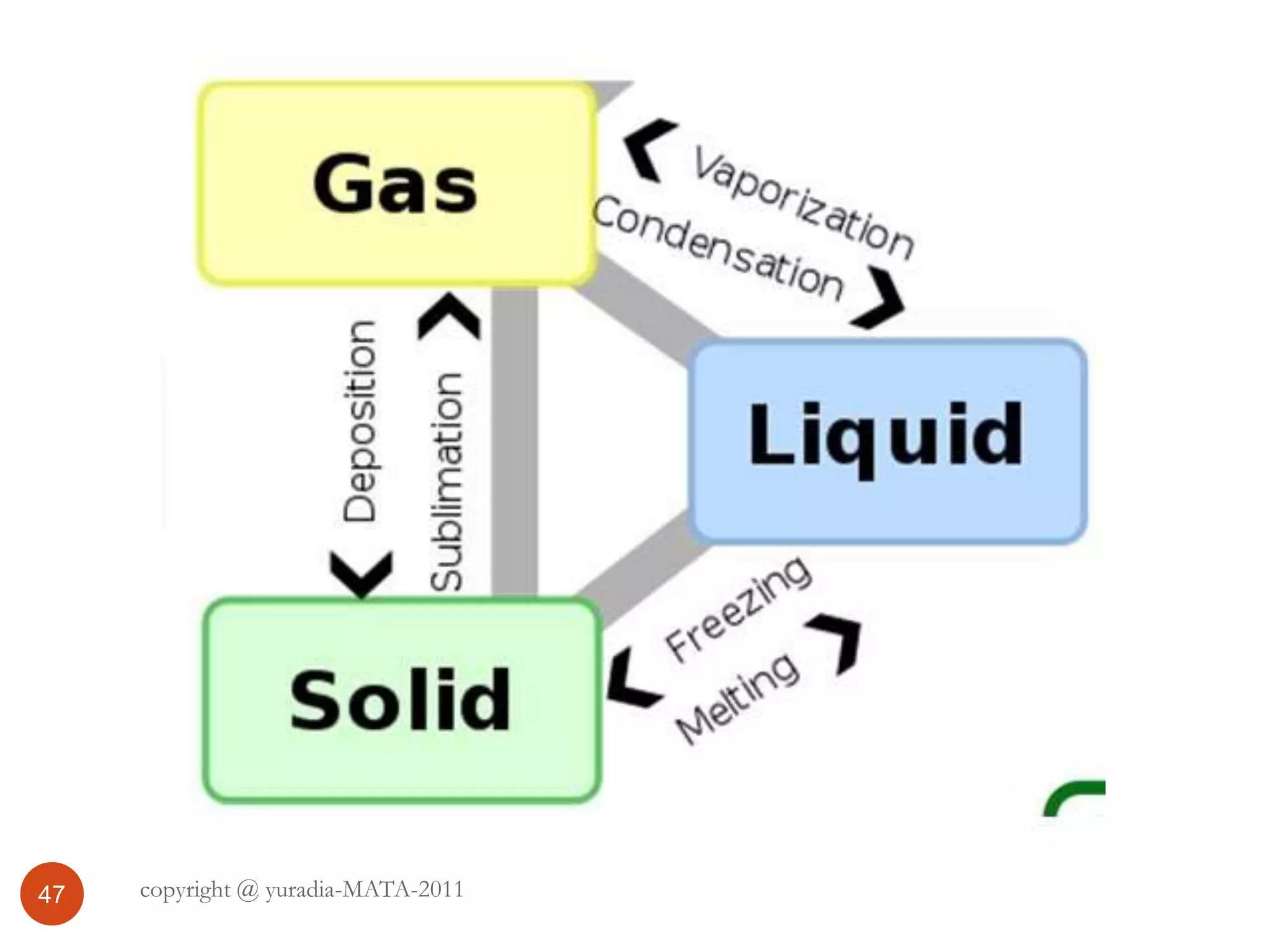
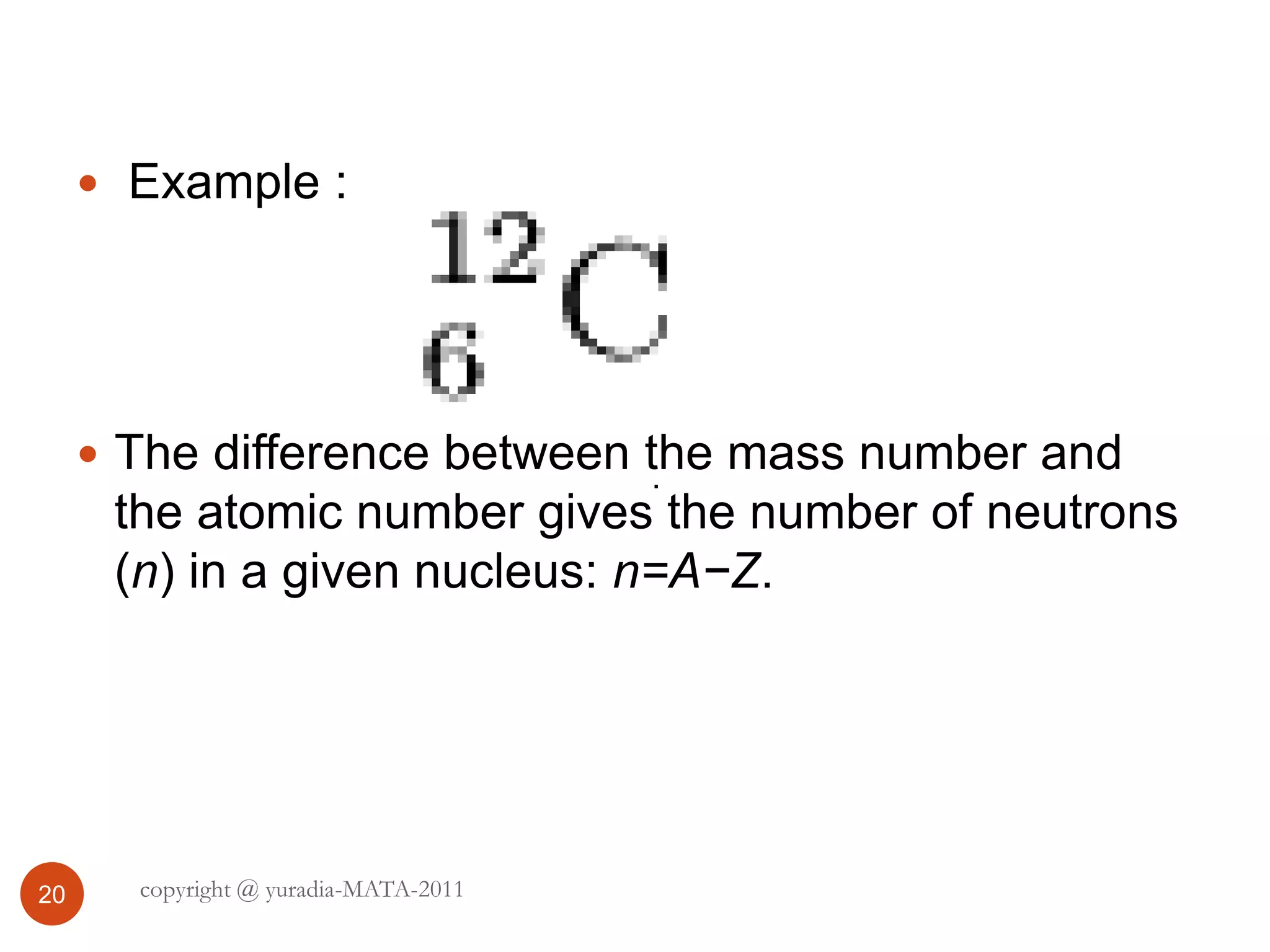The document discusses the three states of matter - solid, liquid, and gas. It provides details about the properties of each state, including density, ability to flow, compressibility, energy content, volume, shape, and particle arrangement. Solids have the highest density and fixed volume/shape due to close particle packing. Liquids are less dense than solids and can flow but maintain a fixed volume. Gases have the lowest density and fill their container due to widely-spaced particles. Changes between the states occur when energy is added or removed, causing changes to particle motion and interaction.