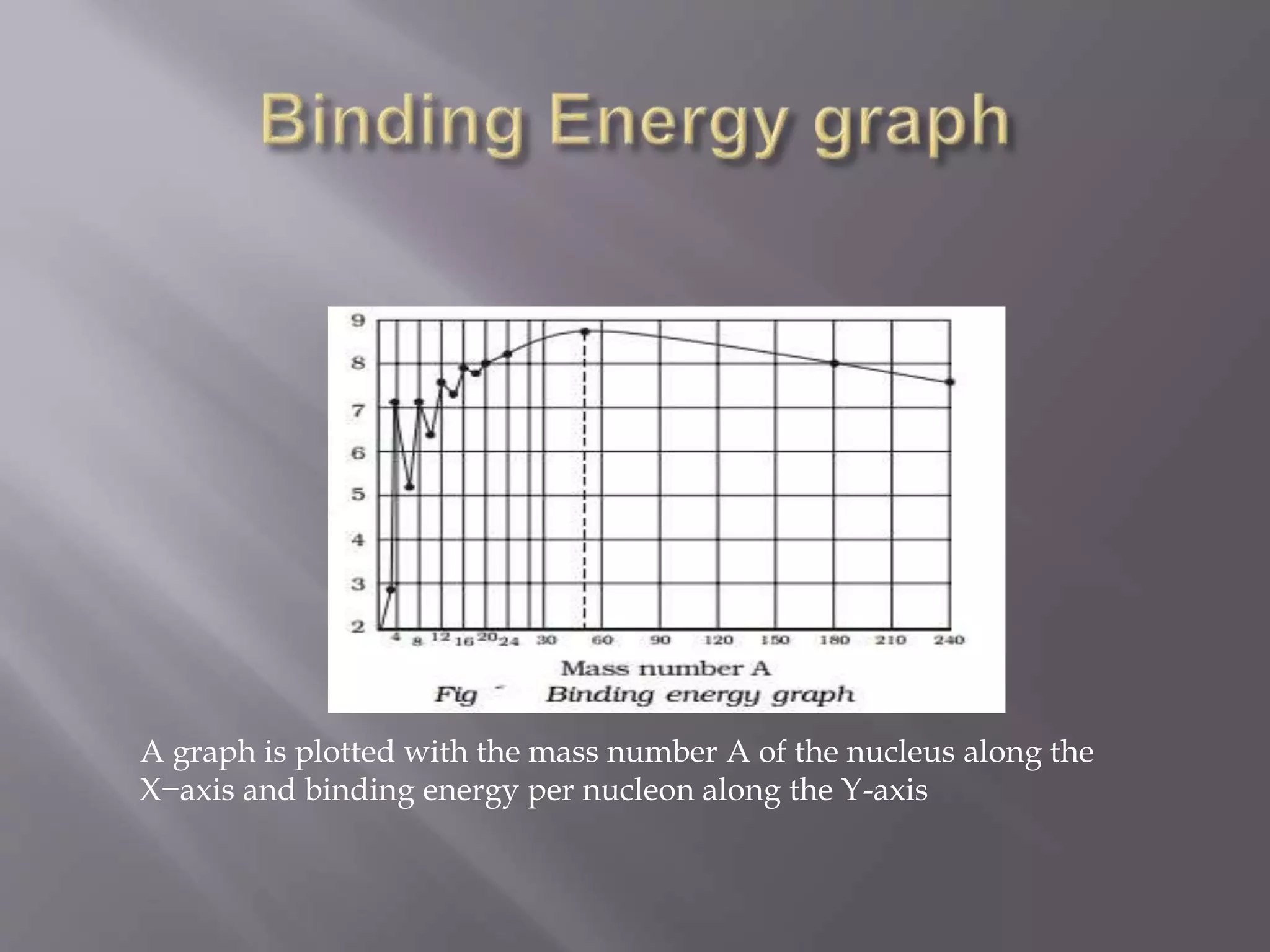
The document discusses the structure of the nucleus. It contains protons and neutrons, collectively known as nucleons. The nucleus is represented by the symbol ZXA, where Z is the number of protons, A is the total number of nucleons, and X is the element's symbol. Isotopes have the same Z but different A, while isobars have the same A but different Z. The nucleus has a small but dense volume due to its high nuclear density. Nuclear reactions and stability are influenced by the binding energy between nucleons. Binding energy reaches a maximum for iron nuclei, making it the most stable.









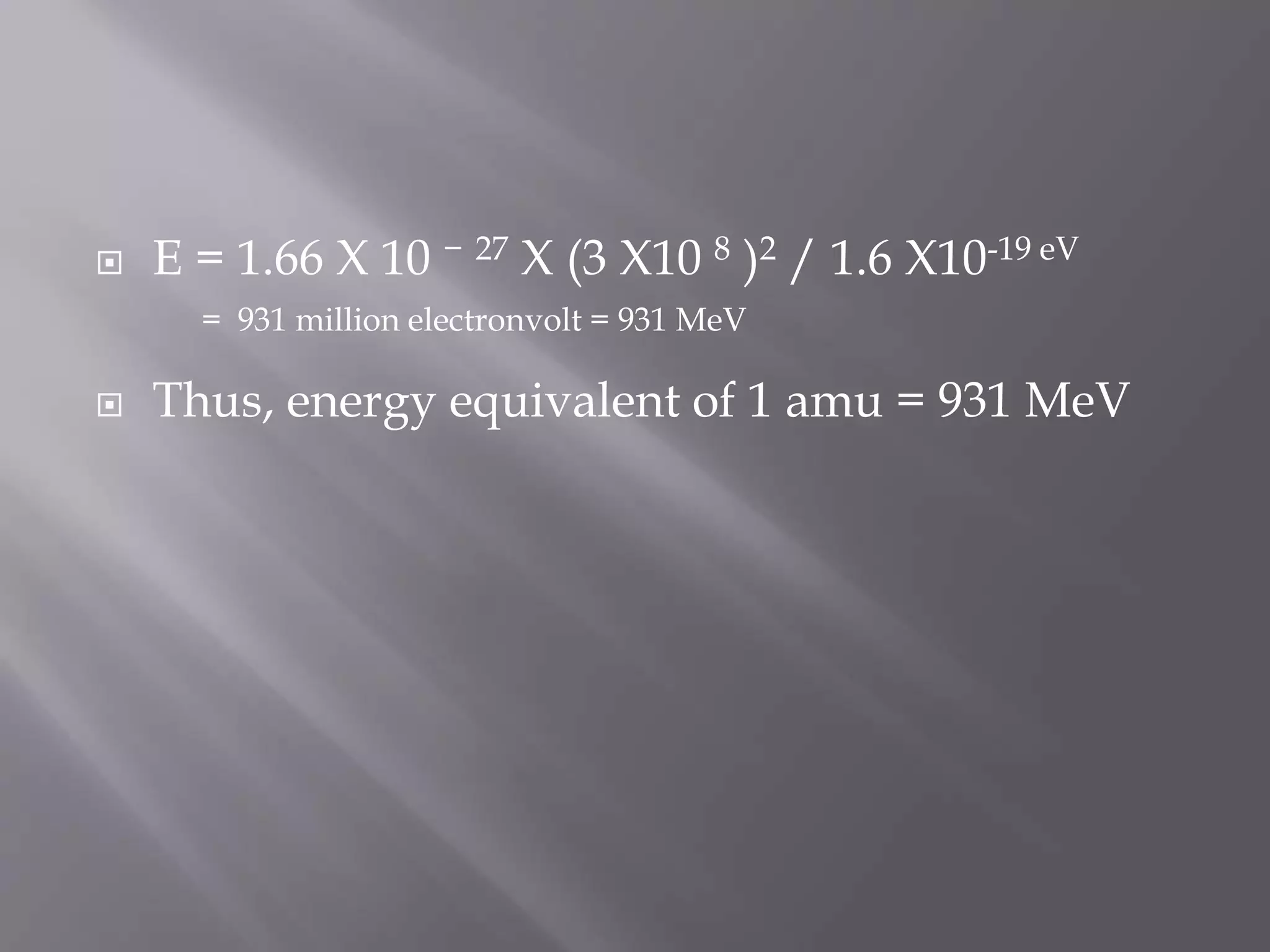

![ When the protons and neutrons combine to form a
nucleus, the mass that disappears (mass defect, ∆m) is
converted into an equivalent amount of energy (∆mc2).
This energy is called the binding energy of the nucleus.
Binding energy = [ZmP + Nmn - m] c2
= ∆m c2
It determines its stability against disintegration.
If the binding energy is large, the nucleus is stable .
The binding energy per nucleon varies from element to
element.](https://image.slidesharecdn.com/classificationofnucleiandpropertiesofnucleus-191105064633/75/Classification-of-nuclei-and-properties-of-nucleus-12-2048.jpg)