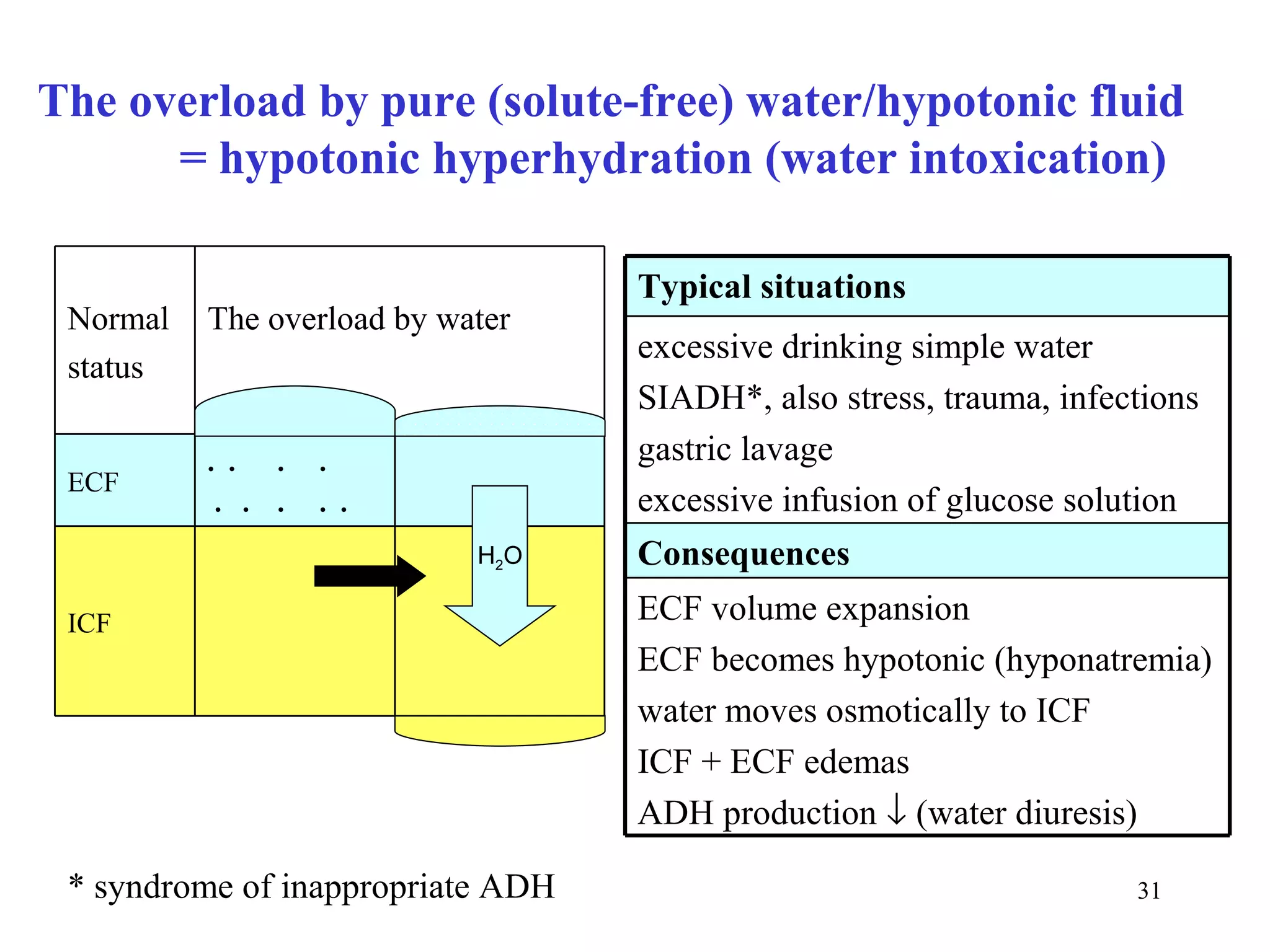
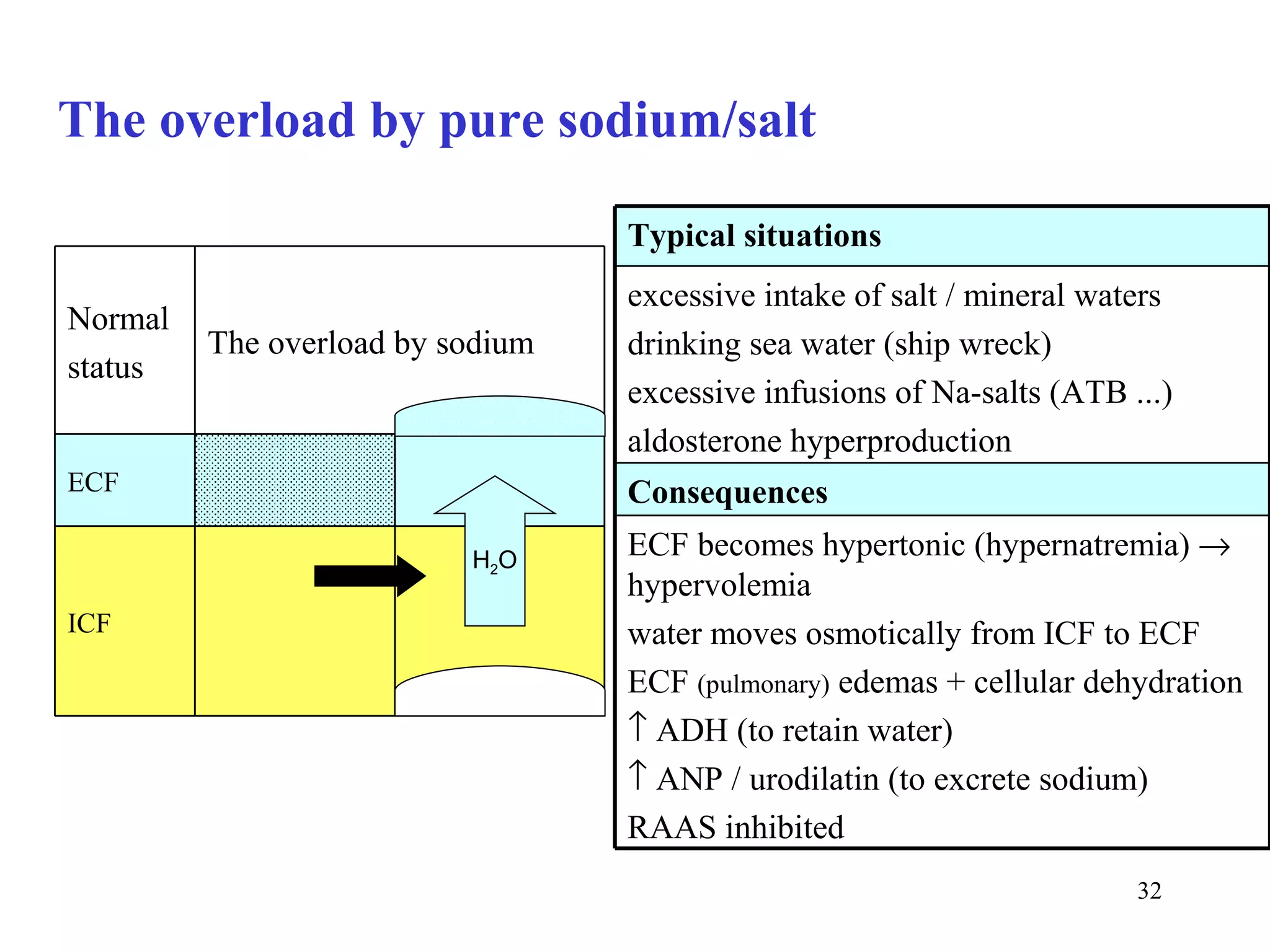
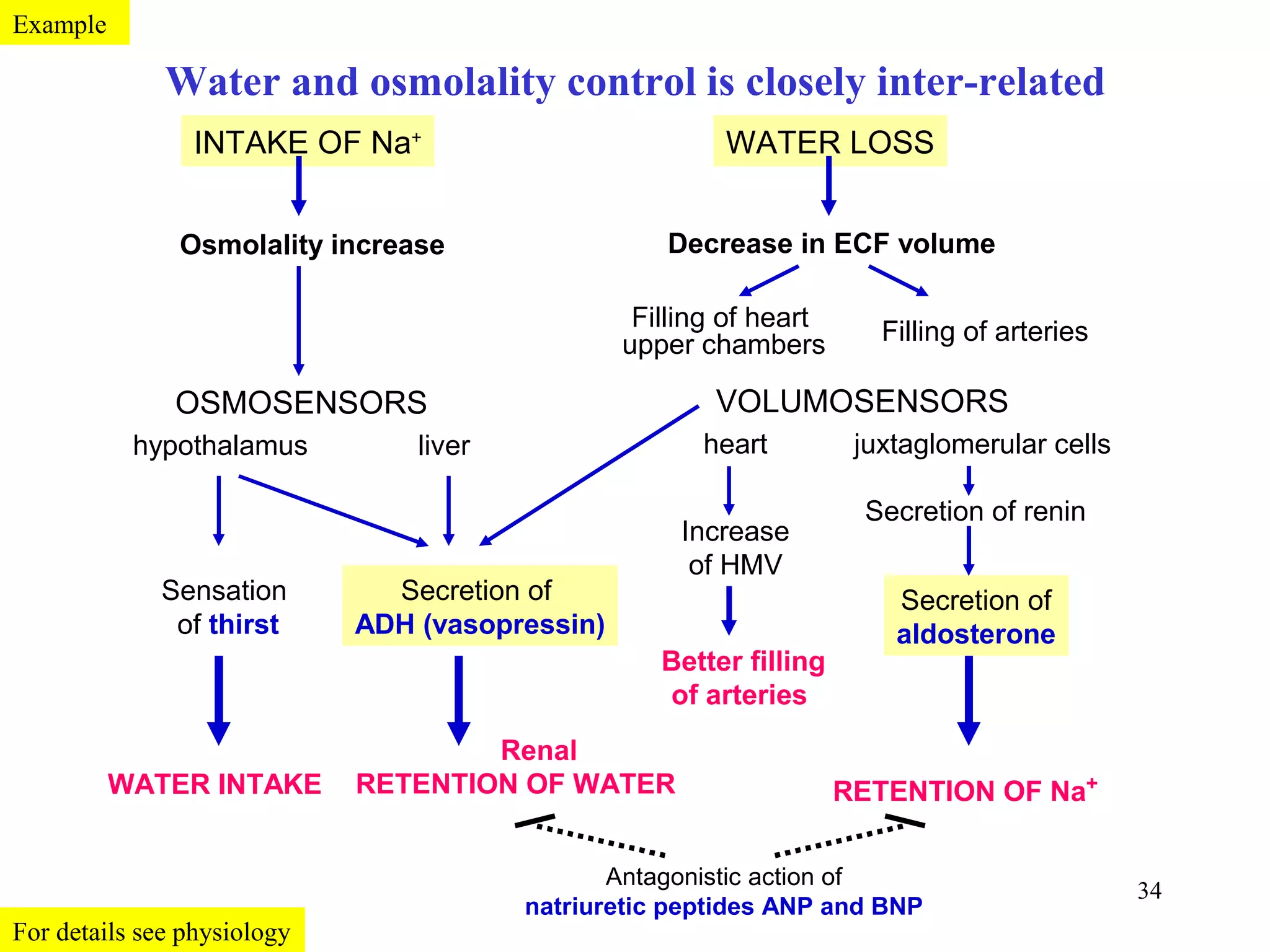
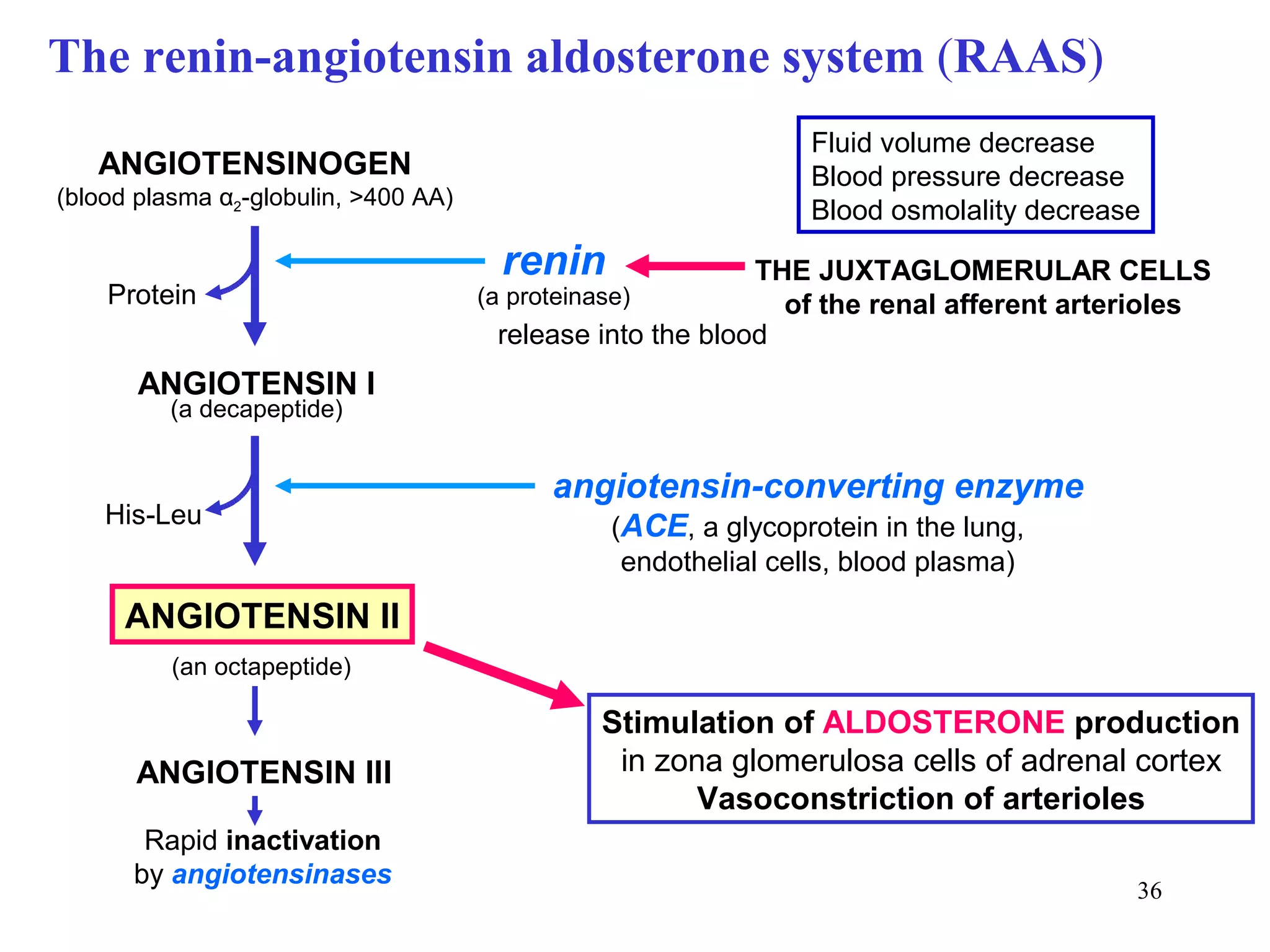
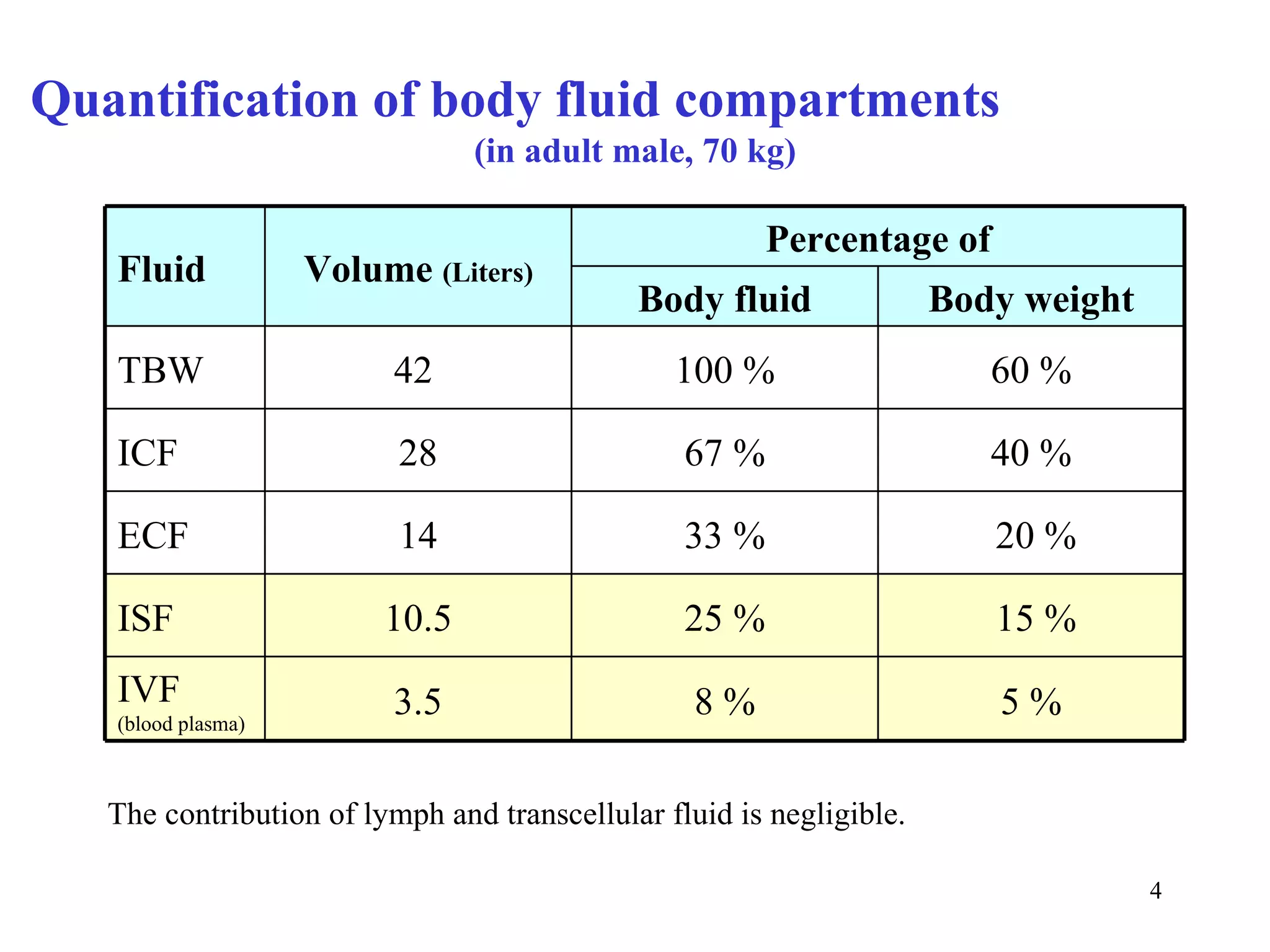
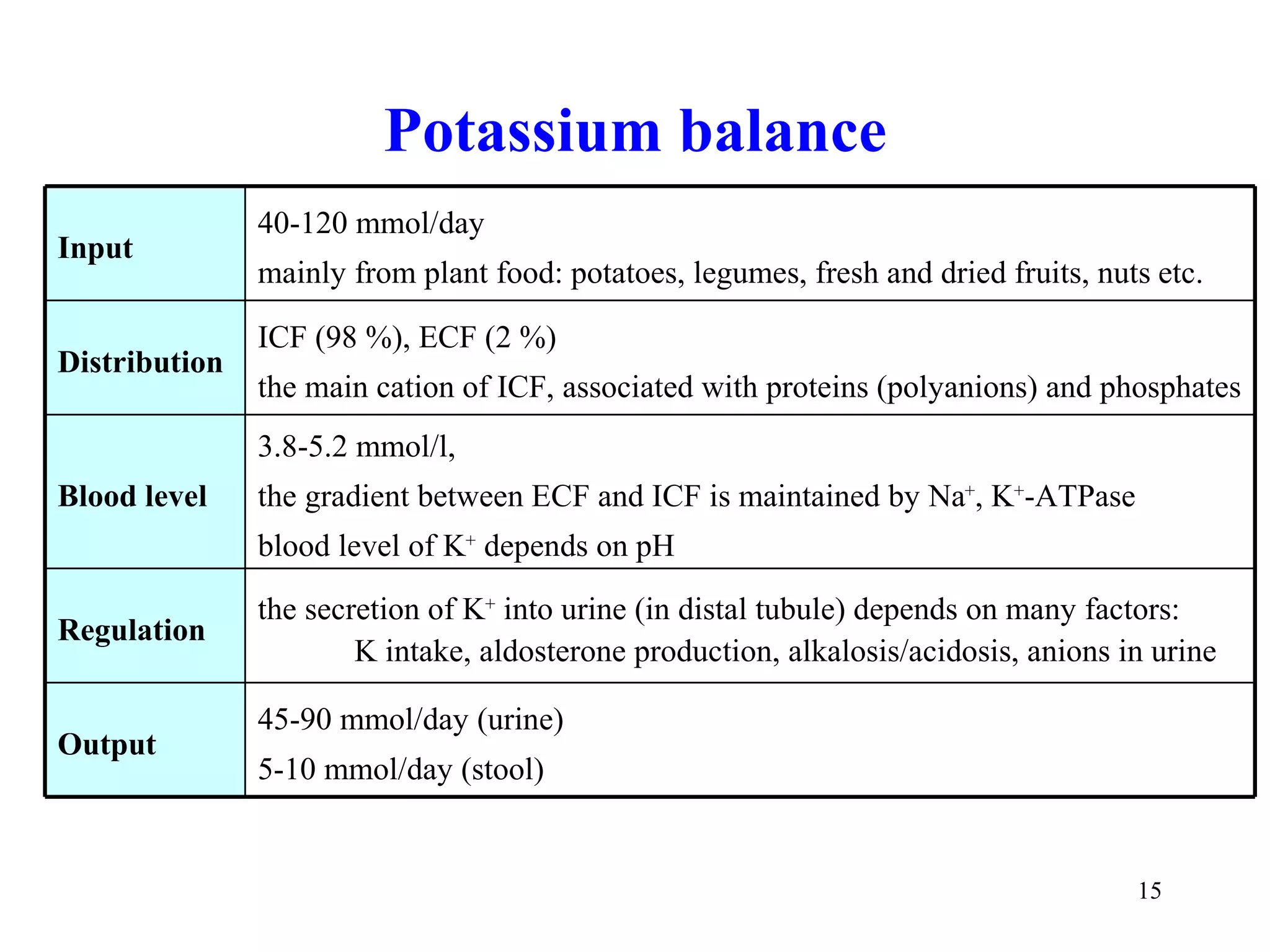
1) The document discusses body water content and fluid balance. It notes that total body water percentage decreases with age from about 75% in newborns to less than 50% in elders.
2) The body's water is divided into intracellular fluid and extracellular fluid compartments. Intracellular fluid makes up about two-thirds of total body water in adults.
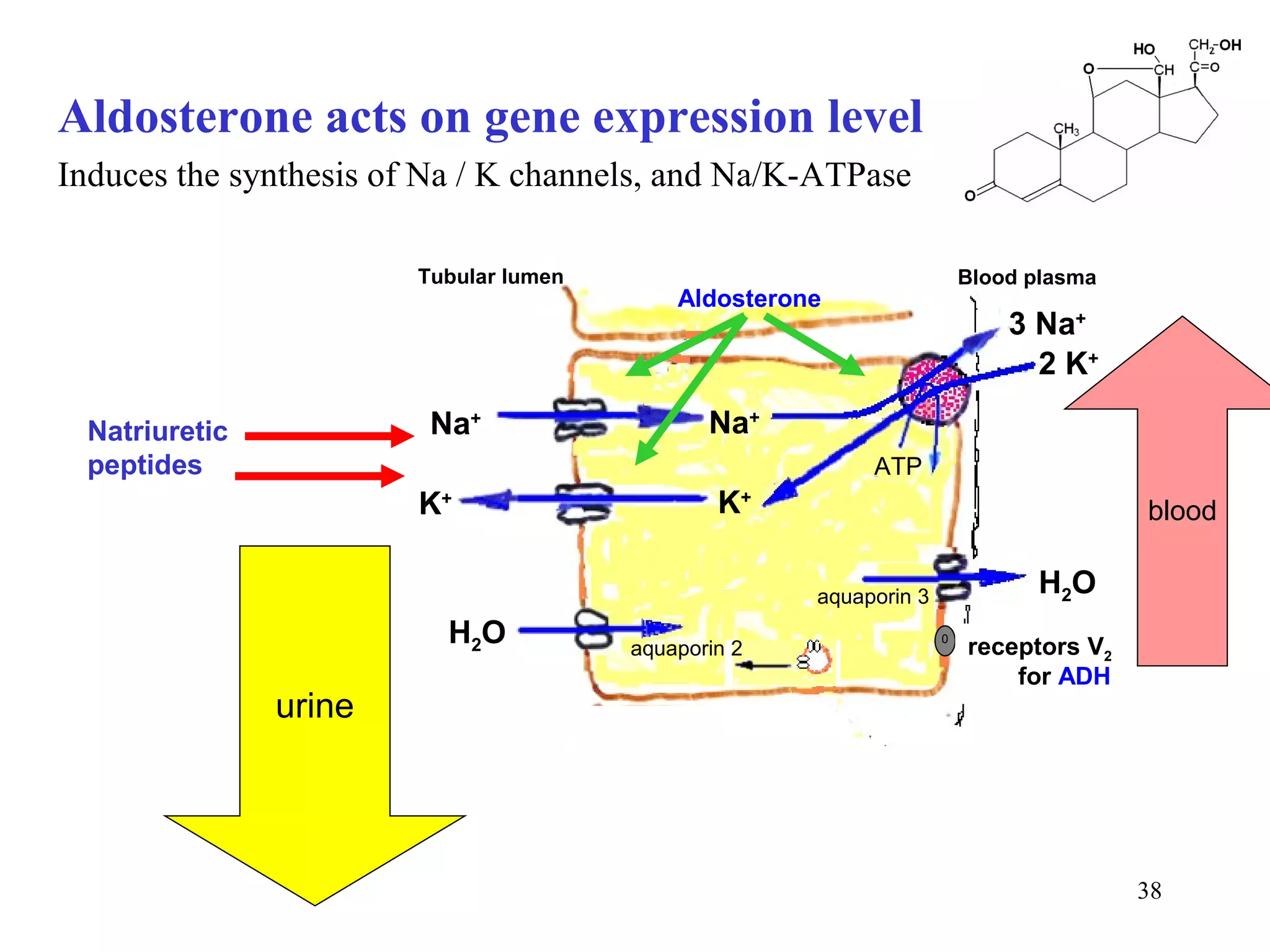
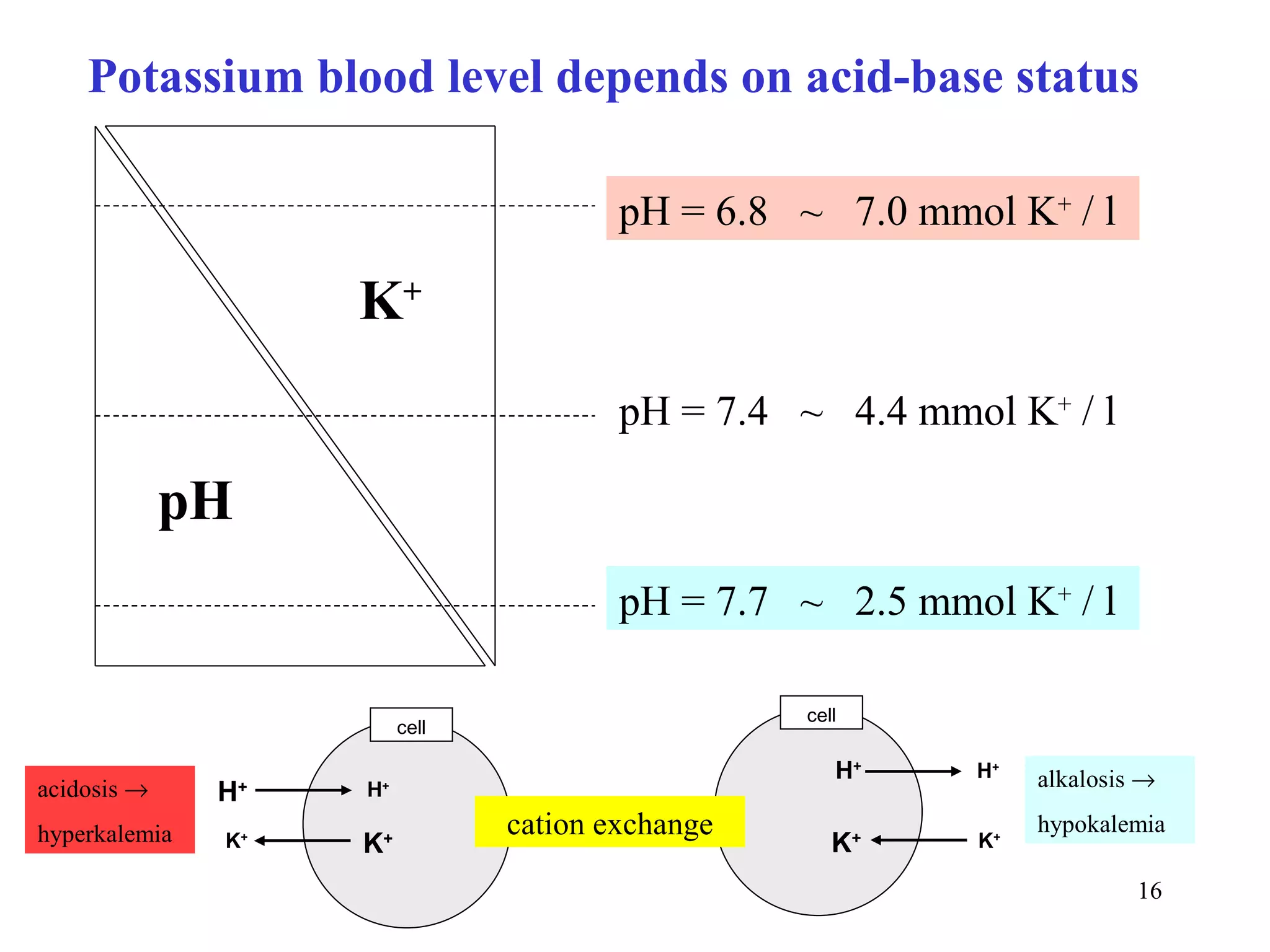
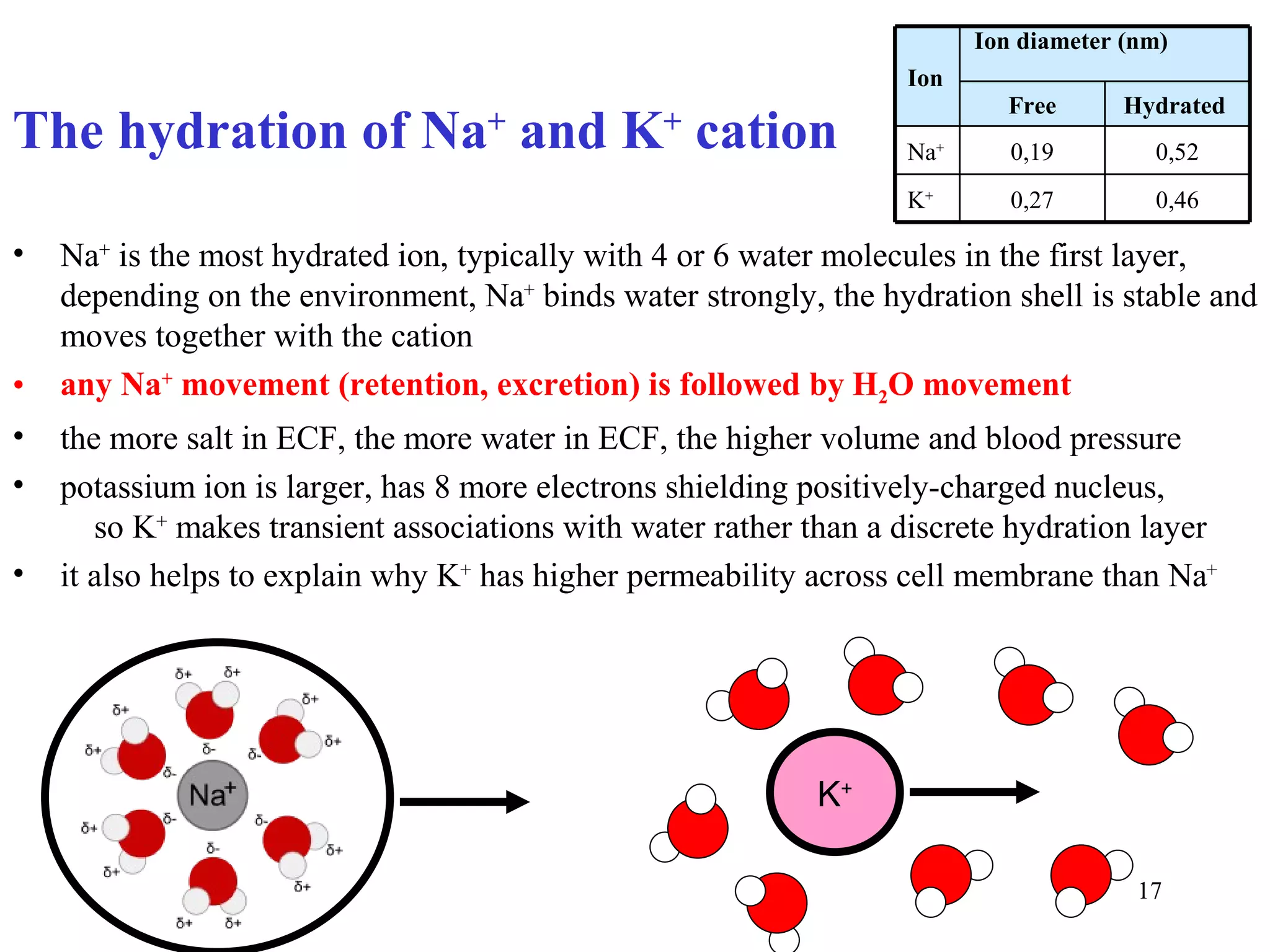
3) Sodium and potassium levels are tightly regulated between intracellular and extracellular fluids through the actions of hormones like aldosterone and atrial natriuretic peptide.






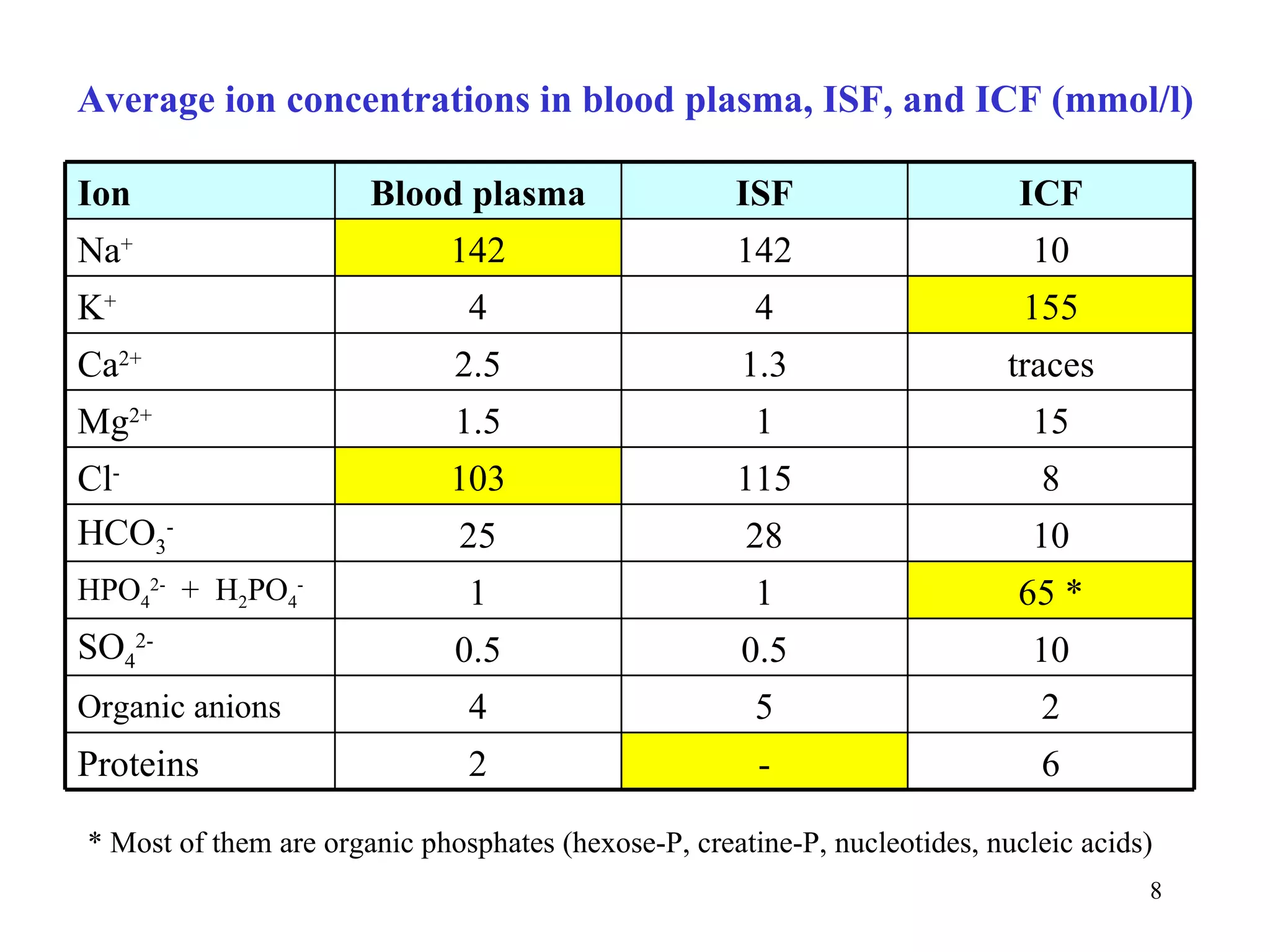
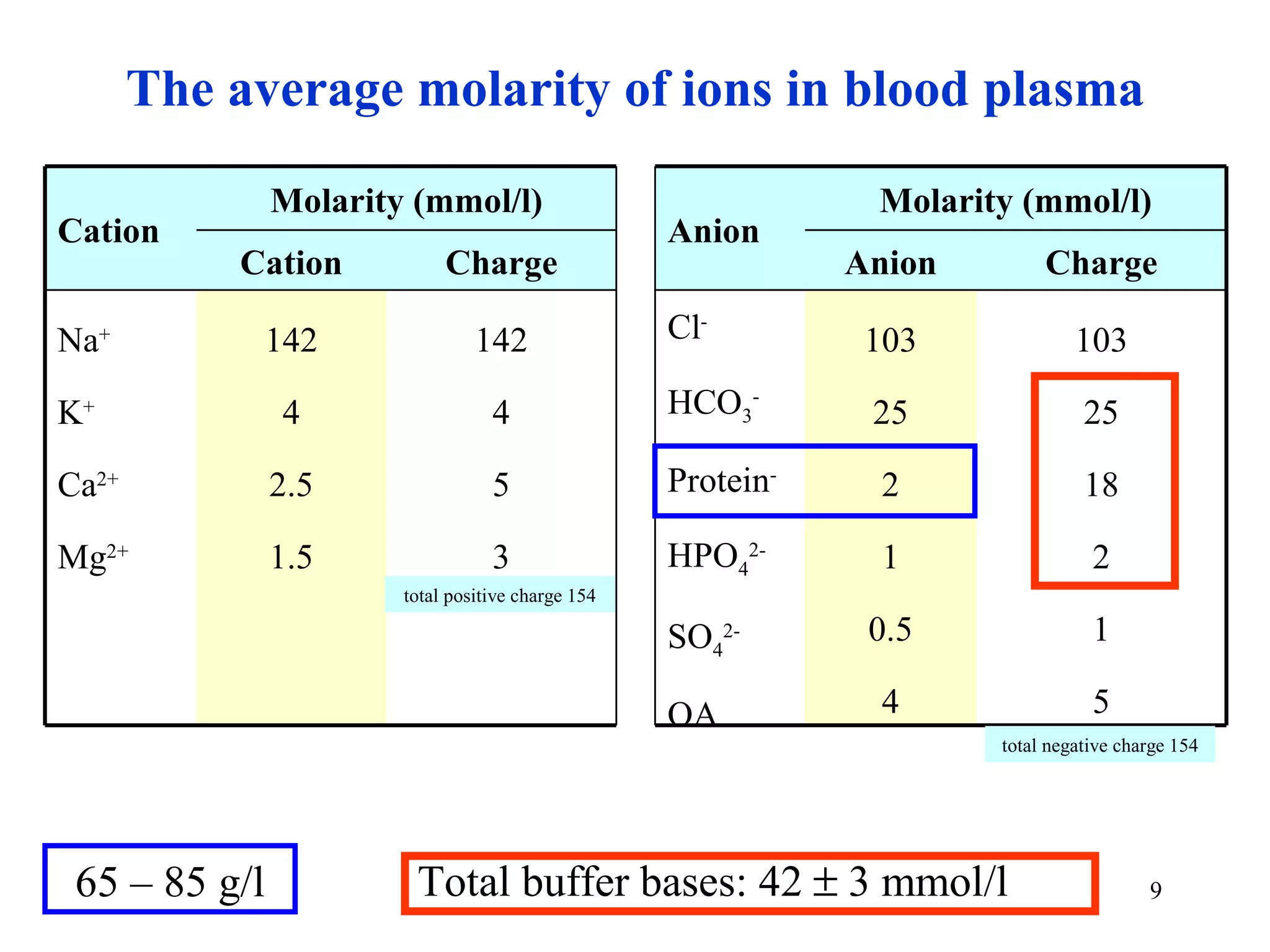
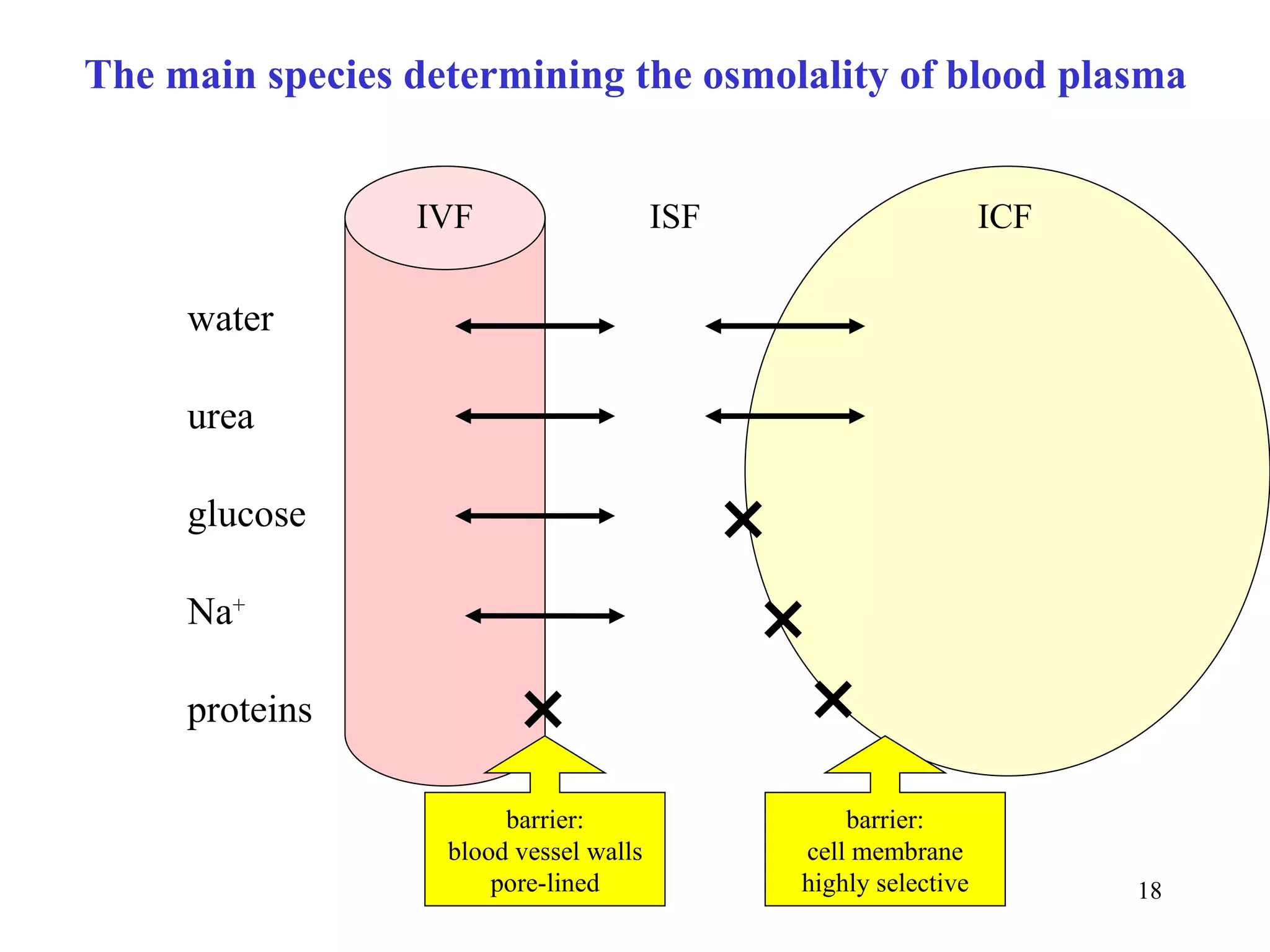
![Comments to ionic composition of body fluids
• blood plasma and ISF have almost identical composition, ISF does not contain proteins
• the main ions of blood plasma are Na+ and Cl-, responsible for osmotic properties of ECF
• the main ions of ICF are K+, organic phosphates, and proteins
• every body fluid is electroneutral ⇒ [total positive charge] = [total negative charge]
• molarity of charge (mmol/l) = mEq/l (miliequivalent per liter)
• in univalent ionic species (e.g. Na+, Cl-, HCO3-) ⇒ molarity of charge = molarity of ion
• in polyvalent ionic species ⇒ molarity of charge = charge × molarity of ion,
e.g. SO42- ⇒ 2 × [SO42-] = 2 × 0.5 = 1 mmol/l
• plasma proteins have pI around 5 ⇒ at pH 7.40 they are polyanions
• OA = low-molecular organic anions: lactate, oxalate, citrate, malate, glutamate, ascorbate, KB ...
• hydrogen carbonate, proteins, and hydrogen phosphate are buffer bases
• total plasma proteins are usually expressed in g/l (mass concetration)
• charge molarity of proteins and org. anions is estimated by empirical formulas
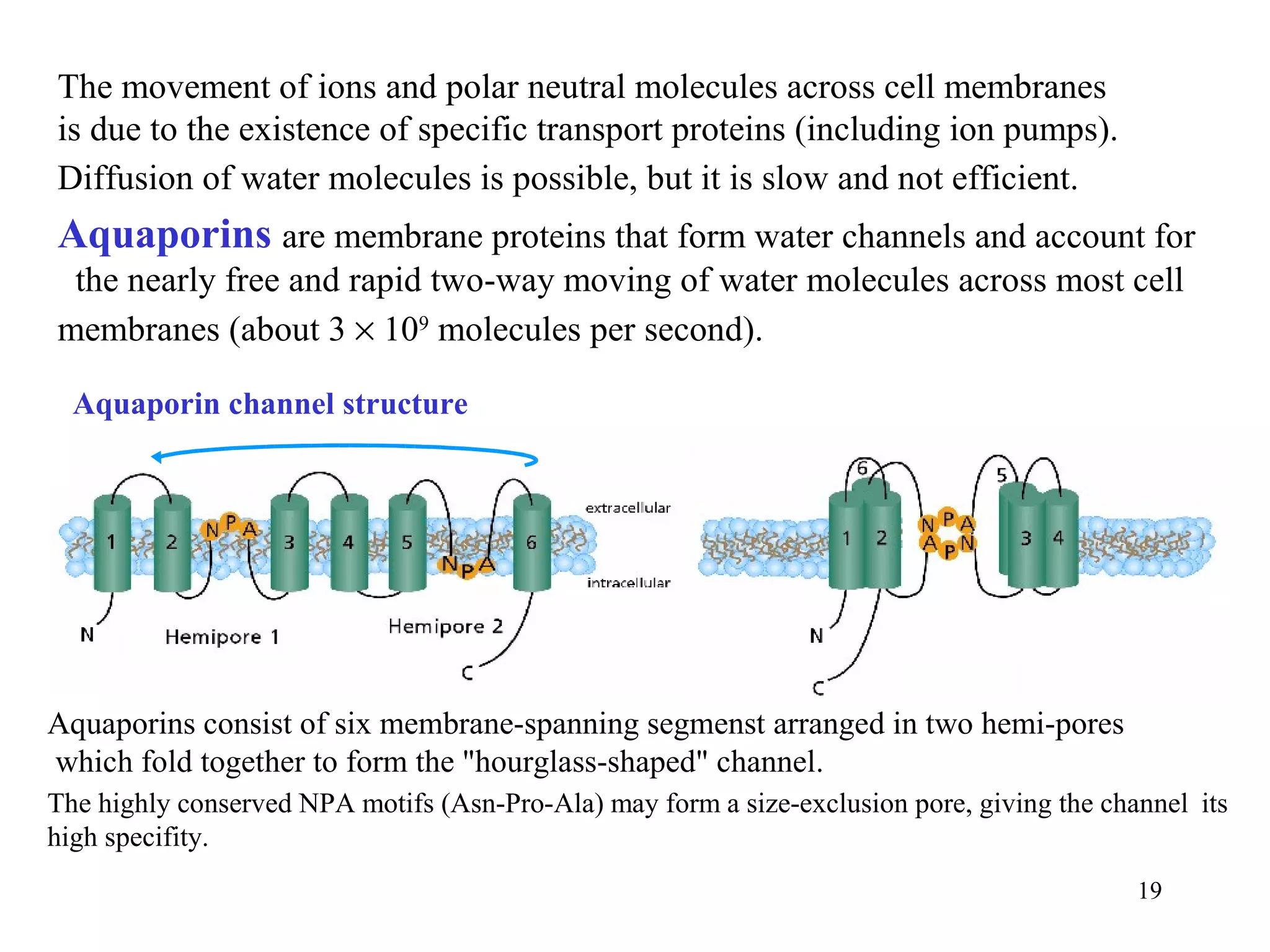
10](https://image.slidesharecdn.com/9-waterions-110920060537-phpapp02/75/9-water-ions-10-2048.jpg)
![Sodium balance
150-280 mmol/day
Input
table salt (NaCl), salty foods (sausages etc.), some mineral waters
ECF (50 %), bones (40 %), ICF (10 %)
Distribution
the main cation of ECF, responsible for osmolality and volume of ECF
130-145 mmol/l
Blood level
gradient between ECF and ICF is created and maintained by Na+, K+-ATPase
aldosterone = salt conserving hormone
Regulation
ANP = atrial natriuretic peptide = antagonist of aldosterone
120-240 mmol/day (urine)
Output [99 % of filtered Na+ is reabsorbed in kidneys]
~10 mmol/day (stool), 10-20 mmol/day (sweat)
11](https://image.slidesharecdn.com/9-waterions-110920060537-phpapp02/75/9-water-ions-11-2048.jpg)




![males 290 ± 10 mmol/kg H2O
Osmolality of blood plasma females 285 ± 10 mmol/kg H2O
Osmolality of biological fluids is measured by osmometers based mostly on the
cryoscopic principle.
Osmolality of blood plasma depends predominantly on the concentrations of Na+,
glucose, and urea. Even if the osmolality of a sample is known (it has been
measured), it is useful to compare the value with the approximate assessment:
osmolality (mmol/kg H2O) ≈ 2 [Na+] + [glucose] + [urea] (mmol/l)
An osmotic gap can be perceived in this way. The measured value is higher
than the calculated rough estimate, if there is a high concentration
of an unionized compound in the sample (e.g. alcohol, ethylene glycol, acetone).
[One gram of ethanol per liter increases the osmolality by about 22 mmol/kg H2O]
21](https://image.slidesharecdn.com/9-waterions-110920060537-phpapp02/75/9-water-ions-21-2048.jpg)