The 2010 SWISS Medical Technology Industry report highlights the maturation of the medtech sector, indicating a shift from a focus on non-reimbursed products to innovation and strategic collaboration. It emphasizes the need for companies to adapt their business models and engage in research and development to sustain growth amidst increasing global competition and market pressure. The overall outlook suggests continued growth potential, particularly through emerging markets and innovative product offerings.







![A STATE OF THE MEDTECH INDUSTRY
Switzerland is among the global hotspots for MedTech
INTERNATIONAL MEDTECH FOCUS [% of respective total]1) COMMENTS
1,1 Switzerland • Switzerland possesses an active medical
0,3 Germany technology industry that can rely on a strong
Employment 0,2 UK network of related companies and institutions
EU that create an ideal breeding ground for the
0,2
USA industry (cluster)
0,2
• Compared to other countries the relative
1,9 domestic share of the SMTI is significantly higher
0,7 • Remarkable – and in line with several other
GDP 0,6 Swiss industries – is the high relative share of
0,5 exports reflecting the international strength of
0,5 Swiss MedTech products
5,0
1,4
Exports 1,7
n.a.
2,8
1) Data from 2008 or, where not available, from 2007
Source: SMTI 2010, Destatis, Eucomed, Eurostat, UK National Statistics 10](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-10-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Switzerland is highly attractive for international companies –
Top 10 companies employ around ¼ of SMTI employees1)
1 Year sales R&D Sales/
Head- Employees in Sales R&D/sales
No. Company Sub-section of market growth expenses employee
quarters Switzerland [CHFm] [%] 3)
[%] [CHFm] [CHFk]
1 Synthes Orthopedics USA 2'900 3'523 6 175 5 329
Roche
2 In-vitro diagnostics CH 1'750 10'055 4 987 10 387
Diagnostics
J&J
3 2) Orthopedics USA 1'400 24'492 2 1'830 7 556
Medical
4 Zimmer Orthopedics USA 1'150 4'250 -1 213 5 518
5 Sonova Hearing systems CH 1'030 1'500 20 87 6 221
6 Ypsomed Injection systems CH 1'000 254 -8 32 12 212
Active and passive implants,
7 Medtronic USA 900 15'151 8 1'406 9 399
vascular diseases and diabetes
8 B. Braun Orthopedics, hospital aids DE 840 5'992 6 207 3 152
9 Straumann Dental implants CH 800 736 5 39 5 339
10 Stryker Orthopedics USA 600 6'977 0 349 5 375
∑ 12'020 Ø 4 Ø 7
1) Approximately, manufacturers only, figures for reporting year 2009/10, exchange rates: USD/CHF: 1.03, EUR/CHF: 1.49 on 31.12.2009
1) J&J Medical: Numbers are the total for orthopaedics, surgery, cardiology and diagnostics
Source: SMTI 2010, Annual reports, Companies press offices, Factiva, Oanda 11](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-11-2048.jpg)

![A STATE OF THE MEDTECH INDUSTRY
The SMTI has a strong home market – Investments in high-class
medical infrastructure meets patients demands
SWISS HEALTHCARE EXPENDITURES [CHF bn per year] COMMENTS
• Continuous investments in up-to-date medical
infrastructure leads to rising healthcare
expenditures – during the last ten years by
CAGR 63,3 nearly 50%
4,0% 61,0
58,1 • The CAGR of healthcare expenditures of
55,2
52,8 4.0% is significantly higher than the
51,0 52,0
47,4
49,3 respective GDP growth of 1.1%
45,6
42,8 FURTHER OBSERVATIONS
• Today Switzerland has the third highest GDP
share of healthcare expenditures (10.6%)
after the USA (16% in 2007) and France
(11% in 2007)
• To constrain rising national healthcare costs,
policies will increasingly focus on cost-
benefit comparison of treatments
• The SMTI companies will have to monitor
upcoming policy changes and be ready to
2000 2001 2002 2003 2004 2005 2006 2007 2008E1) 2009E 2010F1)
adapt the business model accordingly
1) E = Expected 2) F = Forecast
Source: SMTI 2010, BAG, BfS, Healthcare Monitor 2009, KOF 13](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-13-2048.jpg)

![A STATE OF THE MEDTECH INDUSTRY
SMTI manufacturers are evenly segmented across the different
industry categories
MANUFACTURERS BY CATEGORY [%, 2010] COMMENTS
• The manufacturing base of SMTI is
segmented into 16 categories, ranging from a
Hospital share of 16% for hospital hardware to 1% for
Others1)
hardware
biological products
16%
19% • SMTI manufacturers exhibit a broad
technological knowledge base and
Reusable Dental diversification
instruments technology – Bulk risks are reduced
5%
14%
– Benefits interdisciplinary R&D
Radiotherapy
5% collaborations
devices
– Strengthens network innovation
Disposable 6%
items 11%
Inactive
FURTHER OBSERVATIONS
Technical aids 7%
implants
for disabled 8% 8% • There exists a high degree of collaboration
In-vitro Electro- amongst suppliers, manufacturers, and
diagnostics mechanical academia to
– broaden the offering scope
n = 320 – obtain key skills and know-how
1) Others include, in descending order: lab equipment, ophthalmology, patient aids, anaesthetic and respiratory technology,
active implants and biological products
Source: SMTI 2010 15](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-15-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Suppliers and service providers support the MedTech industry with
essential know-how within augmentable value chain processes
SUPPLIERS BY CATEGORY [%, 2010] SERVICE PROVIDERS BY CATEGORY [%, 2010]
Components,
Others1) Financing HR
systems
Auxiliary material, 11% 5%
Analytics IT
green ware 1%
5% 25%
18% 30%
Electronics 7%
Surface
8%
treatment
19% 20%
Machines, auto- 11% Metal Engineering,
mated devices processing planning, 26%
13% Management
design
Plastics consulting
processing
n = 410 n = 410
• The SMTI supplier base is broadly specialized, • IT and management consulting are the most
although components, systems and material prominent categories among service providers
processing contribute to more than 55% • Support SMTI with growth capabilities
1) Others (in descending order) include: medical packaging, measuring, ceramics and sterilization
Source: SMTI 2010 16](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-16-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Manufacturers and traders/distributors are highly focused on
medical devices sales
SHARE OF TURNOVER [%, 2009] COMMENTS
• Many SMTI companies are not exclusively
100 100 100 100 focused on medical technology – the share
15
of sales to other industries and consumers is
19
Turnover from
39%
47
other products 56 • Traders and distributors as well as suppliers
and services Ø 61 show an increased focus on MedTech
compared to 2007
81 85
• Suppliers are more diversified in their
Turnover from 53
44 customer portfolio
medical devices
FURTHER OBSERVATIONS
Suppliers Manu- Traders Service
facturers and providers • A high focus on medical devices is needed in
distributors order to meet the industry specific traits
Turnover and sales requirements
from medical • On the whole, SMTI remains unchanged
39% 84% 80% 59% Ø 65
devices in since 2007, indicating that consolidation has
2007 not been triggered yet by the financial crisis
• For many suppliers from other industries,
2009 n = 212 2007 n = 122 MedTech became highly attractive
Source: SMTI 2010 17](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-17-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Manufacturers form the backbone of the industry –
Average turnover per company is CHF 68 million
AVERAGE COMPANY TURNOVER1) [CHF m, 2009] COMMENTS
• The SMTI has a manufacturer centric market
model
68
• Turnover with medical devices is driven by
– Offering/portfolio strategy, i.e. niche vs. big
player
– Company size
– Upfront investments, i.e. machinery, IP
– Position within the value chain
– Competitive landscape, i.e. number of
competitors within/adjacent to the industry
22 FURTHER OBSERVATIONS
• Future sustainable turnover is mainly
11
determined by the following influencers
3 – Overcome critical mass
– Achieve higher expertise to maintain
Suppliers Manu- Traders and Service compliance
facturers distributors providers – Anticipate power shift towards payers
– Meet changes in sourcing strategies of
n = 171 payers
1) Only with medical devices or components for medical devices
Source: SMTI 2010 18](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-18-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Manufacturers and suppliers provide the highest employment,
accounting for 92% of the total SMTI workforce
DISTRIBUTION OF EMPLOYEES [%, 2009] COMMENTS
• The surveyed companies employ around
27% of the total SMTI sector workforce
56%
• Traders, distributors and service providers
account for 8% of the surveyed workforce
• Across all SMTI firms approximately an
average of 59 people are employed
36% – 126 by manufacturers
– 55 by suppliers
– 25 by traders and distributors
– 7 by service providers
• However, a significant bandwidth has to be
taken into account; of all surveyed companies
– 94% employ less than 250 employees
5% – 75% employ less than 50 employees
3%
– 42% employ less than 10 employees
Suppliers Manu- Traders and Service
facturers distributors providers
n = 199
Source: SMTI 2010 19](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-19-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Average turnover was influenced by high staff levels – This led to
an underperformance of turnover per capita for manufacturers
AVERAGE TURNOVER PER EMPLOYEE AVERAGE GROWTH IN NUMBER OF EMPLOYEES
[CHF k] [% change vs. previous two years]
2008/2009 Ø = +10% 2006/2007 Ø = +20%
244 11
Suppliers
246 7
530 30
Manufacturers
465 5
Traders and 683 15
distributors 818 12
301 2007 n = 185 18 2007 n = 186
Service providers
287 2009 n = 163 21 2009 n = 164
• Traders/distributors experienced growth in turnover per capita, the others remained stable or declined vs. 2007
• The build-up of employees in 2006/2007 led to an overcapacity in 2008/2009, SMTI companies responded by
adapting their recruitment policies
• Unlike many other Swiss industries the SMTI continue to hire above market average, but at a slower pace,
continuing to provide attractive jobs for skilled employees, especially in the fields of R&D, regulatory affairs and
international marketing & sales
Source: SMTI 2010 20](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-20-2048.jpg)
![A STATE OF THE MEDTECH INDUSTRY
Putting past SMTI growth expectations into perspective –
Adjustments were taken fast and early in the downturn
GROWTH EXPECTATIONS FOR 2008 AND 2009 [%] COMMENTS
Growth Share of responses • In 2008, SMTI expected an average growth of
expectations 100% 6%. 77% of CEOs predicted a positive growth
rate1)
Strong growth • The "crisis resistance" of the MedTech
(> 10%) industry became attractive for non-industry
players (mainly suppliers), they were
– Trying to enter a profitable market
– Broaden sales channels and offerings
• As the market turned in Q3 2008 and took its
Moderate growth full impact in 2009 on a global scale, SMTI
(0 to 10%) companies adjusted their growth expectations
– Then, only 54% of companies expected a
Neutral (0%) positive growth rate
Moderate decline – The number of companies expecting a
(-5 to 0%)
negative impact on sales doubled
Strong decline
– Suppliers with a higher non-MedTech
(< -5%)
product portfolio proportion were hit harder
2008 2009 – Manufacturers of non-reimbursed product
n = 280, Ø = 6% n = 21, Ø = 0.1% suffered
1) SMTI 2008 data collected in spring and report released end of August 2008
Source: SMTI 2010 (based on SMTI survey May 2010), CS Economic Research, OECD 21](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-21-2048.jpg)



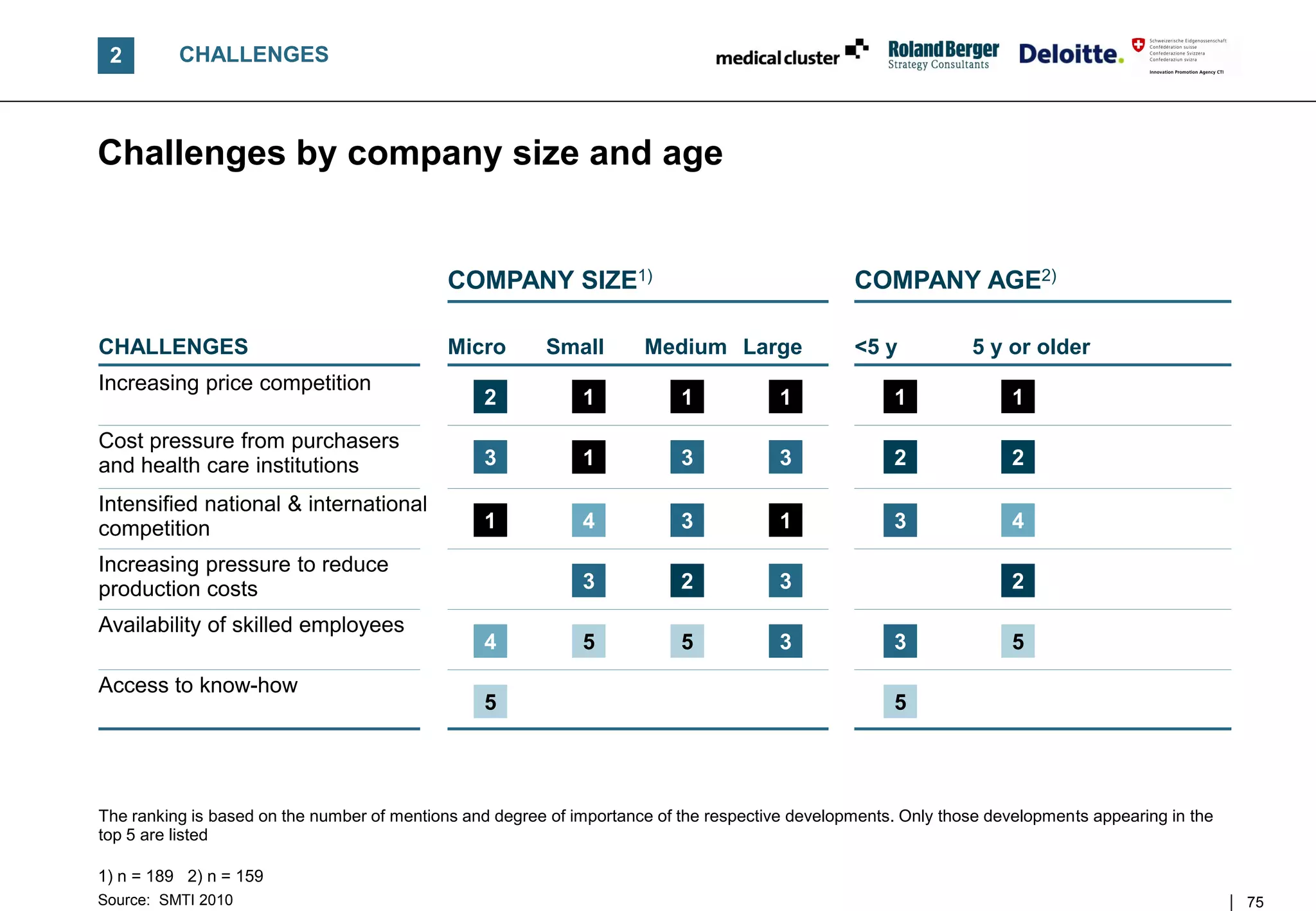
![B TRENDS AND CHALLENGES
Competition for the SMTI intensified severely since 2008 –
Costs and prices are under pressure in a changing market
RANKING OF MAJOR CHALLENGES
[% of answers, 2010]1) COMMENTS
Ranking • As of 2009, SMTI companies experienced
2008
1. Increasing price
intensifying competitive pressures
79 3.
competition – Suppliers with a high proportion of non-
healthcare clients were hit first
2. Increasing cost pres-
77 4. – Manufacturers of non-reimbursed products
sure from purchasers
were hit by tighter consumer spending
3. Intensified competition 75 5. • Today, the majority of SMTI managers are still
in the "crisis mode" observing operational
4. Increasing pressure to
challenges, e.g. higher price competition, cost
68 - pressure and an intensified competition2)
reduce production costs
5. Availability of FURTHER OBSERVATIONS
68 1.
skilled employees
• Current management thinking may fuel
6. Access to specialized consolidation to maintain critical mass to
59 2.
know-how
obtain
7. Organizational issues – A more diverse/innovative product portfolio
53 6.
resulting from growth – Better access to knowledge pools
n = 232; multiple answers possible
1) Challenge is of "high relevance" or "very high relevance" to SMTI companies 2) These represent a "triple challenge" to the SMTI
Source: SMTI 2010 27](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-27-2048.jpg)
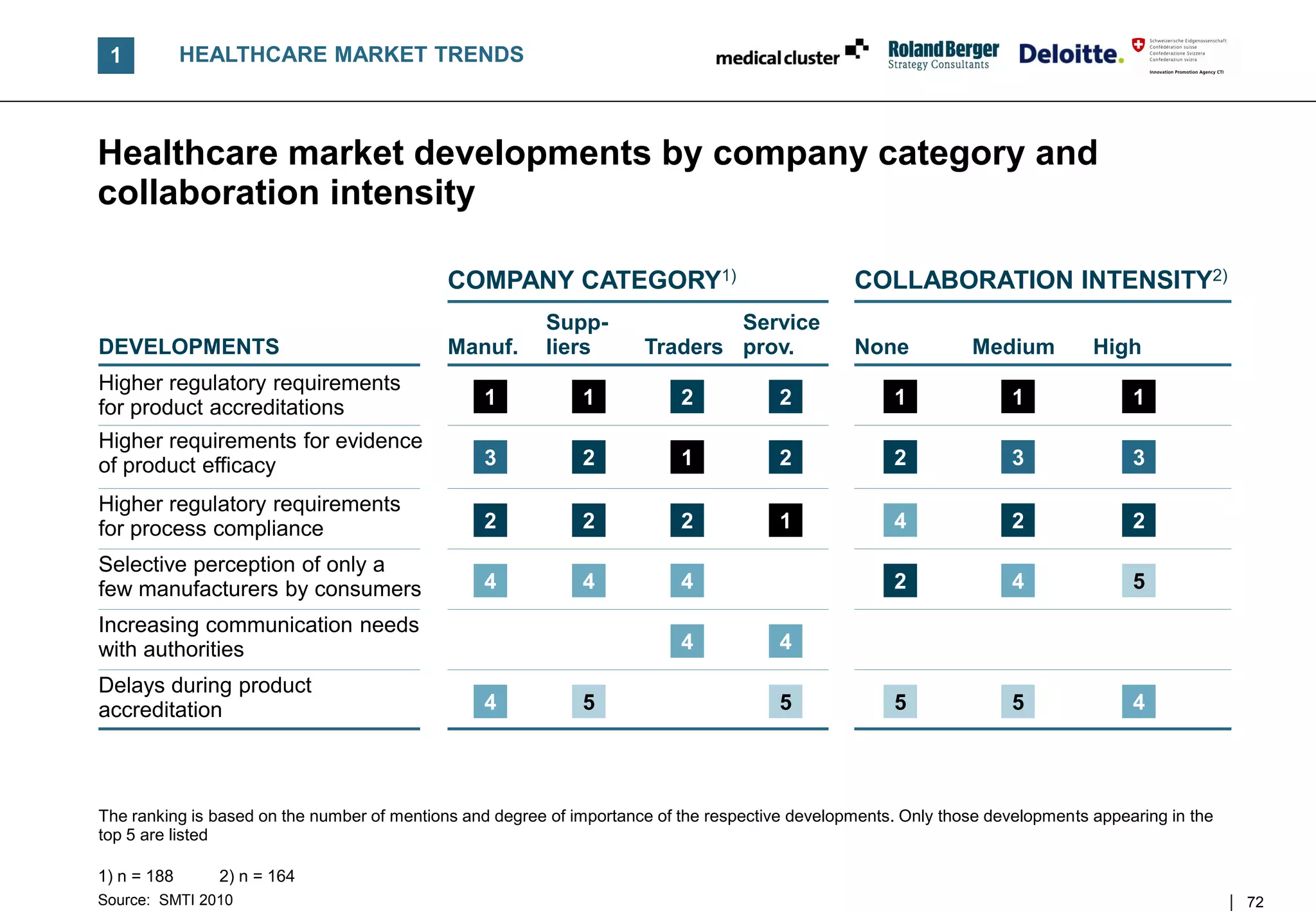
![B TRENDS AND CHALLENGES
Megatrend – Clinical based evidence requirements force
companies to focus on higher product efficacy and communication
DEVELOPMENTS IN HEALTHCARE MARKET
[% of answers, 2010]1) COMMENTS
• Increasing competition and tightening public
Higher requirements for evidence healthcare budgets force companies to invest
of product efficacy 87
more into proofing product efficacy
• Gaining data from comparative effectiveness
Increasing communication needs with research and cost-benefit-analyses will gain
with authorities/healthcare institutions 73
in importance for many SMTI companies
• The communication efforts with customers and
Growth of purchasing cooperatives
61 authorities need to be intensified in order to
(among customers)
optimally position own products
Higher competition for reim- FURTHER OBSERVATIONS
bursement entitlement 56
• Recent healthcare developments will drive
Higher requirements for product – Higher costs to register and fulfil increasing
positioning among DRGs 51 compliance requirements
– Substantial price decreases in mature markets
Negative impact of DRG introduction
45 • EU wide harmonization of pharma prices is
on average product price
currently discussed; this will also influence the
SMTI
n = 134
1) Companies describing the development "applicable" or "partially applicable"; no 2008 data exist
Source: SMTI 2010 28](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-28-2048.jpg)

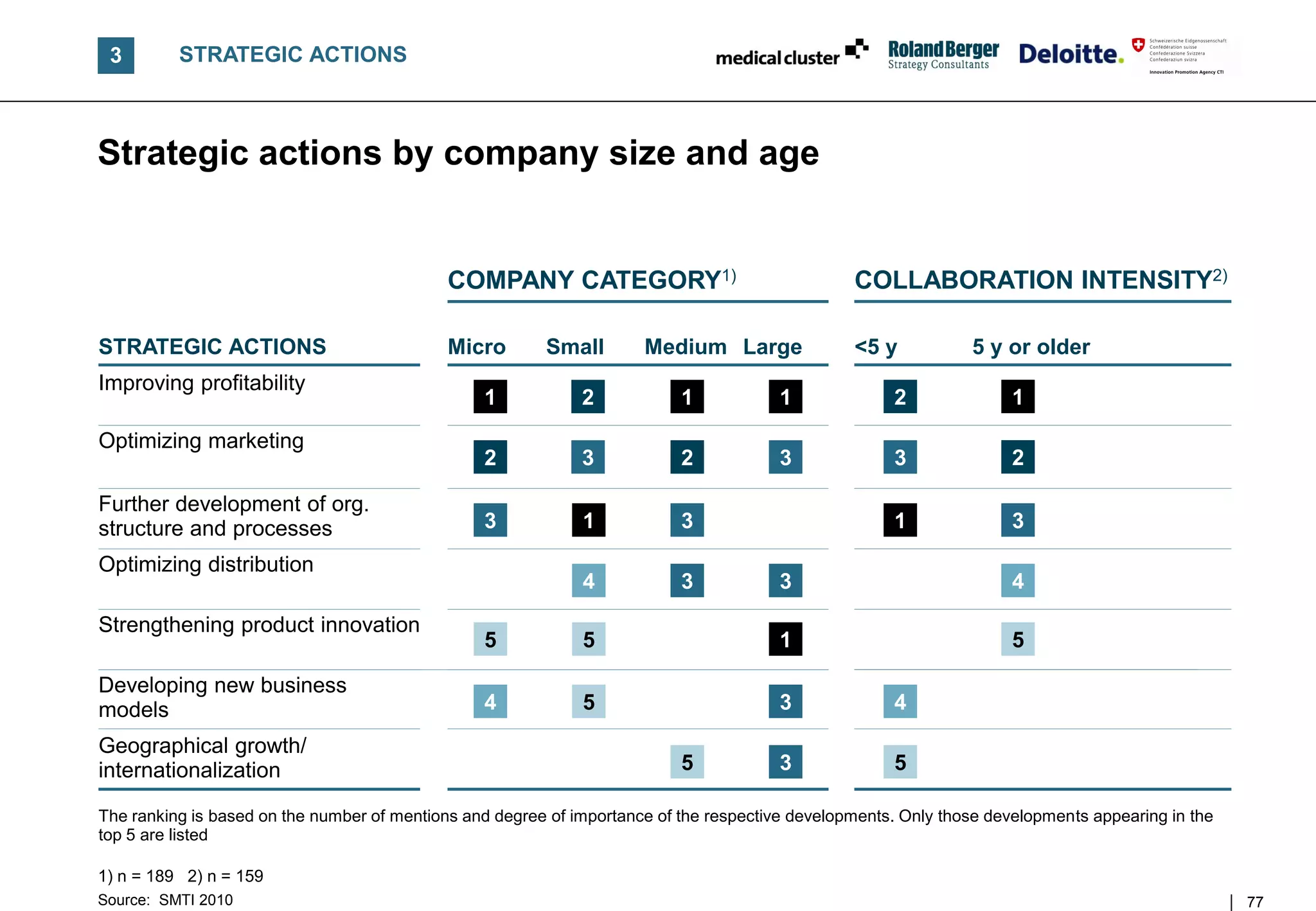
![C STRATEGIC ACTIONS
SMTI companies are focusing on profitability to meet current
challenges at the cost of potentially losing their innovative edge
RANKING OF ACTIONS 2010
[% of answers]1) COMMENTS
Ranking • In contrast to 2007 CEOs express a strong
2008
shift in priorities
1. Improving profitability 85 6.
• To support future growth, three rather
operational actions are in focus
2. Optimizing marketing 77 3.
– Improving profitability
3. Further development of
77 7. – Optimizing marketing & sales effectiveness
org. structure and processes
– Enhancing organizational structures and
4. Optimizing distribution 67 4. processes
5. Strengthening FURTHER OBSERVATIONS
63 1.
product innovation
• SMTI companies seem to approach business
6. Developing new
business models
62 5. conservatively before returning to a "growth
mode"
7. Integrating new partners 59 8.
• An international Life Sciences survey2)
8. Geographic expansion/ indicated contrary management responses
56 2.
internationalization – Developing a robust R&D pipeline
– Accelerating geographical expansion
n = 232; multiple answers possible – Forming alliances with pharma/biotech
1) Strategic actions mentioned as being relevant to SMTI companies 2) Deloitte/The future of the Life Science industries
Source: SMTI 2010 30](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-30-2048.jpg)
![C STRATEGIC ACTIONS
SMTI companies expect 10-12% growth p.a. in the next two years
EXPECTED GROWTH FOR 2010 AND 2011 [%, p.a.] COMMENTS
• On average SMTI CEO's are highly confident
Ø =10% Ø =12%
100% – 10% growth for 2010
– 12% growth for 2011
• Some caution should remain; these
expectations are related to the economic
Strong growth situation in May 2010
55% 59%
(> 10%)
• Start-ups are particularly enthusiastic
• Established companies expect moderate
growth as they face stronger price pressures
Moderate growth
(0 to 10%)
FURTHER OBSERVATIONS
Neutral (0%) 28%
26%
Moderate decline
(-5 to 0%) • If such up-turn is achievable, a shift towards
6% strategic investments and increased
Strong decline 1% 13%
(< -5%) 10% convergence will occur
1% 1% • Current economic forecast for the Swiss Life
2010 2011 Sciences sector as a whole is 6.5%
n = 194 n = 188
Source: SMTI 2010, Credit Suisse, SwissRe 31](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-31-2048.jpg)
![C STRATEGIC ACTIONS
Engaging in the right strategic actions correlates with above-
average growth expectations
EXPECTED GROWTH RATES 2010 AND 2011
[%, companies having the respective strategic action in focus] COMMENTS
16
Companies focusing
15 • Several CEOs focus on a set of strategic
on strengthening Ø 12 actions to enhance their growth potential
regulatory know- Ø 10
how/studies, e.g.
– Strengthening regulatory know-
cost-benefit analyses how/studies
n = 51
– Establishing new production/service
facilities
17 – Engaging in new business models
Companies focusing
on establishing new
12
Ø 12
• If a company focuses e.g. on strengthening
production/service Ø 10 of regulatory know-how/studies, they expect
facilities, e.g. in a growth rate that is 6% above the average
emerging markets
n = 42 for 20101)
15 FURTHER OBSERVATIONS
Companies focusing
on engaging in new
10 Ø 12
business models,
Ø 10 • Companies focusing on new business models
e.g. new kind of do not expect an immediate above-average
services n = 64 effect on their growth rate
2010 2011
Ø = average growth expectations across all survey participants
1) Data does not support a cumulative growth impact of combining the actions
Source: SMTI 2010 32](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-32-2048.jpg)
![C STRATEGIC ACTIONS
The majority of SMTI companies face increasing challenges –
Negative impact on growth is expected
EXPECTED GROWTH RATE [%, companies that are
affected by the mentioned challenges] COMMENTS
Ø 12 • 50% of SMTI companies face a triple
challenge
10 – Increasing price pressure
Ø 10 – Increasing cost pressure
8
– Intensifying competition
• These companies
– Expect a below average growth rate for
2010/2011
– Cannot hope for a fast improvement of their
situation
• All survey participants face at least one of the
three mentioned challenges
FURTHER OBSERVATIONS
• Tighter public healthcare expenditures will
2010 2011
– Keep the pressure on prices
Companies experiencing the "triple challenge"1) – Intensify competition among companies
Ø = Average growth expectations across all survey participants – Force them to decrease their costs basis
• Within this setting companies tend to invest
2010 n = 128, 2011 n = 221 more in product line extensions/renovations
1) Companies stating that increased pressure on prices, costs and intensifying
competition were of "high relevance" or "very high relevance" to them
Source: SMTI 2010 33](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-33-2048.jpg)

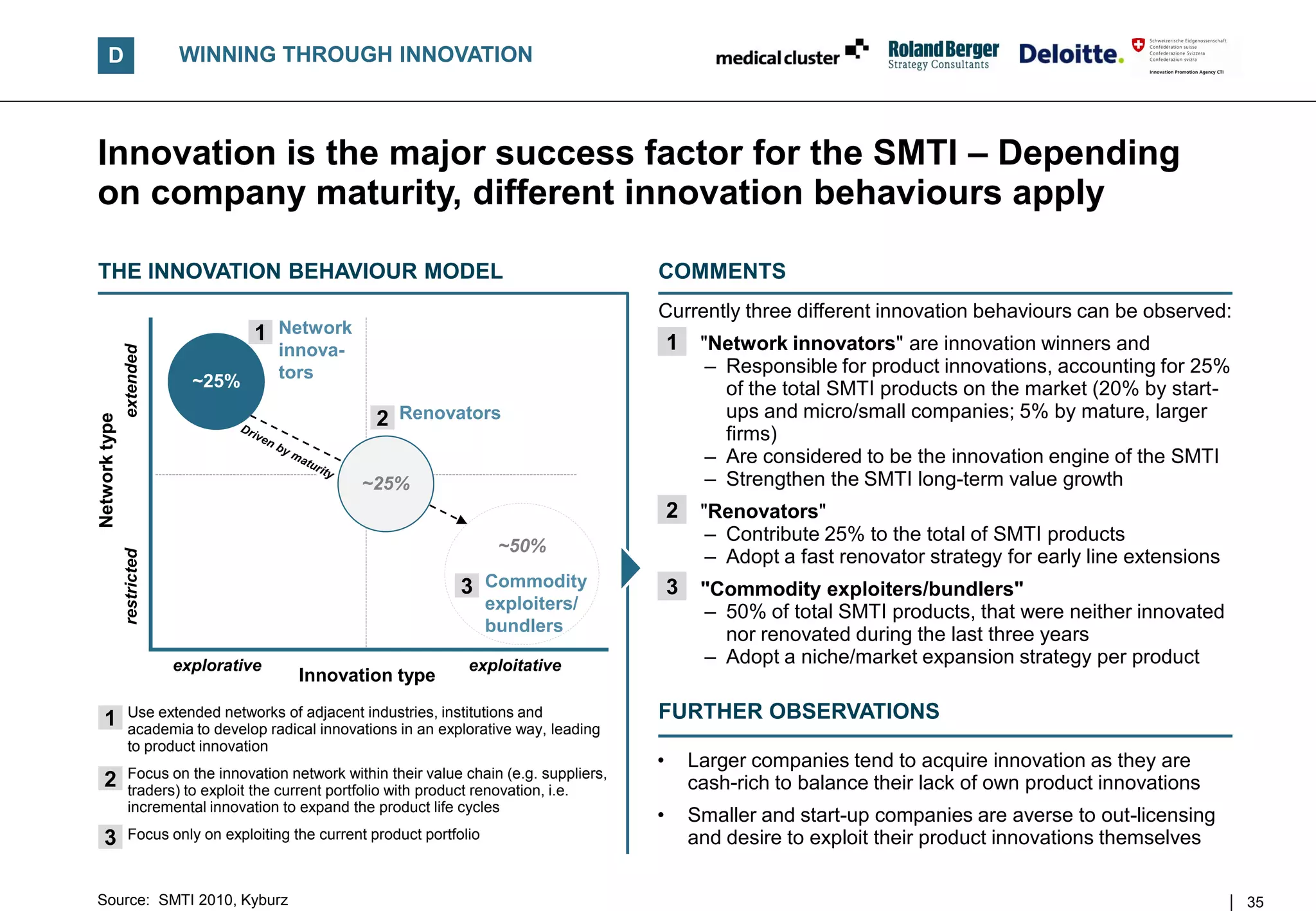
![D WINNING THROUGH INNOVATION
The SMTI shows a balanced product portfolio maturity –
Especially smaller companies drive product innovation
AGE OF PRODUCT PORTFOLIO BY COMPANY SIZE1, 2)
[in %, 2010] COMMENTS
Years
• In general, the maturity of the product
portfolio is well balanced – but the larger the
100%
company, the more mature the portfolio
+10 20%
26% 27% • 25% of the (weighted) SMTI product portfolio
33% is younger than three years
• 81% of the product portfolio of larger
5-10 21% companies is older than three years
27% FURTHER OBSERVATIONS
33% 20%
3-5 25% • Lower priority of product innovation
combined with cost constraining measures
20%
25% is likely to
24% – Lead to fewer novelties
0-3 34% – Prompt more renovation activities
27% • Close attention should be paid to the
22%
16%
maturing product portfolio (see strategic
actions) in order not to fall into an
Micro Small Medium Large innovation gap, as average product
development time is around three years
n = 124
1) Only manufacturers and suppliers 2) In proportion of their product portfolio
Source: SMTI 2010 36](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-36-2048.jpg)
![D WINNING THROUGH INNOVATION
Relative R&D expenditure is highest for small companies –
Manufacturers spend more on innovation than suppliers
BENCHMARKS
R&D EXPENDITURES [% of turnover]1,2) [in % of turnover, 2009]4) COMMENTS
BY COMPANY TYPE • R&D expenditures remained
12 12 stable or even increased since
10 Biotech 22 2007, but were increasingly
5 shifted to incremental innovation
• Manufacturers drive SMTI
innovation; their share of R&D
Pharma 16
2007 2009 spending is higher than for
n = 90
suppliers
• The less turnover a SMTI
Medical
BY TURNOVER company has, the higher the
technology 11
[in CHF and % of turnover 2009]3) industry relative spend on R&D
14
FURTHER OBSERVATIONS
11 10
9 Ø 10.6 Electronics
7 7 5 • Compared with adjacent
industry
industries, the SMTI spends
less on R&D than the biotech
and pharma industry, but
< 5m 5-10m 11-50m n = 90 Machine significantly more than the
4
industry mechanical engineering and
Suppliers Manufacturers electronics industry
1) Only manufacturers and suppliers 2) R&D expenditure was not weighted to turnover
3) Companies with more than CHF 50m turnover were omitted due to a limited sample size 4) Based on a sample of selected companies
Source: SMTI 2010 37](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-37-2048.jpg)
![D WINNING THROUGH INNOVATION
Shortening of the innovation cycle is of significant relevance,
especially to medium and large sized companies
SIGNIFICANCE OF A SHORTENED INNOVATION CYCLE [2010, in %]1)
BY COMPANY TYPE BY COMPANY SIZE
Manu-
45 25 30 Micro 36 26 38
facturers
Suppliers 62 17 21 Small 60 16 24
No or low significance Medium 80 16 4
High significance
Very high significance
Large 78 22 0
n = 129
• The majority of companies feel the market and • Micro companies seem to be flexible enough to adapt
competitive pressures to shorten innovation to shortened innovation life cycles. Their dependency
cycles in order to launch new or improved products on often only one or two products makes it a critical
more rapidly task to stay ahead of competitors
• It seems that manufacturers are partly passing on • The larger a company is, the less flexible it seems to
this pressure, evaluating different suppliers for the speed-up the innovation cycle, mainly because of
most innovative and cost effective solution internal processes and compliance requirements
1) Only manufacturers and suppliers
Source: SMTI 2010 38](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-38-2048.jpg)
![D WINNING THROUGH INNOVATION
Launches during the last three years were difficult – On average
new products achieved less than 30% of companies turnover
REVENUE SHARE OF PRODUCTS THAT WERE LAUNCHED
SINCE 20071) [in % of each company class, 2010] COMMENTS
29 • Product launches in the last three years
Very small 16 were also affected by the global economic
(0%-10%) 13 setbacks
16
• A majority of companies stated that new
31 products contribute only to a minor part
Small 37 (less than 30%) to the current turnover
(10%-30%) 42
45 • For micro companies there seems to be a
clear split:
3
Medium 25 – Either they are older companies with a
(30%-50%) 26 mature "cash-cow" product portfolio
16 – Or they are start-ups with a successful
20
product launch just on the brink of
High 18 growing rapidly
(50%-70%) 13 • The other companies capture less returns
23 from new product launches
17 Micro
Very high 4
(>70%) 6 Small
0 Medium
Large
n = 128
1) Only manufacturers and suppliers
Source: SMTI 2010 39](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-39-2048.jpg)
![D WINNING THROUGH INNOVATION
Companies with a young product portfolio show above-average
growth expectations
EXPECTED GROWTH RATES OF COMPANIES ACCORDING
TO PRODUCT PORTFOLIO MATURITY1) [%] COMMENTS
• Companies with a young product portfolio
17
have significantly higher growth expectations
16 • Companies with a more mature product
portfolio may face – especially if combined
with decreased R&D rates – an innovation
Ø 12
gap that harms future growth rates
• In order to increase growth rates again, these
Ø 10 9 companies need to take into account an
7 average "idea to launch" time of approx. three
years
FURTHER OBSERVATIONS
• The SMTI is an innovation-centred
industry based on network innovation
2010 2011 – Launching new products is an essential
growth factor
Older portfolio (companies with less than 15% of the product portfolio
younger than three years), n = 42 – High priority on profitability2) drives bottom-
Younger portfolio (companies with more than 15% of the product portfolio
line improvements, but does not lead to
younger than three years), n = 56 higher growth rates
1) Only manufacturers and suppliers 2) 85% of survey participants stated improving profitability is a key strategic action
Source: SMTI 2010 40](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-40-2048.jpg)
![D WINNING THROUGH INNOVATION
Companies with a high focus on innovation show above-average
growth expectations
EXPECTED GROWTH RATES AND INNOVATION FOCUS1)
[%] COMMENTS
• Compared to 2008, strategic priority moved
away from strengthening product innovation
15 to profitability improvement measures
14
• Additionally, relative R&D expenditures have
– on average – declined
Ø 12
• Both of these two findings lead to assume a
Ø 10 9 lower focus on innovation
8
• However, firms that still focus on innovation
expect significantly higher growth rates
• They outperform expected average growth by
6% and 3% for 2010 and 2011 respectively,
assigning them a leading role in the further
development of the SMTI
2010 2011
FURTHER OBSERVATIONS
Low focus on innovation (low R&D ratio and strengthening of
product innovation not a priority), n = 33
• Listen to/involve patient groups in prototyping
High focus on innovation (high R&D ratio and strengthening of reviews to enable product generation jumps
product innovation a priority), n = 18
1) Only manufacturers and suppliers
Source: SMTI 2010 41](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-41-2048.jpg)
![D WINNING THROUGH INNOVATION
R&D contribution to growth expectations –
Increased R&D expenditure
GROWTH EXPECTATIONS BY R&D EXPENDITURES
[% of turnover] COMMENTS
Growth R&D expenditures 2009 R&D • The focus of many companies on profitability
expectation expenditures
2011 2007
improvement measures lead to a decrease of
average R&D spend to 10.6%1) from 15% in
0%
2007
Neutral 1
(0% p.a.) • However, a few exceptions exist: suppliers
n.a. 5% and manufacturers must invest more in order
to achieve moderate growth
8 2% • Companies that hardly invest in R&D do not
Moderate growth
(0-10% p.a.)
expect a positive growth rate, strengthening
10 6% the assumption that R&D is a key driver for
SMTI growth
7 10% FURTHER OBSERVATIONS
Strong growth
(> 10% p.a.)
16 15% • Suppliers needed to invest heavily into R&D
to meet shorter innovation cycle time
Suppliers' expenditures on R&D
• They may find themselves in a R&D trap due
Manufacturers' expenditures on R&D
to a high dependency on MedTech
2010 n = 61, 2008 n = 154 manufacturers
1) R&D expenditure was not weighted to turnover
Source: SMTI 2010 42](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-42-2048.jpg)


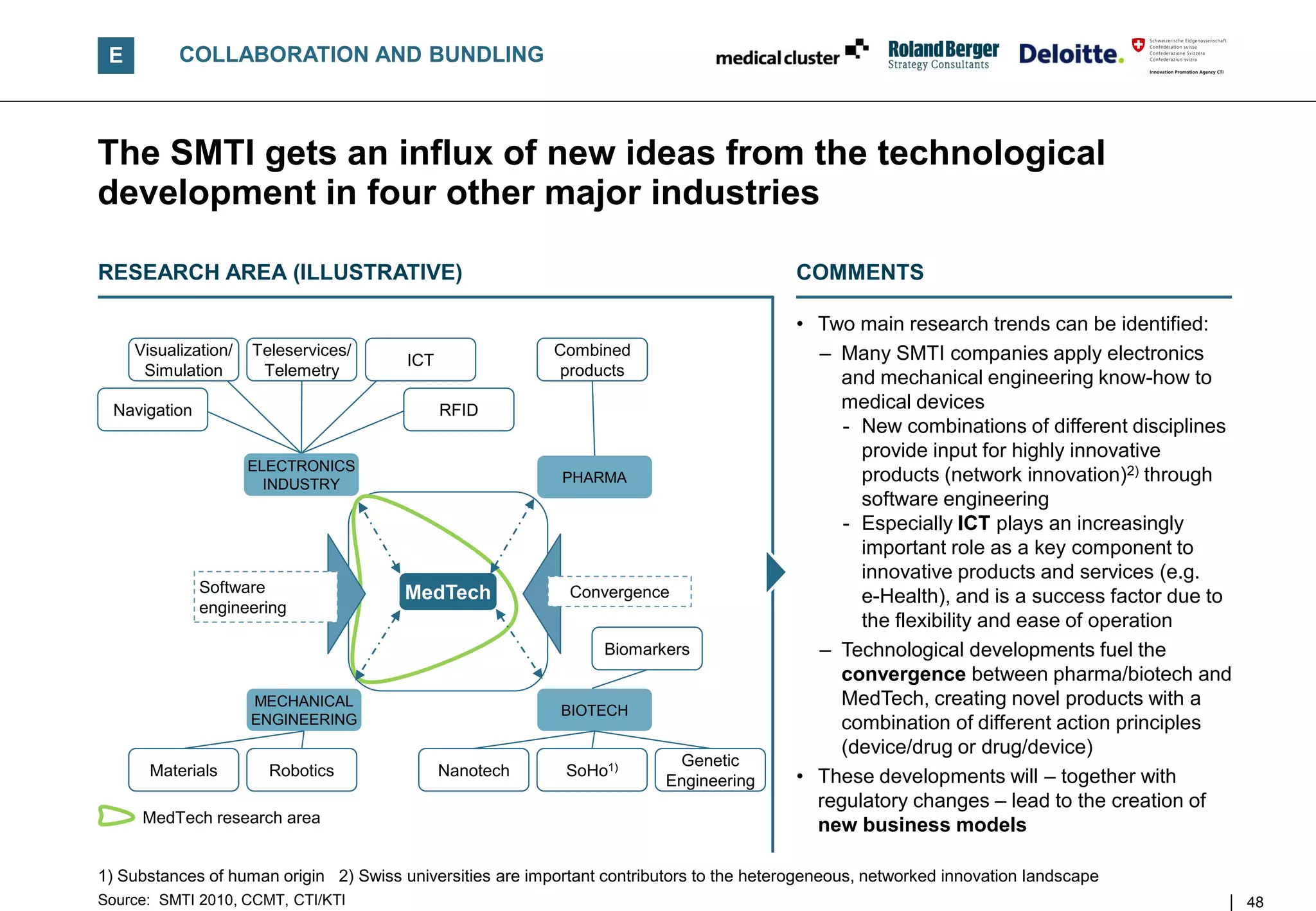
![E COLLABORATION AND BUNDLING
SMTI companies focus on collaborations between manufacturers
and suppliers – Strengthening incremental innovation
SHARE OF COMPANIES WITH COLLABORATIONS
[% of answers, by inter-industry collaboration type, 2010] COMMENTS
• The majority of SMTI companies closely
Manufacturers/ 88 collaborate with other manufacturers and
suppliers 82 suppliers, positioning themselves often as
"Renovators" working on improvements of
43 existing products
Universities
59
• Already second are collaborations with
Technology/IT service 13 universities; there SMTI companies often act
providers 28 as "Network innovators", working on
product innovations
12
Support organizations
24 • On average manufacturers show a higher
degree of collaboration – this is mainly due to
Private research 9
institutes 24 – Technology leadership in projects with
several collaborating parties
Strategy 9 – Higher value chain integration
consultants 4 • Not very widespread are collaborations with
service providers
Suppliers
Manufacturers • Roughly less than a quarter of SMTI
companies have respective collaborations
n = 122, multiple answers possible
Source: SMTI 2010 45](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-45-2048.jpg)
![E COLLABORATION AND BUNDLING
SMTI companies focus on applied research to drive incremental
innovations
FUNCTIONAL COLLABORATION WITH PARTNERS
[% of answers, by type, 2010] COMMENTS
SMTI companies, particularly manufacturers,
Applied research 43
61 engage in functional collaboration. Predominantly,
such activities are sought in the field of:
Quality control 45
46 • Applied research; underlining the focus on
25 incremental innovation
Basic research
35
• Quality control and production certification
Product certification 12 – Strengthening compliance and regulatory
41 know-how
Marketing/sales 30 – Managing innovation cycle
20 – Shifting innovation risks to suppliers
Distribution 17 • Basic research; focus on break-through
28
technology and materials
Training 12
13 • Training, launch/post launch, and after sales
After sales 7
collaborations are of minor importance to SMTI
services 7 firms despite
– Under-proportional returns on new products
5
Product launch
7 – Rising OPEX to sales ratios and process costs
Suppliers – Expected shifts in business models and
offerings
Manufacturers
n = 106, multiple answers possible
Source: SMTI 2010 46](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-46-2048.jpg)
![E COLLABORATION AND BUNDLING
The national innovation programs of CTI/KTI reach 29% of SMTI
companies – More than twice as much as international programs
NATIONAL PROGRAMS INTERNATIONAL PROGRAMS
[% of participation in innovation promotion programs1), 2010] [participation in %, 2010]
65 100 87
OTHER OTHER
CTI/KTI 22 EU
29
35
6
32
29 17 13
2
11
NO YES CTI/KTI micro small medium large NO YES
(distribution by company type)
n = 154 n = 155
EU programs: e.g. FP6, FP7
Other: e.g. COST, EUREKA
1) Suppliers and manufacturers
Source: SMTI 2010 47](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-47-2048.jpg)
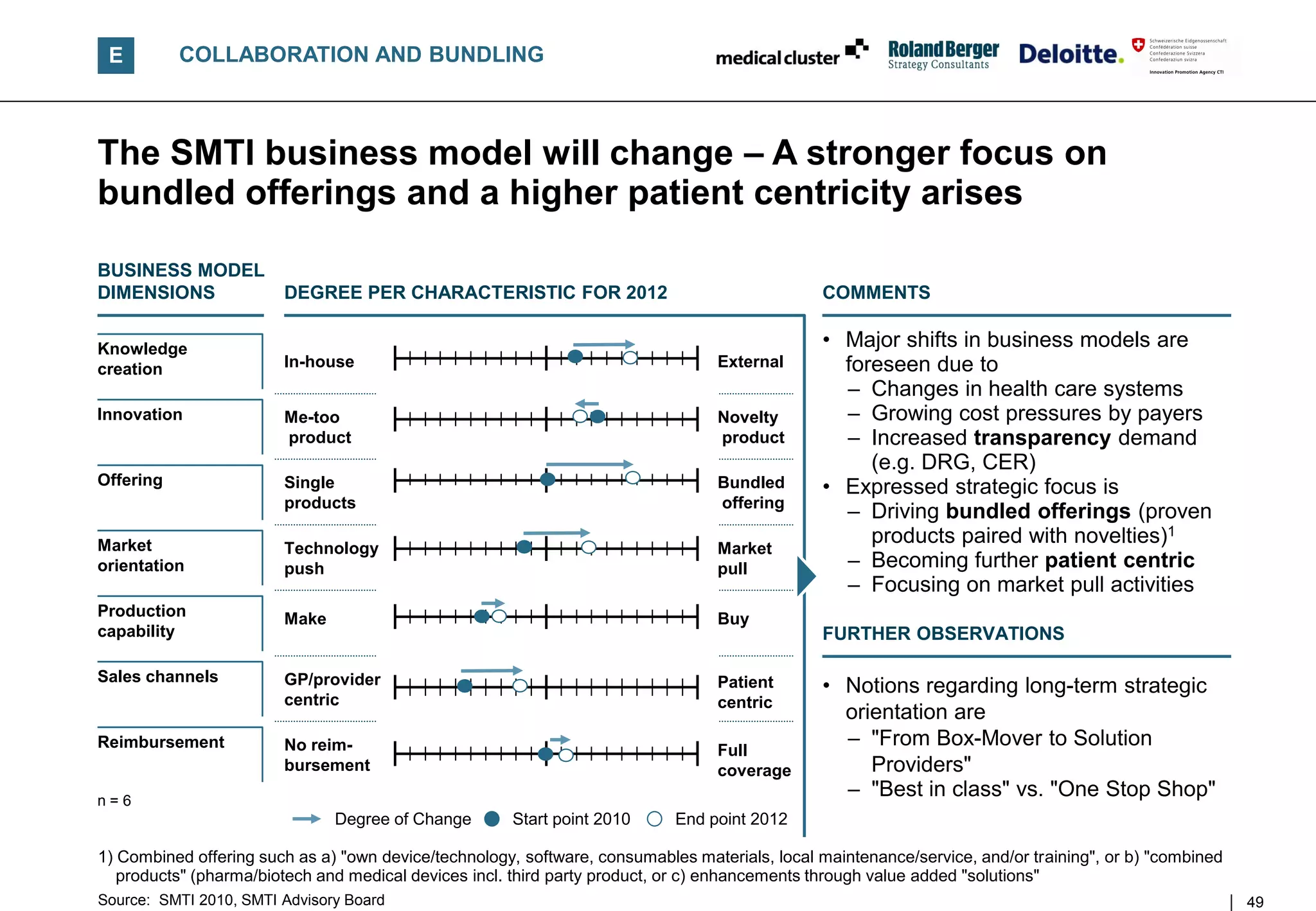
![E COLLABORATION AND BUNDLING
Offerings are challenged by price pressures –
Changes ahead for 2012
POSITIONING AND BREADTH OF
OFFERINGS [illustrative] CHARACTERISTICS OF OFFERING STRATEGIES BY 2012
With reduced funding in health care systems, price pressure will
Best in Class
further increase. Paired with DRG, transparency on patient outcome
high
becomes available to payers
Own product This will influence the companies„ offering strategy – thus they may
choose from the following positioning in the future:
Price
Multiple
indications Commodity • "Best in Class" products
– Unique IP, processes, technology or materials and best
Commodity treatment outcome, thus often benefit drivers
product, single – Yield premium price positions
indication
• "Commodity" (less innovative or "me too"/single) products
low
low
Benefits
high – Aim at high volumes/lower margins pricing
Product categories
– Must provide supply flexibility/reliability
Single product Bundling types
• "Bundling" (to obtain an overall higher price benefit ratio)
own product – Own products, services, consumables, training and IT updates
convergence product – Convergences (combined pharma/biotech and MedTech
solutions product)
– Solutions1 (extended value creating services or complementary
products for a full DRG treatment incl. competitor products)
1) Solutions serving DRG related treatments are currently in early stage discussions amongst leading firms; positioning here is rather indicative
Source: SMTI 2010, SMTI Advisory Board 50](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-50-2048.jpg)

![F INTL OPPORTUNITIES AND THREATS
SMTI companies still perceive the strongest growth potential in the
Swiss home market, closely followed by the EU
COUNTRIES WITH HIGHEST EXPECTED GROWTH
POTENTIAL FOR THE NEXT 2 YEARS [% of mentions, 2010]1) COMMENTS
Switzerland Abroad • Compared to 2008, there were no significant
2010: 35% 2010: 65%
changes:
2008: 34% 2008: 66%
– The Swiss market is still expected to have the
35 34 highest growth potential
– More than half of the SMTI companies expect
30 the highest potential in the EU, USA and BRIC
29
markets
• For the other geographical regions, Swiss
companies tend not to expect important growth
13 13 contributions:
11
10 – In Japan registration and regulatory
7 requirements, as well as cultural differences,
6
are a major hurdle
3 3
2 2
1 1 – The rest of the world market does not yet
contribute significantly to the growth of SMTI
Switzerland EU USA / Japan BRIC Other Other Other companies
Canada countries countries countries
in Asia in Latin
2010 n = 141 America
2008 n = 132
1) Includes suppliers, manufacturers, traders/distributors and service providers
Source: SMTI 2010 52](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-52-2048.jpg)
![F INTL OPPORTUNITIES AND THREATS
Manufacturers are more internationalized than suppliers – More
than 50% perceive strongest growth outside EU and Switzerland
COUNTRIES WITH HIGHEST EXPECTED GROWTH
POTENTIAL FOR THE NEXT 2 YEARS [% of mentions, 2010] COMMENTS
Switzer- • Companies expect the strongest stimulus from EU
land Abroad
countries, followed by Switzerland
• Overall manufacturers seem to be more
35 internationalized than suppliers
32 – About 54% of manufacturers expect highest
30 growth from business with countries outside the
EU and Switzerland
– In the focus are EU, USA/Canada, the BRIC
20 countries and only fourth the Swiss home
18
16 market
14 – Suppliers have a higher focus on the EU and
the Swiss home market, representing for more
8 8 than two thirds of companies the regions with
6 6
the highest growth potential
2 2 2
0 1
FURTHER OBSERVATIONS
Switzerland EU USA / Japan BRIC Other Other Rest of
Canada countries countries the world • Suppliers seem to need a geographical closeness
in Asia in Latin to manufacturers in order to optimally fulfil their
Suppliers n = 49 America
customer requirements
Manufacturers n = 72
Source: SMTI 2010 53](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-53-2048.jpg)
![F INTL OPPORTUNITIES AND THREATS
The economic outlook for main SMTI export markets is positive –
Return to growth expected
GDP GROWTH [% change vs. previous year] COMMENTS
• Most of the main SMTI export markets are
3,1 expected to return to growth in 2010 (0.8 and
2,7 3.1%) and 2011 (1.5 and 2.7%)
2,4
2,1
1,8 1,8 • The OECD average growth rate is expected
1,6 1,5 to slightly outperform Switzerland and to
0,8 recover considerably faster than in the EU-27
0,7
0,5 0,4 • This increases the growth potential through
sales – both in Switzerland and abroad
• Growth prospects are even brighter outside
the traditional European markets, e.g. in the
Switzerland emerging economies in Asia and South
OECD America
-2,4 EU-27
-2,5
USA
-3,3
-4,2
2008 E 2009 E 2010 F 2011 F
E = Estimate, F = Forecast
Source: SMTI 2010, BAG, BfS, KOF 54](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-54-2048.jpg)


![F INTL OPPORTUNITIES AND THREATS
The US Healthcare Reform will also influence SMTI companies
CONVERGENCE OF FOUR FORCES [illustrative] EXPECTED OPPORTUNITIES
• Higher numbers of patients
• Opportunity for highly innovative treatment
demand
• Opportunity to position bundled offerings/solutions
to buying consortiums
• Increased opportunities for preventive/early
indication devices
EXPECTED THREATS
• Higher price transparency
• Higher compliance and registration efforts for new
products, incl. registration fees
• Higher re-negotiation costs of existing products
• Tighter reimbursement for me-too products
• Higher tax burden
Source: SMTI 2010 57](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-57-2048.jpg)
![F INTL OPPORTUNITIES AND THREATS
Registration and introduction of new products are perceived to be
less difficult in Europe than in other markets
ESTIMATED DIFFICULTIES FOR PRODUCT REGISTRATION
AND INTRODUCTION [% of mentions, 2010]1) COMMENTS
• Registration and introduction of new products
1 2 are expected to be a challenging task for 45%
Very difficult 15 13 of manufacturers and suppliers
23
27
26 • However, Switzerland as well as the EU
42 (almost the same processes and structures)
are expected to be less complex than
Difficult 30 38 registration in the US, Japan or BRIC markets
• Japan is considered to be a very difficult
39
52 market due to high requirements regarding
51 28 sensible documents (drawings, IP etc.), high
fees, and long approval times
Neutral 43
• US FDA regulations are perceived as hard to
46 be met, even for large companies
30
20
27 • FDA recently announced higher regulatory
Easy 20 requirements, increasing registration
Very easy 11
4 5 complexity and processing time
1 0 0 0 3 0 3
• Especially smaller SMTI companies face the
Total Switzerland EU USA JAPAN BRIC challenge to meet these requirements due to
n = 90 the lack of internal expertise
1) Only manufacturers and suppliers
Source: SMTI 2010, SMTI Advisory Board, Swissmedic 58](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-58-2048.jpg)

![G INTERNATIONAL POSITIONING
For the first time the SMTI work-force abroad grew at a higher rate
than in Switzerland, showing an increasing internationalization
GROWTH IN LOCAL VS. FOREIGN EMPLOYMENT
[% over last 2 years, end 2007 & 2009]1) COMMENTS
• SMTI companies continue to hire – but at a
21 reduced rate
Switzerland
7
• For the first time growth of employee
16 numbers has been bigger abroad and signals
Abroad
8 a shift in management focus
2007
2007: n = 186 • This result documents the increasing
2009
2009: n = 81 internationalization effort of SMTI companies
• The survey results indicate that no significant
WORKFORCE ABROAD work-force shifts from and to Switzerland
[% of total workforce according to company size, 2009]1) have taken place
• Micro to small companies employ around 4%
Micro 4 of their workforce abroad compared to 20-
40% of medium to large sized companies
Small 4
Medium 20 FURTHER OBSERVATIONS
Large 42
• Micro and small companies are lacking
access to experienced hires in order to obtain
n = 139 international expert know-how
1) Only manufacturers and suppliers
Source: SMTI 2010 60](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-60-2048.jpg)
![G INTERNATIONAL POSITIONING
Manufacturers and suppliers are strongly export driven and
achieve on average 63% of revenues abroad
AVERAGE SHARE OF EXPORTS
[% of turnover, 2009] 1) COMMENTS
78 • Manufacturers generate around 78% of sales
abroad
Ø 63
• Suppliers play an important role in the local
53 market as 47% of their products and services
are sold within Switzerland
35 • Their export share decreased significantly,
reflecting economical difficulties in many
Ø 29
markets, e.g. declining demand, fiercer
16 competition, and adverse currency
fluctuations
• The same applies to a majority of
Suppliers Manu- Traders Service traders/distributors; they only achieved an
facturers and providers2) export share of 16% (-50% vs. 2007)
distributors
• Service providers are generating 35% of their
2007 data 62% 74% 34% turnover abroad, benefitting from the high
expertise gained in the home market
n = 174
1) Average for suppliers and manufacturers , based on average export share of all companies, not based on value of exports. Therefore varies from the
value mentioned in the management summary 2) Share of export of service providers was not surveyed in 2008
Source: SMTI 2010 61](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-61-2048.jpg)
![G INTERNATIONAL POSITIONING
A young product portfolio helps manufacturers to increase their
export share
EXPORT SHARE
[%, according to age of product portfolio, 2010] COMMENTS
• Suppliers and manufacturers realize different
82 export shares in respect to the age of their
Ø 78 product portfolio
72
• Suppliers
61
– Can export above-average mature
products
Ø 53
46 – Were faced with difficulties in capturing
export shares with young product
• Manufacturers
– Can capitalize on an innovative product
range in export markets
Younger PP1) – Tend to introduce new products or line
extensions faster abroad
Older PP2)
– Expect below avg. growth from mature
Suppliers Manufacturers products
n = 93
1) More than 50% of product portfolio younger than 5 years
2) More than 50% of product portfolio older than 5 years
Source: SMTI 2010 62](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-62-2048.jpg)
![G INTERNATIONAL POSITIONING
Growth expectations and export share – Positive correlation for
manufacturers, negative correlation for suppliers
EXPECTED GROWTH ACCORDING TO EXPORT SHARE
[%, only manufacturers and suppliers, 2010] COMMENTS
• Export is a “Must do” for growth
18
• Manufacturers are much stronger export
focused than suppliers
• Manufacturers with an above-average export
share
– expect significantly higher growth rates
10 – will outperform the market
Ø 10
– will outperform suppliers by far
7
• For suppliers, the situation is reversed – the
home market remains the key to growth.
4 They face difficulties to
– Overcome the external market access trap
Suppliers
– Find niches in a growing commodity market
Manufacturers
• By focusing on export, firms can
Low export share1) High export share2)
– Expand the potential customer base
– Drive regional account management
n = 114
1) Defined as less than 44%/81% of revenue from exports for suppliers/manufacturers
2) Defined as more than 44%/81% or revenue from exports for suppliers/manufacturers
Source: SMTI 2010 63](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-63-2048.jpg)









![4 INNOVATION AND SUPPORT
Share of newly introduced products and distribution of product
portfolio
SHARE OF SALES OF NEWLY INTRODUCED DISTRIBUTION OF PRODUCT PORTFOLIO
PRODUCTS (SINCE 2007) ACCORDING TO PRODUCT AGE
[% of answers, 2010] [% of answers, 2010]
28 28
27
Very small proportion 21
25
(0% - 10%) 18 24
23 23
22
Small proportion 36
(10% - 30%) 38
Medium proportion 21
(30% - 50%) 16
High proportion 16
(50% - 70%) 20
Very high proportion 6
(> 70%) 8
0-3 years 3-5 years 5-10 years `+10 years
Suppliers
Manufacturers
n = 127
Source: SMTI 2010 78](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-78-2048.jpg)
![4 INNOVATION AND SUPPORT
Share of inter-industry collaborations by partner
BY COMPANY CATEGORY [% of partners, 2010] BY COMPANY SIZE [% of partners, 2010]
88 94
Manufacturers/ Manufacturers/ 88
suppliers 81 suppliers 83
73
50
43 43
Universitites Universitites
59 55
91
13 9
IT service providers IT service providers 18
28 28
36
13
12 20
Support organizations Support organizations
24 14
27
9 16
Private research Private research 15
institutes 24 institutes 21
18 Micro
Suppliers 6 Small
9 8
Strategic consultants Manu- Strategic consultants Medium
4 facturers
7
9 Large
n = 105, multiple answers possible n = 105, multiple answers possible
Source: SMTI 2010 79](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-79-2048.jpg)
![4 INNOVATION AND SUPPORT
Share of functional collaborations
BY COMPANY CATEGORY [% by type, 2010] BY COMPANY SIZE [% by type, 2010]
43 54
Applied research Applied research 39 52
61 80
50
Quality control 45 Quality control 55
46 37
30
21
25 27
Basic research Basic research 37
35 40
21
12 24
Product certification Product certification 30
41
30
30 29
Marketing/sales Marketing/sales 21 33
20
17 15 25
Distribution Distribution
28 19 40
4
12 15
Training Training 19
13
20
7 Micro
After sales service 7 After sales service
Suppliers 7 Small
7 20
Manu- 4 Medium
Product launch 5 Product launch
7 facturers 3 11 Large
10
n = 105, multiple answers possible n = 105, multiple answers possible
Source: SMTI 2010 80](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-80-2048.jpg)

![4 INNOVATION AND SUPPORT
CTI/KTI initiatives are well known – The use of knowledge transfer
institutions is limited
KNOWLEDGE ABOUT CTI/KTI INNOVATION
PROMOTION PROGRAMS1) USE OF KNOWLEDGE AND TECHNOLOGY
[% of respondents that know CTI/KTI, 2010] TRANSFER INSTITUTIONS1) [%, 2010]
77 69
53
27
26 25
4
R&D Project Start-up Innovation Platform & No Yes Do not know any
Promotion Promotion Check Network Transfer Institutions
Promotion
n = 81, multiple answers possible n = 154
1) Suppliers and manufacturers
Source: SMTI 2010, CTI/KTI 82](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-82-2048.jpg)
![4 INNOVATION AND SUPPORT
Companies of all sizes use CTI/KTI services and support
DISTRIBUTION OF CTI/KTI USERS ACCORDING TO
COMPANY TURNOVER [%, 2010] COMMENTS
SUPPLIERS • If analyzed by company turnover, the
56 distribution of CTI/KTI users roughly
Ø 56
represents the total survey sample
32
• One major exception are manufacturers,
Ø 24 where companies with more than CHF 50m in
Ø 17
6 6 turnover are overrepresented
Ø3 – 50% of manufacturers that use CTI/KTI
programs have more than CHF 50m
CHF m <5 5-10 10-50 > 50
n = 16
turnover
– But only 23% of companies in the sample
have a turnover of more than CHF 50m
MANUFACTURERS 50
• However, these findings should not be over-
Ø 36
interpreted, as
19 19 Ø 23 Ø 23 – larger manufacturers usually have several
Ø 18 12 R&D projects running in parallel
– one CTI/KTI supported project therefore
only represents a fraction of all R&D
CHF m <5 5-10 10-50 > 50 projects
n = 16 – early CTI start-ups are not covered by this
Distribution of total SMTI sample, n = 105 report
Source: SMTI 2010, CTI/KTI 83](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-83-2048.jpg)
![4 INNOVATION AND SUPPORT
Distribution of CTI/KTI users according to company age shows
little deviation
DISTRIBUTION OF CTI/KTI USERS ACCORDING TO
COMPANY AGE1) [%, 2010] COMMENTS
61 • If analysed according to company age, the
Ø 61
distribution of CTI/KTI users shows little
deviation from the survey average
• Very young companies (< 5 years) account
for 15% of CTI/KTI users – slightly more
than their 12% share of the survey
• Young companies (5 to 10 years) make up
Ø 26 24 24% of CTI/KTI users – slightly less than
their 26% survey share
15
Ø 12 • Older companies (> 10 years) account for
61% of both CTI/KTI users and share of
overall survey participants
Very young Young Older
(< 5 years) (5 to 10 years) (> 10 years)
n = 41
= Distribution of total sample n = 137
1) Suppliers and manufacturers, CTI/KTI start-up support is not represented
Source: SMTI 2010, CTI/KTI 84](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-84-2048.jpg)
![5 GROWTH EXPECTATIONS
Growth expectations by segment and company size
GROWTH EXPECTATIONS BY SEGMENT GROWTH EXPECTATIONS BY COMPANY SIZE
[% of answers] [% of answers]
9 13
Suppliers Micro
11 14
13 9
Manufacturers Small
12 11
Tracers and 9 8
Medium
distributors 9 11
10 8
Service Providers Large
16 7
2010 2011
n = 215
Source: SMTI 2010 85](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-85-2048.jpg)
![6 SMTI REPORT DATABASE
The SMTI 2010 survey sample represents the whole industry
fairly well
Distribution of participating companies according to turnover
ACCORDING TO COMPANY TURNOVER ACCORDING TO COMPANY SIZE
[CHF] [number of employees]
Large
> 50m (250 and more)
8% 7%
Medium
(50 to 249) Micro
21%
11-50m
24% 40% (< 10)
68%
< 11m
32%
Small (10 to 49)
Source: SMTI 2010 87](https://image.slidesharecdn.com/swissmedicaltechnologyindustry2010report-110225052853-phpapp02/75/Swiss-medical-technology-industry-2010-report-87-2048.jpg)









