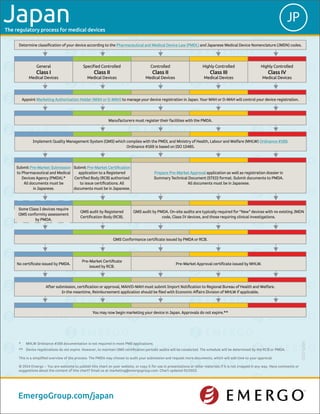
Japan medical device approval chart - Emergo
- 1. The regulatory process for medical devices You may now begin marketing your device in Japan. Approvals do not expire.** After submission, certification or approval, MAH/D-MAH must submit Import Notification to Regional Bureau of Health and Welfare. In the meantime, Reimbursement application should be filed with Economic Affairs Division of MHLW if applicable. No certificate issued by PMDA. Pre-Market Certificate issued by RCB. Pre-Market Approval certificate issued by MHLW. QMS Conformance certificate issued by PMDA or RCB. Some Class I devices require QMS conformity assessment by PMDA. QMS audit by Registered Certification Body (RCB). QMS audit by PMDA. On-site audits are typically required for “New” devices with no existing JMDN code, Class IV devices, and those requiring clinical investigations. Submit Pre-Market Submission to Pharmaceutical and Medical Devices Agency (PMDA).* All documents must be in Japanese. Submit Pre-Market Certification application to a Registered Certified Body (RCB) authorized to issue certifications. All documents must be in Japanese. Prepare Pre-Market Approval application as well as registration dossier in Summary Technical Document (STED) format. Submit documents to PMDA. All documents must be in Japanese. 5000-0115 * MHLW Ordinance #169 documentation is not required in most PMS applications. ** Device registrations do not expire. However, to maintain QMS certification periodic audits will be conducted. The schedule will be determined by the RCB or PMDA. This is a simplified overview of the process. The PMDA may choose to audit your submission and request more documents, which will add time to your approval. © 2014 Emergo – You are welcome to publish this chart on your website, or copy it for use in presentations or other materials if it is not cropped in any way. Have comments or suggestions about the content of this chart? Email us at marketing@emergogroup.com. Chart updated 01/2015. EmergoGroup.com/japan JPJapan Determine classification of your device according to the Pharmaceutical and Medical Device Law (PMDL) and Japanese Medical Device Nomenclature (JMDN) codes. General Class I Medical Devices Specified Controlled Class II Medical Devices Controlled Class II Medical Devices Highly Controlled Class III Medical Devices Highly Controlled Class IV Medical Devices Appoint Marketing Authorization Holder (MAH or D-MAH) to manage your device registration in Japan. Your MAH or D-MAH will control your device registration. Manufacturers must register their facilities with the PMDA. Implement Quality Management System (QMS) which complies with the PMDL and Ministry of Health, Labour and Welfare (MHLW) Ordinance #169. Ordinance #169 is based on ISO 13485.
- 2. The regulatory process for medical devices Device classification in Japan How long you should expect to wait after submission until approval is granted. (See note 1) Validity period for device registrations. (See note 2) Registration renewal should be started this far in advance. (See note 2) Complexity of the registration process for this classification. (See note 3) Overall cost of gaining regulatory approval. (See note 4) CLASS I General Medical Devices 1 month Does not expire 1 month Simple Complex Low High CLASS II Specified Controlled Medical Devices 3-5 months Does not expire 1 month Simple Complex Low High CLASS II Controlled Medical Devices 7-9 months Does not expire 1 month Simple Complex Low High CLASS III Highly Controlled Medical Devices 9-11 months Does not expire 1 month Simple Complex Low High CLASS IV Highly Controlled Medical Devices 13-16 months Does not expire 1 month Simple Complex Low High JPJapan EmergoGroup.com/japan NOTE 1: The time frames shown above are typical for the majority of medical device submissions but assume that your device does not contain animal tissue, medicinal substances or employ entirely novel technology. Your length of approval will depend on the quality and completeness of your technical documentation and how much time you take to address additional information requests from authorities after submission. YOUR SUBMISSION(S) MAY TAKE MORE TIME THAN WHAT IS SHOWN ABOVE. NOTE 2: Device registration does not expire, however you must pay Foreign Manufacturer Registration (FMR) renewal fees. Failure to renew FMR will result in your permission to market your device being revoked. NOTE 3: Our rating of the complexity of the registration process is based on our experience and the opinion of nearly 1,000 QA/RA professionals worldwide who were asked to rate the difficulty of registering a device in each country in January 2014. The European CE Marking process is considered the mid-point to which all other markets are compared. NOTE 4: Low = Less than US$5000; Midpoint = US$15000-$30000: High = More than US$50000. Overall cost includes registration application fees, product testing, in- country representation, submission preparation consulting and translation of registration documents but not IFU. Does not include cost of implementing or auditing a quality management system compliant with ISO 13485 or US FDA 21 CFR Part 820, if applicable. © 2014 Emergo Group – You are welcome to publish this chart on your website, or copy it for use in presentations or other materials if it is not cropped in any way. Have comments or suggestions about the content of this chart? Email us at marketing@emergogroup.com. Chart updated 01/2015.
