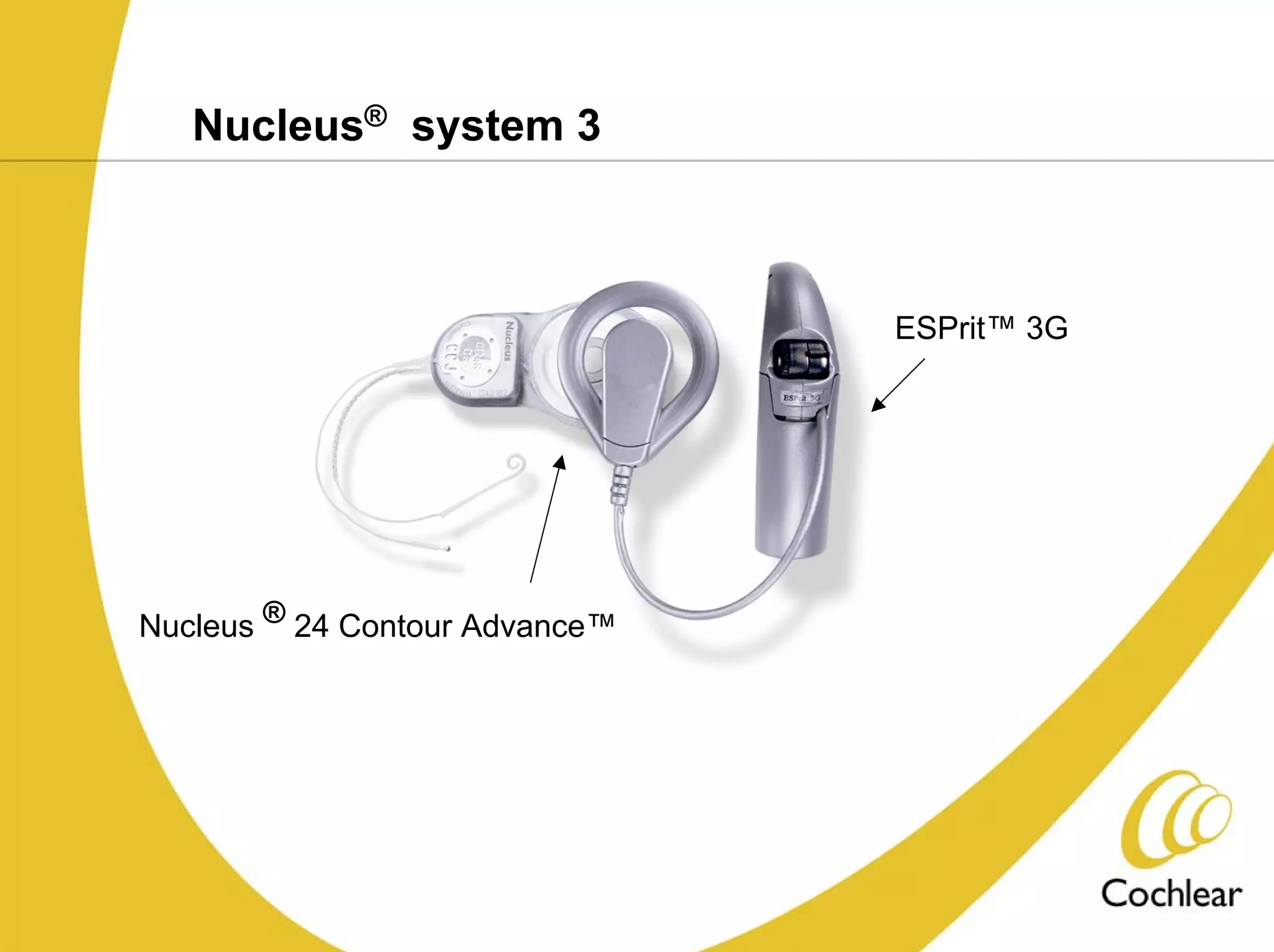
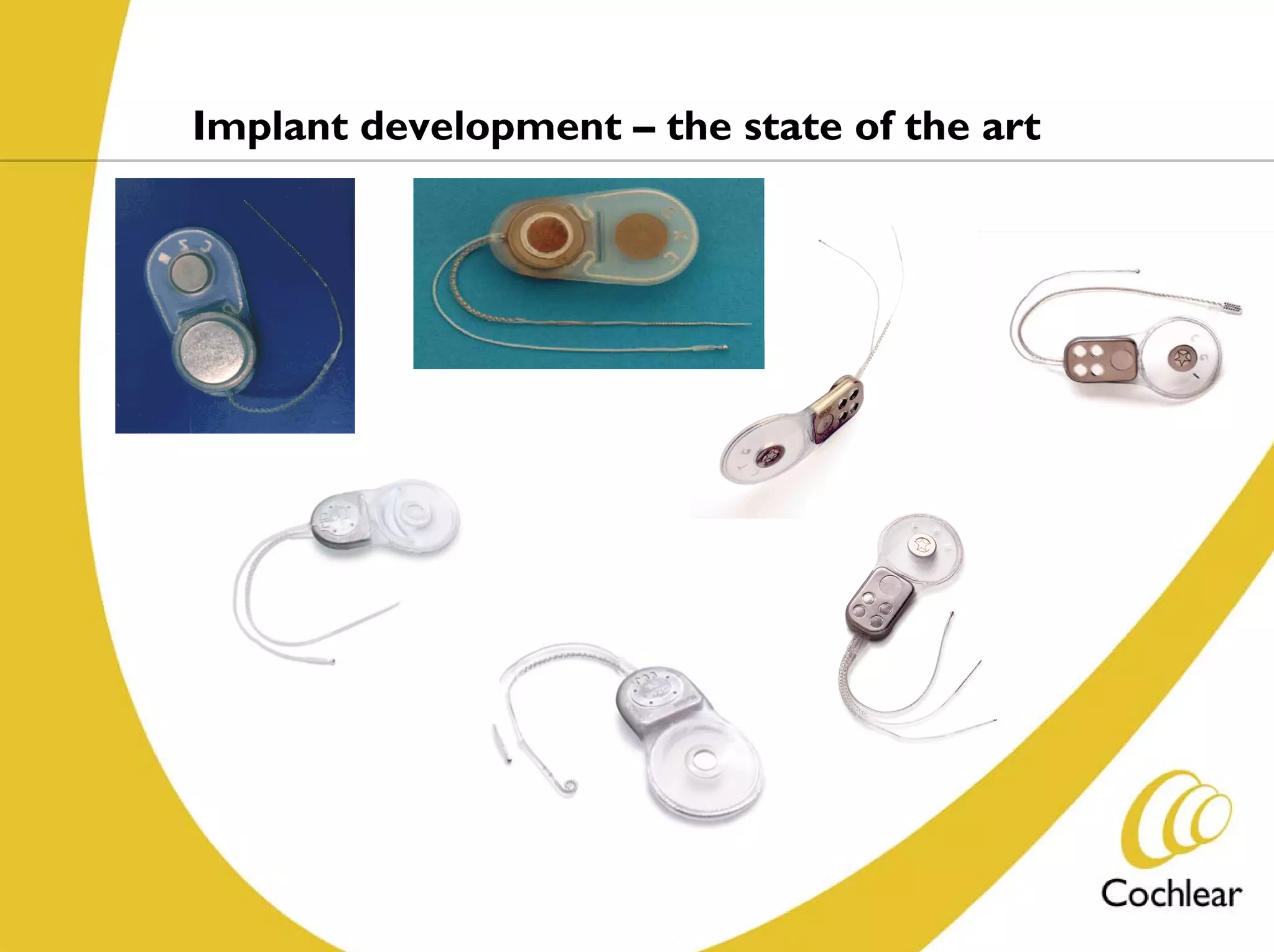
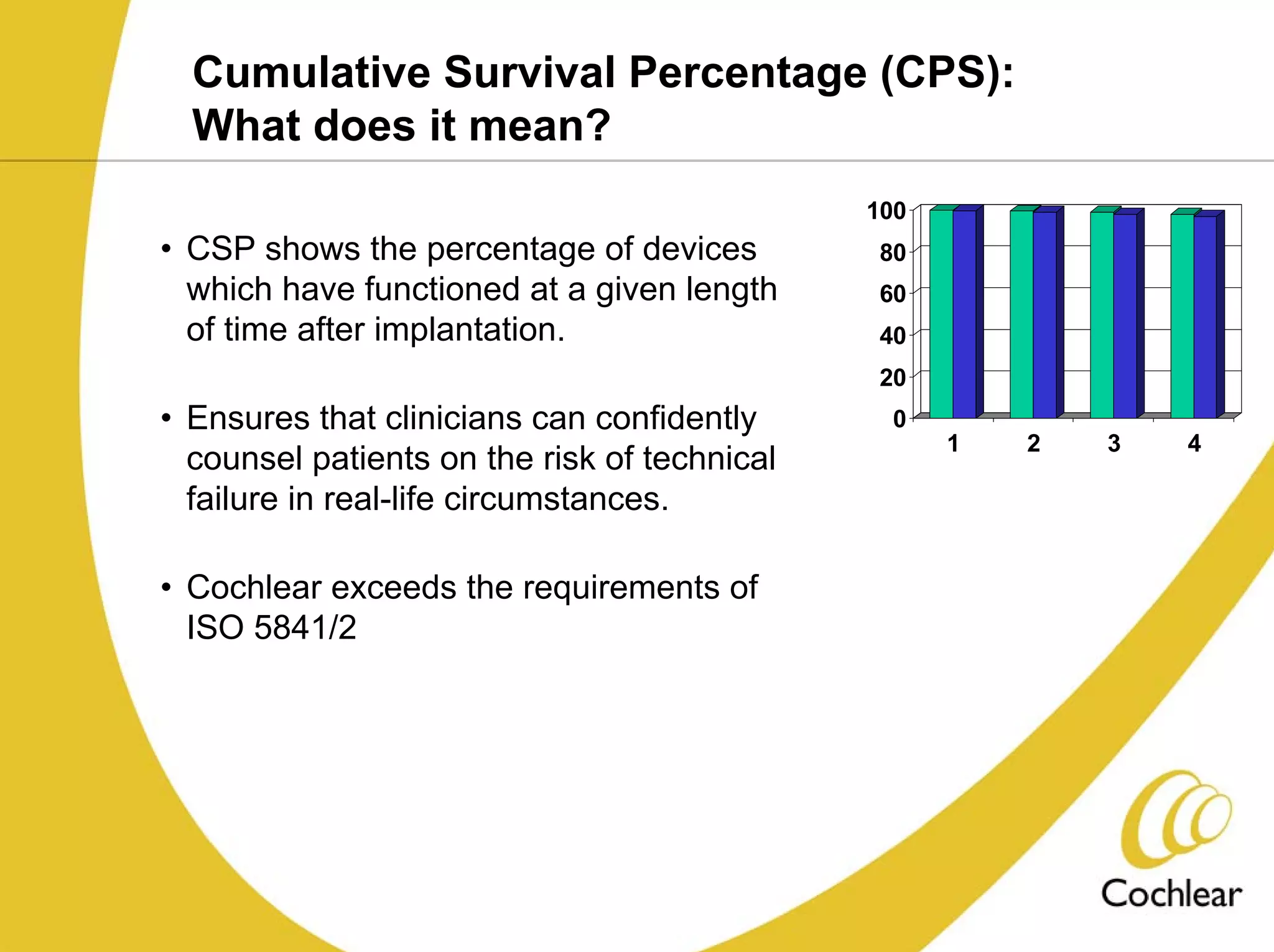
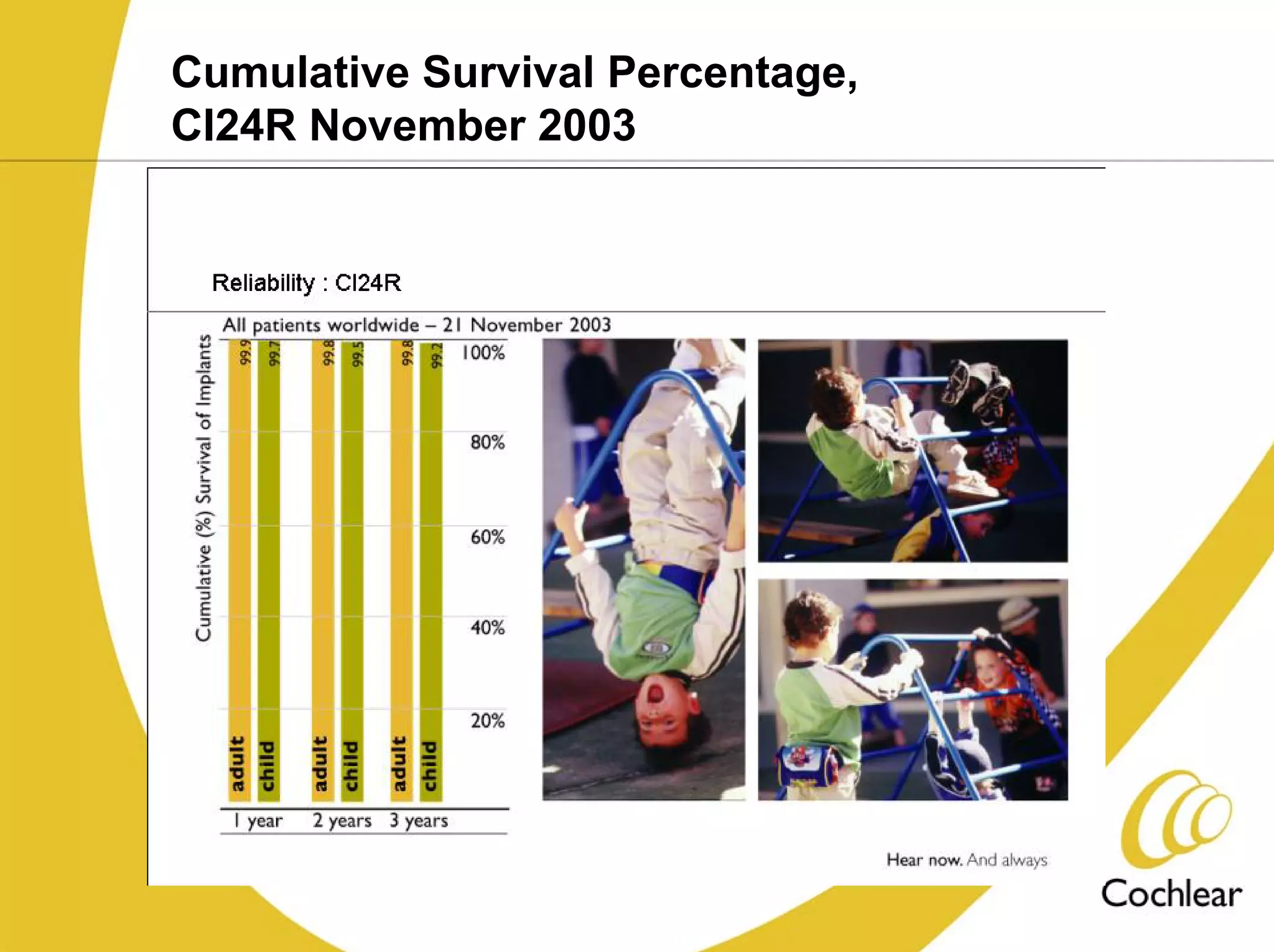
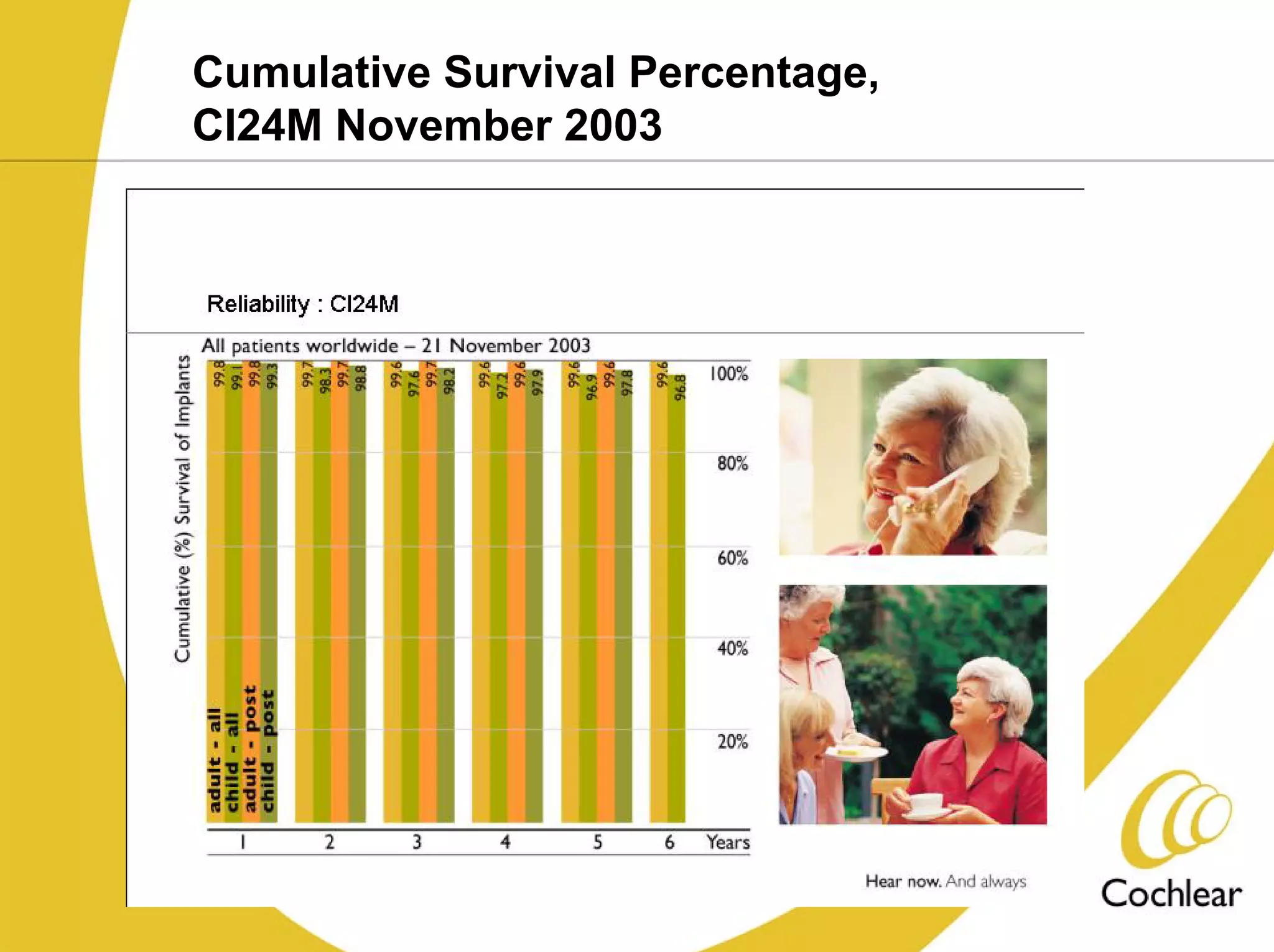
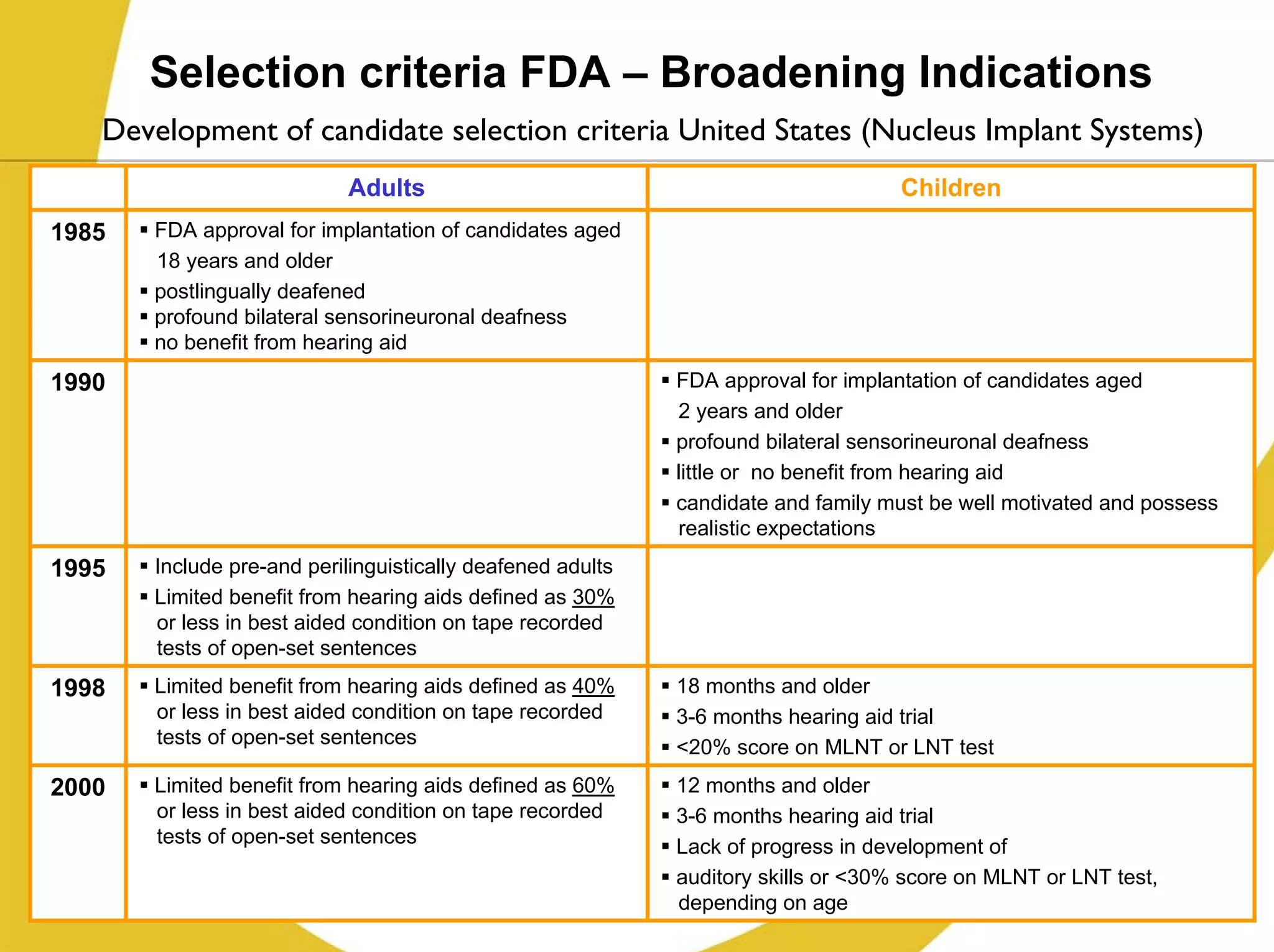
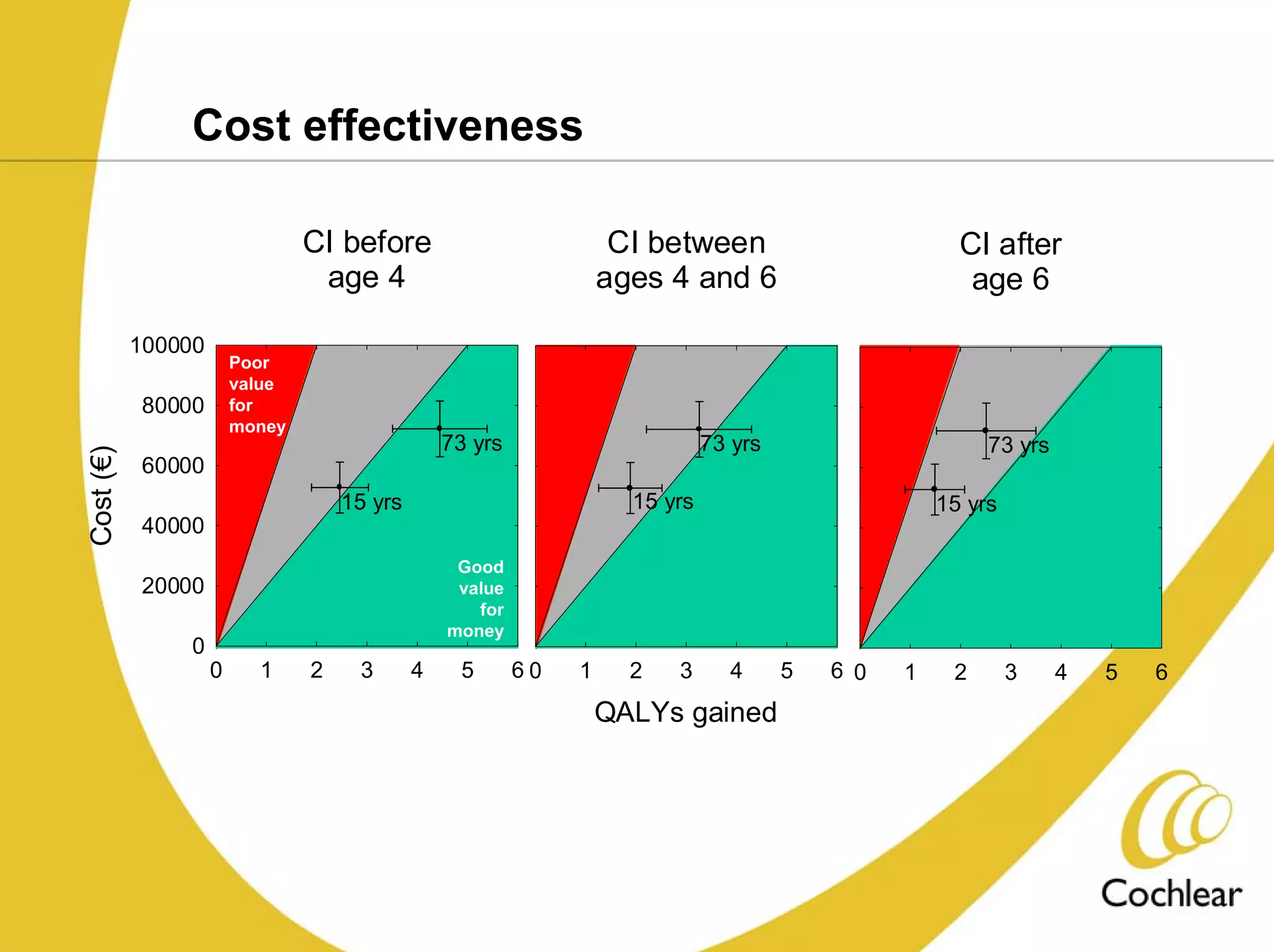
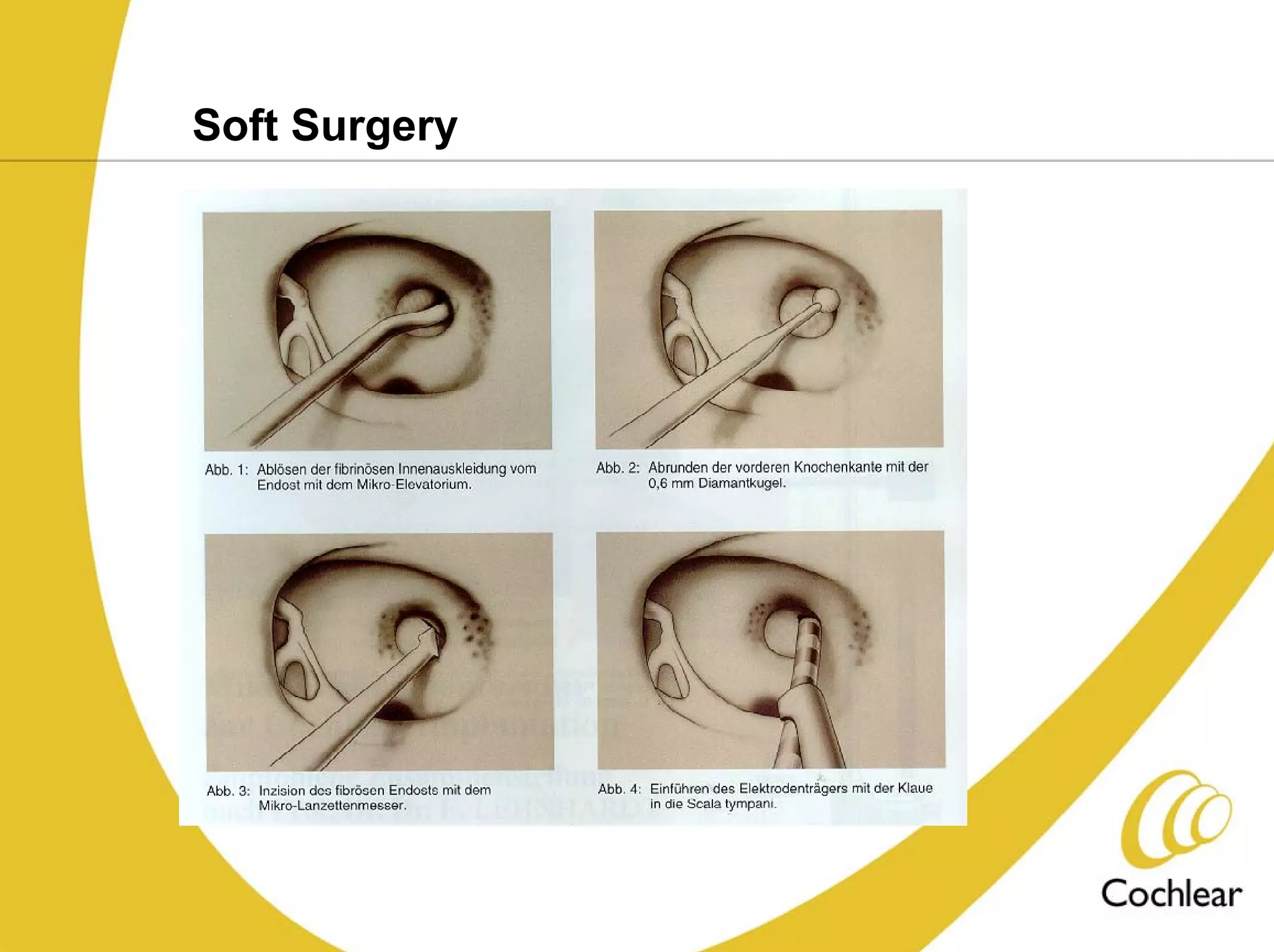
The document discusses cochlear implants, including their history and development. It covers improvements in implant technology and speech processors over time. It also discusses failures, categorizing them as either hard failures that require explantation, or soft failures that can be resolved through programming. Survival rates of different implant models are presented. The roles of early screening, appropriate candidacy criteria, rehabilitation approaches like auditory-verbal therapy, and parent participation are addressed. While cochlear implants provide benefits, the document suggests they should not be considered miracles.